Neural correlates of switching set as measured in fast, event-related functional magnetic resonance imaging - PubMed (original) (raw)
Neural correlates of switching set as measured in fast, event-related functional magnetic resonance imaging
Anna B Smith et al. Hum Brain Mapp. 2004 Apr.
Abstract
Attentional switching has shown to involve several prefrontal and parietal brain regions. Most cognitive paradigms used to measure cognitive switching such as the Wisconsin Card Sorting Task (WCST) involve additional cognitive processes besides switching, in particular working memory (WM). It is, therefore, questionable whether prefrontal brain regions activated in these conditions, especially dorsolateral prefrontal cortex (DLPFC), are involved in cognitive switching per se, or are related to WM components involved in switching tasks. Functional magnetic resonance imaging (fMRI) was used to examine neural correlates of pure switching using a paradigm purposely designed to minimize WM functions. The switching paradigm required subjects to switch unpredictably between two spatial dimensions, clearly indicated throughout the task before each trial. Fast, event-related fMRI was used to compare neural activation associated with switch trials to that related to repeat trials in 20 healthy, right-handed, adult males. A large cluster of activation was observed in the right hemisphere, extending from inferior prefrontal and pre- and postcentral gyri to superior temporal and inferior parietal cortices. A smaller and more caudal cluster of homologous activation in the left hemisphere was accompanied by activation of left dorsolateral prefrontal cortex (DLPFC). We conclude that left DLPFC activation is involved directly in cognitive switching, in conjunction with parietal and temporal brain regions. Pre- and postcentral gyrus activation may be related to motor components of switching set.
Copyright 2004 Wiley-Liss, Inc.
Figures
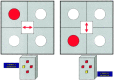
Figure 1
Illustration of switch task and correct response.
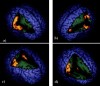
Figure 2
Regions of brain activation associated with switch contrasted with repeat trials. a: Whole brain cut away to reveal large activation cluster in the right hemisphere extending from inferior prefrontal lobe, pre‐ and post‐central gyri to middle temporal gyrus. b: Whole brain cut away to reveal activation cluster in the left hemisphere including DLPFC, post‐central gyrus, and superior temporal gyrus. c: Whole brain cut away to reveal activation in anterior cingulate gyrus and right hemispheric precentral and superior temporal activation (also seen in a). d: Whole brain cut away to reveal putamen activation and right hemisphere activation of post‐central gyrus (also seen in a).
References
- Alho K, Connolly JF, Cheour M, Lehtokoski A, Huotilainen M, Virtanen J, Aulanko R, Ilmoniemi RJ ( 1998): Hemispheric lateralization in preattentive processing of speech sounds. Neurosci Lett 258: 9–12. -PubMed
- Baker SC, Frith CD, Frackowiak RSJ, Dolan RJ ( 1996): The neural substrate of active memory for shape and spatial location in man. Cereb Cortex 6: 612–619. -PubMed
- Berg EA ( 1948): A simple objective technique for measuring flexibility of thinking. J Gen Psychol 39: 15–22. -PubMed
- Berman KF, Ostrem JL, Randolph C, Gold J, Goldberg TE, Coppola R, Carson RE, Herscovitch P, Weinberger DR ( 1995): Physiological activation of a cortical network during performance of the Wisconsin Card Sorting Test: a positron emission tomography study. Neuropsychologist 33: 1027–1046. -PubMed
- Botvinick M, Nystrom L, Fissell K, Carter CS, Cohen JD ( 1999): Conflict monitoring versus selection for action in anterior cingulate cortex. Nature 402: 179–181. -PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical