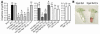Endocytic delivery of lipocalin-siderophore-iron complex rescues the kidney from ischemia-reperfusion injury - PubMed (original) (raw)
. 2005 Mar;115(3):610-21.
doi: 10.1172/JCI23056.
H Thomas Lee, Dana Rapoport, Ian R Drexler, Kirk Foster, Jun Yang, Kai M Schmidt-Ott, Xia Chen, Jau Yi Li, Stacey Weiss, Jaya Mishra, Faisal H Cheema, Glenn Markowitz, Takayoshi Suganami, Kazutomo Sawai, Masashi Mukoyama, Cheryl Kunis, Vivette D'Agati, Prasad Devarajan, Jonathan Barasch
Affiliations
- PMID: 15711640
- PMCID: PMC548316
- DOI: 10.1172/JCI23056
Endocytic delivery of lipocalin-siderophore-iron complex rescues the kidney from ischemia-reperfusion injury
Kiyoshi Mori et al. J Clin Invest. 2005 Mar.
Abstract
Neutrophil gelatinase-associated lipocalin (Ngal), also known as siderocalin, forms a complex with iron-binding siderophores (Ngal:siderophore:Fe). This complex converts renal progenitors into epithelial tubules. In this study, we tested the hypothesis that Ngal:siderophore:Fe protects adult kidney epithelial cells or accelerates their recovery from damage. Using a mouse model of severe renal failure, ischemia-reperfusion injury, we show that a single dose of Ngal (10 microg), introduced during the initial phase of the disease, dramatically protects the kidney and mitigates azotemia. Ngal activity depends on delivery of the protein and its siderophore to the proximal tubule. Iron must also be delivered, since blockade of the siderophore with gallium inhibits the rescue from ischemia. The Ngal:siderophore:Fe complex upregulates heme oxygenase-1, a protective enzyme, preserves proximal tubule N-cadherin, and inhibits cell death. Because mouse urine contains an Ngal-dependent siderophore-like activity, endogenous Ngal might also play a protective role. Indeed, Ngal is highly accumulated in the human kidney cortical tubules and in the blood and urine after nephrotoxic and ischemic injury. We reveal what we believe to be a novel pathway of iron traffic that is activated in human and mouse renal diseases, and it provides a unique method for their treatment.
Figures
Figure 1
Ngal expression in ATN of human (A–D) and mouse (E). (A) Monoclonal anti–human Ngal (Mo) and polyclonal anti–mouse Ngal (Po) antibodies recognized recombinant (21-kDa) and native (25-kDa) human and mouse Ngal. Occasionally, higher–molecular weight species (approximately 35 kDa and 66 kDa) were present in recombinant and native preparations; these might represent dimers and trimers of Ngal. A standard curve was constructed with 25, 5, 1, and 0.2 ng recombinant proteins on nonreducing gels. Human urine samples (0.1–20 μl) from patients with ATN showed high levels of Ngal, whereas samples from patients with chronic renal failure (CRF), patients with liver cirrhosis, hemochromatosis, or pancreatic carcinoma but lacking a renal diagnosis (Others), or normal subjects (Normal) had low levels of Ngal. (B) Similar data were obtained from human serum. (C and D) Geometric means (bar ± SD) of urinary (C) and serum (D) Ngal were compared in normal, CRF, and ATN groups. ATN was further divided into sepsis and nonsepsis. *P < 0.05, **P < 0.01, ***P < 0.001 vs. normal. (E) Mouse ATN was also associated with elevated urinary Ngal. The renal pedicle was cross-clamped for 30 minutes, and urine was collected at 24 hours of reperfusion and analyzed by immunoblot (5 μl/lane).
Figure 2
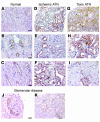
Ngal is expressed in cortical tubules in human acute renal failure. Ngal was detected with affinity-purified polyclonal antibody. (A) The normal kidney had little staining for Ngal. (B and C) At high power, focal staining of distal tubule cells (occupying 10% of the cortical area) and collecting ducts was found. There was no staining of proximal tubules. (D–I) Ischemic ATN caused by sepsis (D), by hypovolemia due to vomiting and diarrhea (E), or by heart failure (F), or nephrotoxic ATN caused by bisphosphonate (G), by cephalosporin (H), or by hemoglobinuria (I), produced intense staining of nearly 50% of the cortical tubules. Staining was heterogeneous and most intense in epithelial cells that displayed histologic features of cell injury, including simplification and enlarged reparative nuclei and prominent nucleoli. (J and K) In glomerular disease, Ngal was weakly expressed by crescents (J) and the proximal tubules of nephrosis (K). Scale bars: A, D, G, and K, 11 μm; B, C, E, F, and H–J, 5 μm.
Figure 3
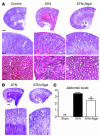
Rescue of mouse ATN by Ngal. (A) Holo-Ngal (100 μg) was injected into the peritoneum 15 minutes before renal pedicle cross-clamp and 30 minutes of ischemia. Kidneys were harvested after 24 hours of reperfusion for H&E staining. The ischemic kidneys (ATN) demonstrated loss of tubular nuclei (ATN, bottom) as well as the presence of cortical and medullary intratubular casts (ATN, middle). In contrast, Ngal pretreatment resulted in preservation of cortical tubules (ATN+Ngal, bottom) and reduced cortical-medullary casts (ATN+Ngal, middle). (B) PAS staining highlighted the luminal casts in the ischemic kidney (ATN) as well as the rescue of cortical tubules by pretreatment with Ngal (ATN+Ngal). (C) Area of proximal convoluted tubular necrosis was evaluated by the Jablonski scale, which demonstrated rescue of the ischemic cortex by Ngal (0: no necrosis; 1: isolated necrotic cells; 2: focal necrosis in inner cortex; 3: diffuse necrosis in inner cortex; 4: necrosis involving whole cortex). *P < 0.05, **P < 0.01 vs. untreated ischemic kidneys. Scale bars: A, top row, 800 μm; middle row, 24 μm; bottom row, 11 μm; B, top row, 800 μm; bottom row, 24 μm.
Figure 4
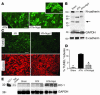
Correlates of ATN. Kidneys were harvested 24 hours after reperfusion. (A) N-cadherin staining was nearly abolished by ischemia-reperfusion (ATN), but it predominated apical cell-cell junctions after treatment (100 μg holo-Ngal; ATN+Ngal). (B) Full-length N-cadherin (130 kDa) was rescued by Ngal. Note the presence of N-cadherin fragments (28 kDa) in ischemia-reperfusion and sham-treated animals, but their suppression in Ngal-treated animals (arrow). In contrast, there was little change in the level of E-cadherin protein. GAPDH (38 kDa) was the loading control. (C) Tubules with TUNEL+ apoptotic cells (green fluorescence in ischemia-reperfusion injury; I/R) were reduced by pretreatment with Ngal (I/R+Ngal). TO-PRO-l was the nuclear counterstain (red) for the same field. (D) Percentage of tubules containing at least 1 apoptotic nucleus. Ischemia-reperfusion injury increased the number of positive tubules 22-fold, and Ngal reduced the activity fourfold. *P < 0.001 vs. I/R. (E) Ngal upregulated HO-1 (32 kDa) expression in ischemic kidneys. Kidneys harvested 24 hours after ischemia-reperfusion (ATN) expressed HO-1, but when animals were treated with Ngal, HO-1 expression was enhanced (ATN+Ngal). Purified HO-1 (HO-1) and rat cortex are included for comparison. GAPDH was the loading control. Scale bars: A and C, 9 μm; A, inset, 4.6 μm.
Figure 5
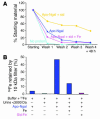
Clearance of Ngal. Ngal (100 μg) was introduced into the peritoneum, and serum (5 μl) and urine (1 μl) samples collected after the time indicated were analyzed by immunoblot.
Figure 6
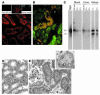
Clearance of Ngal by the proximal tubule. (A) Fluorescent Ngal (100 μg, labeled with Alexa 568) was introduced into the peritoneum, and after 1 hour the kidney was harvested and sectioned. Fluorescent Ngal was localized to large vesicles in the proximal tubule (bottom panel) but not in the glomerulus (G) or medulla (small top panels). Uncoupled dye did not label the kidney (Alexa 568 only). (B) Alexa 568–Ngal colocalized with FITC-dextran in S1 and S2 segments of the proximal tubule. Dextran was introduced into the animal 1 day before the Ngal injection in order to label lysosomes. (C) 125I-Ngal was introduced into mice, and the samples were assayed by SDS-PAGE. Lanes were loaded with 1,000 cpm each. The lane marked “Ngal” shows the initial preparation of 125I-Ngal. Subsequent lanes of urine or blood show loss of signal, suggesting degradation to small fragments. However, full-length Ngal and a 14-kDa fragment of Ngal were found in the kidney 1 and 5 hours after injection. (D and E) Radioautograph of kidney 1 hour after i.p. injection of 55Fe-loaded Ngal:siderophore. Radioactivity was not found in the medulla (D). In contrast, radioactive decay was found in the cortex and was associated with the apical zones of proximal tubule cells (E). Original magnification, A and B, ×40; A, inset, ×10. Scale bar: D and E, 2 μm.
Figure 7
Rescue of ATN by Ngal. (A) Plasma creatinine in mice subjected to 30 minutes of ischemia followed by 24 hours of reperfusion. The first panel shows that holo-Ngal (≥10 μg) from XL1-Blue bacteria (containing siderophore and iron) rescued renal function when introduced 15 minutes before ischemia or within 1 hour after ischemia (+ 1 h). However, Ngal was ineffective when administered later (+ 2 h). The second panel shows that apo-Ngal from BL21 bacteria (siderophore free) was minimally active, but that, when loaded with a siderophore (enterochelin), the protein was protective: both iron-free (apo-Ngal:Sid) and iron-loaded siderophores (apo-Ngal:Sid:Fe) had a protective effect. In comparison, the gallium-loaded complex (apo-Ngal:Sid:gallium) was ineffective, as was a single dose of the iron chelator DFO or the free siderophore (Sid). Retinol-binding protein (RBP), a lipocalin that is also filtered and reabsorbed by the proximal tubule, was ineffective. *P < 0.001 vs. ATN. #P < 0.01 vs. apo-Ngal:Sid (10 μg). The numbers in parentheses show the number of animals analyzed. (B) Preparations of Ngal. Ngal:Sid contains enterochelin, but not iron. Ngal:Sid:Fe contains enterochelin and iron.
Figure 8
Iron-binding cofactor in urine. (A) Buffer was mixed with 55Fe (No protein, light blue) and with apo-Ngal (dark blue), apo-Ngal plus siderophore (yellow), or apo-Ngal plus siderophore plus unlabeled iron (pink). The samples were then washed 3 times on a 10-kDa filter, and small aliquots were measured for retention of radioactivity (Washes 1–3). After 48 hours at 4°C the samples were washed again (Wash 4 + 48 h). Note the retention of 55Fe by apo-Ngal plus siderophore but not by apo-Ngal alone or apo-Ngal ligated by the iron-saturated siderophore, which demonstrates that an unsaturated siderophore is required for retention of 55Fe by Ngal. 55Fe binding to apo-Ngal plus siderophore was stable for 48 hours. (B) Urine (<3,000 Da) was mixed with 55Fe and with apo-Ngal, as indicated, and then washed 3 times on a 10-kDa filter. While urine (<3,000 Da) or apo-Ngal in buffer did not retain 55Fe on a 10-kDa filter, apo-Ngal plus urine retained 55Fe. 55Fe retention was blocked by the addition of excess iron citrate (Fe) or of iron-saturated enterochelin (Sid:Fe).
Similar articles
- Neutrophil gelatinase-associated lipocalin-mediated iron traffic in kidney epithelia.
Schmidt-Ott KM, Mori K, Kalandadze A, Li JY, Paragas N, Nicholas T, Devarajan P, Barasch J. Schmidt-Ott KM, et al. Curr Opin Nephrol Hypertens. 2006 Jul;15(4):442-9. doi: 10.1097/01.mnh.0000232886.81142.58. Curr Opin Nephrol Hypertens. 2006. PMID: 16775460 Review. - Neutrophil gelatinase-associated lipocalin, a siderophore-binding eukaryotic protein.
Borregaard N, Cowland JB. Borregaard N, et al. Biometals. 2006 Apr;19(2):211-5. doi: 10.1007/s10534-005-3251-7. Biometals. 2006. PMID: 16718606 Review. - Dual action of neutrophil gelatinase-associated lipocalin.
Schmidt-Ott KM, Mori K, Li JY, Kalandadze A, Cohen DJ, Devarajan P, Barasch J. Schmidt-Ott KM, et al. J Am Soc Nephrol. 2007 Feb;18(2):407-13. doi: 10.1681/ASN.2006080882. Epub 2007 Jan 17. J Am Soc Nephrol. 2007. PMID: 17229907 Review. - Amelioration of ischemic acute renal injury by neutrophil gelatinase-associated lipocalin.
Mishra J, Mori K, Ma Q, Kelly C, Yang J, Mitsnefes M, Barasch J, Devarajan P. Mishra J, et al. J Am Soc Nephrol. 2004 Dec;15(12):3073-82. doi: 10.1097/01.ASN.0000145013.44578.45. J Am Soc Nephrol. 2004. PMID: 15579510 - Identification of neutrophil gelatinase-associated lipocalin as a novel early urinary biomarker for ischemic renal injury.
Mishra J, Ma Q, Prada A, Mitsnefes M, Zahedi K, Yang J, Barasch J, Devarajan P. Mishra J, et al. J Am Soc Nephrol. 2003 Oct;14(10):2534-43. doi: 10.1097/01.asn.0000088027.54400.c6. J Am Soc Nephrol. 2003. PMID: 14514731
Cited by
- Angiotensin-II type 2 receptor-mediated renoprotection is independent of receptor Mas in obese Zucker rats fed high-sodium diet.
Patel SN, Kulkarni K, Faisal T, Hussain T. Patel SN, et al. Front Pharmacol. 2024 Jul 29;15:1409313. doi: 10.3389/fphar.2024.1409313. eCollection 2024. Front Pharmacol. 2024. PMID: 39135807 Free PMC article. - Blood and urine biomarkers of disease progression in IgA nephropathy.
Duan ZY, Zhang C, Chen XM, Cai GY. Duan ZY, et al. Biomark Res. 2024 Jul 29;12(1):72. doi: 10.1186/s40364-024-00619-4. Biomark Res. 2024. PMID: 39075557 Free PMC article. Review. - Renal Biomarkers in Heart Failure: Systematic Review and Meta-Analysis.
Kumar A, Chidambaram V, Geetha HS, Majella MG, Bavineni M, Pona PK, Jain N, Sharalaya Z, Al'Aref SJ, Asnani A, Lau ES, Mehta JL. Kumar A, et al. JACC Adv. 2024 Jan 5;3(2):100765. doi: 10.1016/j.jacadv.2023.100765. eCollection 2024 Feb. JACC Adv. 2024. PMID: 38939376 Free PMC article. - The Role of Biomarkers in Diagnosis of Sepsis and Acute Kidney Injury.
Ferreira GS, Frota ML, Gonzaga MJD, Vattimo MFF, Lima C. Ferreira GS, et al. Biomedicines. 2024 Apr 23;12(5):931. doi: 10.3390/biomedicines12050931. Biomedicines. 2024. PMID: 38790893 Free PMC article. Review. - Plasma proenkephalin and neutrophil gelatinase-associated lipocalin predict mortality in ICU patients with acute kidney injury.
Zhang M, Yang Y, Zhu L, Cui K, Zhang S, Xu Y, Jiang Y. Zhang M, et al. BMC Nephrol. 2024 May 22;25(1):181. doi: 10.1186/s12882-024-03611-0. BMC Nephrol. 2024. PMID: 38778257 Free PMC article.
References
- Lieberthal W, Nigam SK. Acute renal failure. II. Experimental models of acute renal failure: imperfect but indispensable. Am. J. Physiol. Renal Physiol. 2000;278:F1–F12. - PubMed
- Bonventre JV. Dedifferentiation and proliferation of surviving epithelial cells in acute renal failure. J. Am. Soc. Nephrol. 2003;14(Suppl. 1):S55–S61. - PubMed
- Halliwell B, Gutteridge JM. Role of free radicals and catalytic metal ions in human disease: an overview. Methods Enzymol. 1990;186:1–85. - PubMed
- McCord JM. Oxygen-derived free radicals in postischemic tissue injury. N. Engl. J. Med. 1985;312:159–163. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous