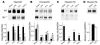Hemojuvelin is essential for dietary iron sensing, and its mutation leads to severe iron overload - PubMed (original) (raw)
Hemojuvelin is essential for dietary iron sensing, and its mutation leads to severe iron overload
Vera Niederkofler et al. J Clin Invest. 2005 Aug.
Abstract
Iron homeostasis plays a critical role in many physiological processes, notably synthesis of heme proteins. Dietary iron sensing and inflammation converge in the control of iron absorption and retention by regulating the expression of hepcidin, a regulator of the iron exporter ferroportin. Human mutations in the glycosylphosphatidylinositol-anchored protein hemojuvelin (HJV; also known as RGMc and HFE2) cause juvenile hemochromatosis, a severe iron overload disease, but the way in which HJV intersects with the iron regulatory network has been unclear. Here we show that, within the liver, mouse Hjv is selectively expressed by periportal hepatocytes and also that Hjv-mutant mice exhibit iron overload as well as a dramatic decrease in hepcidin expression. Our findings define a key role for Hjv in dietary iron sensing and also reveal that cytokine-induced inflammation regulates hepcidin expression through an Hjv-independent pathway.
Figures
Figure 1
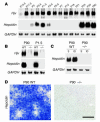
Hjv expression in periportal hepatocytes. (A) Targeting strategy used for homologous recombination in ES cells to eliminate Hjv gene function. The Hjv locus contains 3 coding exons (yellow). A targeting construct containing eGFP (dark gray) followed by IRES-NLS-lacZ-pA (blue) and thymidine kinase neomycin (TK-neo) (light gray) cassettes was integrated in frame into the second coding exon of Hjv. The probe used for genomic Southern blot analysis is indicated in blue. Integrated cassette is not drawn to scale. STOP, carboxyterminal stop codon. (B) Genomic Southern blot of Hjv+/+, Hjv+/–, and Hjv–/– genomic DNA using the probe indicated in A. (C) Northern blot analysis of total RNA isolated from P21 hindlimb muscles of Hjv+/+ and Hjv–/– mice probed for the expression of Hjv (top) and GAPDH (bottom). (D) Schematic drawing depicting the territories of liver lobules. Portal tracts (PT) are indicated in blue; central veins (CVs) are shown in red. Note that solid lines in D–G outline the hexagonally shaped hepatic lobule with PTs at the corners. (E–G) Detection of enzymatic lacZ activity in liver from 3-month-old Hjv+/– mice analyzed on vibratome (E) or cryostat (F and G) sections. Red circles indicate CV, blue circles indicate PT. Inset in (G) depicts high magnification of individual binuclear hepatocytes that express lacZ. (H–K) Immunohistochemical detection of HNF4α (H, J, and K: red), lacZ (I, J, and K: green), and SYTOX green (nuclei; K: blue) in liver from 3-month-old Hjv+/– mice. Arrows point to binuclear _Hjv_-expressing hepatocytes. Scale bar: 530 μm (E); 260 μm (F); 70 μm (G); 30 μm (inset in G); 40 μm (H–K)
Figure 2
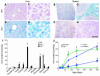
Iron accumulation in _Hjv-_mutant mice. (A–H) Histological detection of iron content on cryostat sections of liver (A–D) and spleen (E–H) of wild-type (A, C, E, and G) and Hjv–/– (B, D, F, and H) mice. Note uniform iron accumulation in the liver of 2.5-month-old _Hjv-_mutant mice and absence thereof in the red pulp of the spleen. (I) Quantitative determination of iron content (μmol/g dry weight) in various organs of 2.5-month-old wild-type (white), Hjv+/– (gray), and Hjv–/– (black) mice (n = 5 for each group). Asterisks indicate significant changes (P < 0.05) in Hjv–/– mice as compared with wild-type littermates. (J) Time course (P12–P300) of iron content (μmol/g dry weight) determined in Hjv–/– mice (squares) compared with pooled wild-type and Hjv+/– mice (triangles). Liver (green) and spleen (blue) are depicted in the graph. At least 3 animals per time point and genotype were included in the analysis. Asterisks indicate significant changes (P < 0.05) in Hjv–/– mice as compared with pooled wild-type and Hjv+/– littermates. Scale bar: 270 μm (A and B); 45 μm (C and D); 1.2 mm (E and F); 100 μm (G and H).
Figure 3
Lack of hepcidin expression in _Hjv-_mutant mice. (A) Developmental time course (E13.5–P90) of Hjv, hepcidin, and GAPDH expression levels as determined by Northern blot analysis on total RNA isolated from liver. (B) Northern blot analysis of total RNA isolated from adult (P90) or P1.5 liver of wild-type and Hjv–/– mice probed for the expression of Hjv, hepcidin, and GAPDH. (C) Northern blot analysis of total RNA isolated from adult (P90) liver of wild-type and Hjv–/– mice sacrificed 7 days after sham injection (S) or injection with iron-dextran (ID) and probed for the expression of hepcidin and GAPDH. Scale bar: 100 μm. (D) In situ hybridization on cryostat sections of liver isolated from adult (P90) wild-type and Hjv–/– mice probed for the expression of hepcidin.
Figure 4
Selective suppression of Hjv during inflammatory response. (A–C) Northern blot analysis of hepcidin (A), ferroportin (B), and Hjv (C) expression on total RNA isolated from liver of wild-type or _Hjv-_mutant mice. Before isolation of total RNA, mice were injected intraperitoneally with PBS (sham), LPS, IL-6, or TNF-α. At least 3 animals per experimental condition were analyzed, and 1 representative example is shown. Quantification of expression levels was performed by normalization of each sample to GAPDH expression probed sequentially on the same blots (data not shown). Histograms depict wild-type mice in black and _Hjv-_mutant mice in grey. Asterisks indicate significant changes (P < 0.05) in animals treated with LPS, IL-6, or TNF-α as compared with sham-injected animals of the same genotype. (D) Northern blot analysis of Hjv expression on total RNA isolated from skeletal muscle of wild-type mice after sham or LPS injection. Quantification was performed as described in (A–C). Histogram depicts sham-injected mice in black and LPS-injected mice in white.
Figure 5
An essential role for Hjv in the iron-sensing pathway. (A) Model depicting the dietary iron-sensing pathway in wild-type and _Hjv-_mutant mice. In wild-type mice, green arrows indicate responses in the presence of high iron; red arrows show responses in the presence of low iron. Balanced regulation of this pathway adjusts iron levels to the needs of the healthy organism (follow green or red arrows from left to right). In _Hjv-_mutant mice, iron-sensing is defective due to the absence of Hjv (indicated by gray double line). Despite high iron, this leads to an essentially complete absence of hepcidin expression, iron overload, and hemochromatosis (green arrows in Hjv mutant). Fpn, ferroportin. (B) Model depicting the impact of acute inflammation on the iron-sensing pathway (blue arrows). Acute-phase cytokines IL-6 and TNF-α act to coordinately downregulate Hjv expression in the liver while simultaneously inducing hepcidin expression. The reduction of Hjv results in a blockade of the dietary iron-sensing pathway (indicated by blue double line). This mechanism efficiently suppresses the iron-sensing pathway during the inflammatory response, which results in a low iron serum concentration inhibiting pathogenic growth.
Comment in
- Of mice and men: the iron age.
Vaulont S, Lou DQ, Viatte L, Kahn A. Vaulont S, et al. J Clin Invest. 2005 Aug;115(8):2079-82. doi: 10.1172/JCI25642. J Clin Invest. 2005. PMID: 16075054 Free PMC article.
Similar articles
- Conditional disruption of mouse HFE2 gene: maintenance of systemic iron homeostasis requires hepatic but not skeletal muscle hemojuvelin.
Gkouvatsos K, Wagner J, Papanikolaou G, Sebastiani G, Pantopoulos K. Gkouvatsos K, et al. Hepatology. 2011 Nov;54(5):1800-7. doi: 10.1002/hep.24547. Epub 2011 Aug 24. Hepatology. 2011. PMID: 21748766 - A mouse model of juvenile hemochromatosis.
Huang FW, Pinkus JL, Pinkus GS, Fleming MD, Andrews NC. Huang FW, et al. J Clin Invest. 2005 Aug;115(8):2187-91. doi: 10.1172/JCI25049. J Clin Invest. 2005. PMID: 16075059 Free PMC article. - Hfe and Hjv exhibit overlapping functions for iron signaling to hepcidin.
Kent P, Wilkinson N, Constante M, Fillebeen C, Gkouvatsos K, Wagner J, Buffler M, Becker C, Schümann K, Santos MM, Pantopoulos K. Kent P, et al. J Mol Med (Berl). 2015 May;93(5):489-98. doi: 10.1007/s00109-015-1253-7. Epub 2015 Jan 23. J Mol Med (Berl). 2015. PMID: 25609138 - Hemojuvelin: the hepcidin story continues.
Malyszko J. Malyszko J. Kidney Blood Press Res. 2009;32(2):71-6. doi: 10.1159/000208988. Epub 2009 Mar 14. Kidney Blood Press Res. 2009. PMID: 19287179 Review. - New insights into the regulation of iron homeostasis.
Deicher R, Hörl WH. Deicher R, et al. Eur J Clin Invest. 2006 May;36(5):301-9. doi: 10.1111/j.1365-2362.2006.01633.x. Eur J Clin Invest. 2006. PMID: 16634833 Review.
Cited by
- Iron metabolism: current facts and future directions.
Tandara L, Salamunic I. Tandara L, et al. Biochem Med (Zagreb). 2012;22(3):311-28. doi: 10.11613/bm.2012.034. Biochem Med (Zagreb). 2012. PMID: 23092063 Free PMC article. Review. - Effects of Excess Iron on the Retina: Insights From Clinical Cases and Animal Models of Iron Disorders.
Shahandeh A, Bui BV, Finkelstein DI, Nguyen CTO. Shahandeh A, et al. Front Neurosci. 2022 Feb 3;15:794809. doi: 10.3389/fnins.2021.794809. eCollection 2021. Front Neurosci. 2022. PMID: 35185447 Free PMC article. Review. - Physiology and Inflammation Driven Pathophysiology of Iron Homeostasis-Mechanistic Insights into Anemia of Inflammation and Its Treatment.
Lanser L, Fuchs D, Kurz K, Weiss G. Lanser L, et al. Nutrients. 2021 Oct 22;13(11):3732. doi: 10.3390/nu13113732. Nutrients. 2021. PMID: 34835988 Free PMC article. Review. - Iron and inflammation: cross-talk between pathways regulating hepcidin.
Fleming RE. Fleming RE. J Mol Med (Berl). 2008 May;86(5):491-4. doi: 10.1007/s00109-008-0349-8. J Mol Med (Berl). 2008. PMID: 18425494 No abstract available. - Iron-dependent regulation of hepcidin in Hjv-/- mice: evidence that hemojuvelin is dispensable for sensing body iron levels.
Gkouvatsos K, Fillebeen C, Daba A, Wagner J, Sebastiani G, Pantopoulos K. Gkouvatsos K, et al. PLoS One. 2014 Jan 7;9(1):e85530. doi: 10.1371/journal.pone.0085530. eCollection 2014. PLoS One. 2014. PMID: 24409331 Free PMC article.
References
- Hentze MW, Muckenthaler MU, Andrews NC. Balancing acts: molecular control of mammalian iron metabolism [review] Cell. 2004;117:285–297. - PubMed
- Andrews NC. Molecular control of iron metabolism. Best. Pract. Res. Clin. Haematol. 2005;18:159–169. - PubMed
- Nemeth E, et al. Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science. 2004;306:2090–2093. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases