Detection of a sulfotransferase (HEC-GlcNAc6ST) in high endothelial venules of lymph nodes and in high endothelial venule-like vessels within ectopic lymphoid aggregates: relationship to the MECA-79 epitope - PubMed (original) (raw)
Detection of a sulfotransferase (HEC-GlcNAc6ST) in high endothelial venules of lymph nodes and in high endothelial venule-like vessels within ectopic lymphoid aggregates: relationship to the MECA-79 epitope
Annette Bistrup et al. Am J Pathol. 2004 May.
Abstract
The interaction of L-selectin on lymphocytes with sulfated ligands on high endothelial venules (HEVs) of lymph nodes results in lymphocyte rolling and is essential for lymphocyte homing. The MECA-79 monoclonal antibody reports HEV-expressed ligands for L-selectin by recognizing a critical sulfation-dependent determinant on these ligands. HEC-GlcNAc6ST, a HEV-localized sulfotransferase, is essential for the elaboration of functional ligands within lymph nodes, as well as the generation of the MECA-79 epitope. Here, we use an antibody against murine HEC-GlcNAc6ST to study its expression in relationship to the MECA-79 epitope. In lymph nodes, the enzyme is expressed in the Golgi apparatus of high endothelial cells, in close correspondence with luminal staining by MECA-79. In lymph node HEVs of HEC-GlcNAc6ST-null mice, luminal staining by MECA-79 is almost abolished, whereas abluminal staining persists although reduced in intensity. HEV-like vessels in several examples of inflammation-associated lymphoid neogenesis, including nonobese diabetic mice, also exhibit concomitant expression of the sulfotransferase and luminal MECA-79 reactivity. The correlation extends to ectopic lymphoid aggregates within the pancreas of RIP-BLC mice, in which CXCL13 is expressed in islets. Analysis of the progeny of RIP-BLC by HEC-GlcNAc6ST-null mice establishes that the enzyme is responsible for the MECA-79 defined luminal ligands.
Figures
Figure 1
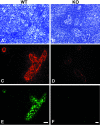
Staining of lymph nodes with an anti-HEC-GlcNAc6ST serum. An anti-HEC-GlcNAc6ST antiserum was produced by immunization of an individual rabbit with three peptides derived from the HEC-GlcNAc6ST sequence, as described in Materials and Methods. The serum was used to stain cryostat sections of mouse PN in the absence (B) or presence (D) of peptide 3 (RGKGMGQHAFHTNC). A and C show the bright-field images (hematoxylin stained) corresponding to B and D. Scale bar, 25 μm.
Figure 2
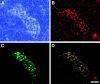
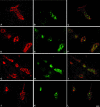
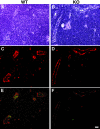
Expression of HEC-GlcNAc6ST and the MECA-79 epitope in mouse lymph node. Serial cryostat sections were prepared from PNs of wild-type (A, C, E) or −/− (B, D, F) mice. Sections were stained by immunofluorescence with MECA-79 (C, D) or the anti-HEC-GlcNAc6ST antibody (E, F). The bright-field images (A, B) establish the presence of HEVs in both wild-type and −/− lymph nodes. Scale bar, 25 μm.
Figure 3
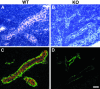
Golgi localization of HEC-GlcNAC6ST. A cryostat section from a rat lymph node was dual stained with an antibody for a Golgi marker (B; GM-130, Cy3, red) and the anti-HEC-GlcNAc6ST antibody (C; Cy2, green). D: Overlay of the two fluorescent images; yellow indicates overlap of the two stains. A is the bright-field image of the same field showing a single HEV. Scale bar, 25 μm.
Figure 4
Contribution of HEC-GlcNAC6ST to the generation of luminal and abluminal MECA-79 epitopes. Cryostat sections of PN were prepared from wild-type (A, C) and −/− (B, D) mice that had received intravenous injections of unlabeled MECA-79. The sections were stained first with Cy3-conjugated secondary reagents to detect injected luminally bound MECA-79 and then with biotinylated MECA-79 followed by streptavidin-Cy2 to detect abluminal MECA-79 epitopes. C and D: Overlay of the two fluorescent images. A and B show the corresponding bright-field images. Scale bar, 25 μm.
Figure 5
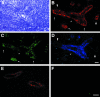
Expression of HEC-GlcNAc6ST in MNs and PPs. Cryostat sections from MNs of wild-type mice were stained simultaneously for MECA-79 (B; Cy3, red), HEC-GlcNAc6ST (C; Cy2, green), and MAdCAM-1 (D; aminomethylcoumarin, blue). Large arrowheads in B and D indicate vessels or segments of vessels that are MAdCAM-1-negative but contain luminal MECA-79 epitopes. Arrows in B and C indicate the correspondence between expression of HEC-GlcNAc6ST and luminal MECA-79 epitopes. Small arrowheads in B, C, and D indicate a segment of a vessel that is positive for MAdCAM-1, negative for HEC-GlcNAc6ST, and in which MECA-79 is confined to the luminal aspect. In E and F, a section of PP was dual stained for MECA-79 (E; Cy3, red) and HEC-GlcNAc6ST (F; Cy2, green). Scale bar, 25 μm.
Figure 6
Expression of HEC-GlcNAc6ST in HEV-like vessels at sites of inflammation. Cryostat sections from hyperplastic AKR thymus (A–C), NOD pancreas (D–F), NOD lacrimal glands (G–I), and NOD salivary glands (J–L) were dual stained for MECA-79 (A, D, G, J; Cy3, red) and HEC-GlcNAc6ST (B, E, H, K; Cy2, green). C, F, I, and L are the respective overlays. Scale bar, 25 μm.
Figure 7
Contribution of HEC-GlcNAc6ST to luminal MECA-79 epitopes and lymphoid aggregates in the pancreas of RIP-BLC mice. Cryostat sections from infiltrated pancreas of RIP-BLC/HEC-GlcNAc6ST+/+ (A, C, and E) and RIP-BLC/HEC-GlcNAc6ST−/− (B, D, and F) mice were dual stained for MECA-79 (Cy3, red) and HEC-GlcNAc6ST (Cy2, green). Shown are the bright-field images (A, B), the MECA-79 staining (C, D), and the merged MECA-79 and anti-HEC-GlcNAc6ST staining (E, F). Scale bar, 25 μm.
Similar articles
- A HEV-restricted sulfotransferase is expressed in rheumatoid arthritis synovium and is induced by lymphotoxin-alpha/beta and TNF-alpha in cultured endothelial cells.
Pablos JL, Santiago B, Tsay D, Singer MS, Palao G, Galindo M, Rosen SD. Pablos JL, et al. BMC Immunol. 2005 Mar 7;6:6. doi: 10.1186/1471-2172-6-6. BMC Immunol. 2005. PMID: 15752429 Free PMC article. - Lymphocyte-HEV interactions in lymph nodes of a sulfotransferase-deficient mouse.
van Zante A, Gauguet JM, Bistrup A, Tsay D, von Andrian UH, Rosen SD. van Zante A, et al. J Exp Med. 2003 Nov 3;198(9):1289-300. doi: 10.1084/jem.20030057. J Exp Med. 2003. PMID: 14597732 Free PMC article. - N-acetylglucosamine 6-O-sulfotransferase-1 regulates expression of L-selectin ligands and lymphocyte homing.
Uchimura K, Kadomatsu K, El-Fasakhany FM, Singer MS, Izawa M, Kannagi R, Takeda N, Rosen SD, Muramatsu T. Uchimura K, et al. J Biol Chem. 2004 Aug 13;279(33):35001-8. doi: 10.1074/jbc.M404456200. Epub 2004 Jun 2. J Biol Chem. 2004. PMID: 15175329 - Sulfated L-selectin ligands as a therapeutic target in chronic inflammation.
Uchimura K, Rosen SD. Uchimura K, et al. Trends Immunol. 2006 Dec;27(12):559-65. doi: 10.1016/j.it.2006.10.007. Epub 2006 Oct 17. Trends Immunol. 2006. PMID: 17049924 Review. - Sulphated endothelial ligands for L-selectin in lymphocyte homing and inflammation.
van Zante A, Rosen SD. van Zante A, et al. Biochem Soc Trans. 2003 Apr;31(2):313-7. doi: 10.1042/bst0310313. Biochem Soc Trans. 2003. PMID: 12653627 Review.
Cited by
- High endothelial venules as potential gateways for therapeutics.
Hussain B, Kasinath V, Ashton-Rickardt GP, Clancy T, Uchimura K, Tsokos G, Abdi R. Hussain B, et al. Trends Immunol. 2022 Sep;43(9):728-740. doi: 10.1016/j.it.2022.07.002. Epub 2022 Aug 3. Trends Immunol. 2022. PMID: 35931612 Free PMC article. Review. - Mechanism of action and efficacy of RX-111, a thieno[2,3-c]pyridine derivative and small molecule inhibitor of protein interaction with glycosaminoglycans (SMIGs), in delayed-type hypersensitivity, TNBS-induced colitis and experimental autoimmune encephalomyelitis.
Harris N, Koppel J, Zsila F, Juhas S, Il'kova G, Kogan FY, Lahmy O, Wildbaum G, Karin N, Zhuk R, Gregor P. Harris N, et al. Inflamm Res. 2016 Apr;65(4):285-94. doi: 10.1007/s00011-016-0915-4. Epub 2016 Jan 21. Inflamm Res. 2016. PMID: 26794621 - Induction of PNAd and N-acetylglucosamine 6-O-sulfotransferases 1 and 2 in mouse collagen-induced arthritis.
Yang J, Rosen SD, Bendele P, Hemmerich S. Yang J, et al. BMC Immunol. 2006 Jun 13;7:12. doi: 10.1186/1471-2172-7-12. BMC Immunol. 2006. PMID: 16772045 Free PMC article. - Lymphotoxin plays a crucial role in the development and function of nasal-associated lymphoid tissue through regulation of chemokines and peripheral node addressin.
Ying X, Chan K, Shenoy P, Hill M, Ruddle NH. Ying X, et al. Am J Pathol. 2005 Jan;166(1):135-46. doi: 10.1016/S0002-9440(10)62239-0. Am J Pathol. 2005. PMID: 15632007 Free PMC article. - Lymphatic vessels and tertiary lymphoid organs.
Ruddle NH. Ruddle NH. J Clin Invest. 2014 Mar;124(3):953-9. doi: 10.1172/JCI71611. Epub 2014 Mar 3. J Clin Invest. 2014. PMID: 24590281 Free PMC article. Review.
References
- Butcher EC, Picker LJ. Lymphocyte homing and homeostasis. Science. 1996;272:60–66. - PubMed
- Ruddle NH. Lymphoid neo-organogenesis: lymphotoxin’s role in inflammation and development. Immunol Res. 1999;19:119–125. - PubMed
- von Andrian UH, Mackay CR. T-cell function and migration. Two sides of the same coin. N Engl J Med. 2000;343:1020–1034. - PubMed
- Rosen SD. Ligands for L-selectin: homing, inflammation and beyond. Ann Rev Immunol. 2004;22:129–156. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R37 GM23547/GM/NIGMS NIH HHS/United States
- R01AI45073/AI/NIAID NIH HHS/United States
- R01 GM057411/GM/NIGMS NIH HHS/United States
- R01 AI045073/AI/NIAID NIH HHS/United States
- R01 CA016885/CA/NCI NIH HHS/United States
- R01 GM57411/GM/NIGMS NIH HHS/United States
- CA16885/CA/NCI NIH HHS/United States
- R01 DK057731/DK/NIDDK NIH HHS/United States
- R01 DK57731/DK/NIDDK NIH HHS/United States
- R37 GM023547/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous