Long-term reinfection of the human genome by endogenous retroviruses - PubMed (original) (raw)
Long-term reinfection of the human genome by endogenous retroviruses
Robert Belshaw et al. Proc Natl Acad Sci U S A. 2004.
Abstract
Endogenous retrovirus (ERV) families are derived from their exogenous counterparts by means of a process of germ-line infection and proliferation within the host genome. Several families in the human and mouse genomes now consist of many hundreds of elements and, although several candidates have been proposed, the mechanism behind this proliferation has remained uncertain. To investigate this mechanism, we reconstructed the ratio of nonsynonymous to synonymous changes and the acquisition of stop codons during the evolution of the human ERV family HERV-K(HML2). We show that all genes, including the env gene, which is necessary only for movement between cells, have been under continuous purifying selection. This finding strongly suggests that the proliferation of this family has been almost entirely due to germ-line reinfection, rather than retrotransposition in cis or complementation in trans, and that an infectious pool of endogenous retroviruses has persisted within the primate lineage throughout the past 30 million years. Because many elements within this pool would have been unfixed, it is possible that the HERV-K(HML2) family still contains infectious elements at present, despite their apparent absence in the human genome sequence. Analysis of the env gene of eight other HERV families indicated that reinfection is likely to be the most common mechanism by which endogenous retroviruses proliferate in their hosts.
Figures
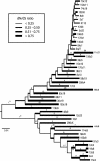
Fig. 1.
Bayesian estimate of the phylogenies for the internal region and LTRs (those of the same element are linked by lines). Posterior probabilities for the internal region are shown above the branches except (for clarity) in the human-specific clade. Internal branches are thickened if they were also recovered in a strict consensus of the most parsimonious trees. Thick terminal branches in the LTR tree represent 5′ LTRs, and thin terminal branches represent 3′ LTRs. Boxes represent elements whose 5′ and 3′ LTRs do not cluster together, with a thick box showing that they are polyphyletic in both Bayesian and MP trees, and a thin box showing that they are paraphyletic in both, or monophyletic in one. Filled circles denote elements containing the 292-bp deletion spanning the _pol_-env junction [Type I HERV-K(HML2) subgroup], which is absent in all other elements [Type II HERV-K(HML2) subgroup]. The clade created by segmental duplication of the host genome is also indicated, as are the branches indicating probable changes in the distribution of elements among primates.
Fig. 2.
Phylogenetic profile obtained by using
phylpro
. The darkened line shows the profile for sequence 12c22 only. Negative peaks for groups A and B (members of which have almost identical profiles) are indicated. The vertical dashed lines extend from the break-point positions identified by
lard
for 12c22 and the two other group A elements (HKY model, with κ and α estimated from neighbor joining trees for each triplet). P values are calculated from
seq-gen
using 100 replicates from a Bayesian tree (HKY model: κ, α and base frequencies taken from the tree). The window size in
phylpro
extends to include 100 variable positions either side of the sliding point. The ordinate shows the linear correlation score of uncorrected pairwise distances from the two windows.
Fig. 3.
Phylogeny of the internal region. The estimated number of stop codons acquired on each branch is shown (with R representing a reversal) and the _d_N/_d_S ratio is indicated by the line thickness. Superscript letters show the five potential acquisitions of stop codons on internal branches discussed in the text. Note that a short branch may represent insufficient changes for a reliable estimate to be made for that branch.
Similar articles
- High copy number in human endogenous retrovirus families is associated with copying mechanisms in addition to reinfection.
Belshaw R, Katzourakis A, Paces J, Burt A, Tristem M. Belshaw R, et al. Mol Biol Evol. 2005 Apr;22(4):814-7. doi: 10.1093/molbev/msi088. Epub 2005 Jan 19. Mol Biol Evol. 2005. PMID: 15659556 - Tracking the Fate of Endogenous Retrovirus Segregation in Wild and Domestic Cats.
Ngo MH, Arnal M, Sumi R, Kawasaki J, Miyake A, Grant CK, Otoi T, Fernández de Luco D, Nishigaki K. Ngo MH, et al. J Virol. 2019 Nov 26;93(24):e01324-19. doi: 10.1128/JVI.01324-19. Print 2019 Dec 15. J Virol. 2019. PMID: 31534037 Free PMC article. - HERV-W group evolutionary history in non-human primates: characterization of ERV-W orthologs in Catarrhini and related ERV groups in Platyrrhini.
Grandi N, Cadeddu M, Blomberg J, Mayer J, Tramontano E. Grandi N, et al. BMC Evol Biol. 2018 Jan 19;18(1):6. doi: 10.1186/s12862-018-1125-1. BMC Evol Biol. 2018. PMID: 29351742 Free PMC article. - Human endogenous retroviruses in the primate lineage and their influence on host genomes.
Mayer J, Meese E. Mayer J, et al. Cytogenet Genome Res. 2005;110(1-4):448-56. doi: 10.1159/000084977. Cytogenet Genome Res. 2005. PMID: 16093697 Review. - Human endogenous retroviruses: friend or foe?
Weiss RA. Weiss RA. APMIS. 2016 Jan-Feb;124(1-2):4-10. doi: 10.1111/apm.12476. APMIS. 2016. PMID: 26818257 Review.
Cited by
- The decline of human endogenous retroviruses: extinction and survival.
Magiorkinis G, Blanco-Melo D, Belshaw R. Magiorkinis G, et al. Retrovirology. 2015 Feb 2;12:8. doi: 10.1186/s12977-015-0136-x. Retrovirology. 2015. PMID: 25640971 Free PMC article. - Env-less endogenous retroviruses are genomic superspreaders.
Magiorkinis G, Gifford RJ, Katzourakis A, De Ranter J, Belshaw R. Magiorkinis G, et al. Proc Natl Acad Sci U S A. 2012 May 8;109(19):7385-90. doi: 10.1073/pnas.1200913109. Epub 2012 Apr 23. Proc Natl Acad Sci U S A. 2012. PMID: 22529376 Free PMC article. - Viral Metagenomic Profiling of Croatian Bat Population Reveals Sample and Habitat Dependent Diversity.
Šimić I, Zorec TM, Lojkić I, Krešić N, Poljak M, Cliquet F, Picard-Meyer E, Wasniewski M, Zrnčić V, Ćukušić A, Bedeković T. Šimić I, et al. Viruses. 2020 Aug 14;12(8):891. doi: 10.3390/v12080891. Viruses. 2020. PMID: 32824037 Free PMC article. - Integration targeting by avian sarcoma-leukosis virus and human immunodeficiency virus in the chicken genome.
Barr SD, Leipzig J, Shinn P, Ecker JR, Bushman FD. Barr SD, et al. J Virol. 2005 Sep;79(18):12035-44. doi: 10.1128/JVI.79.18.12035-12044.2005. J Virol. 2005. PMID: 16140779 Free PMC article. - Identification of endogenous retroviral reading frames in the human genome.
Villesen P, Aagaard L, Wiuf C, Pedersen FS. Villesen P, et al. Retrovirology. 2004 Oct 11;1:32. doi: 10.1186/1742-4690-1-32. Retrovirology. 2004. PMID: 15476554 Free PMC article.
References
- Boeke, J. D. & Stoye, J. P. (1997) in Retroviruses, eds. Coffin, J. M., Hughes, S. H. & Varmus, H. E. (Cold Spring Harbor Lab. Press, Plainview, NY), pp. 343–435. - PubMed
- International Human Genome Sequencing Consortium (2001) Nature 409, 860–921. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous