Reduced cortical activity due to a shift in the balance between excitation and inhibition in a mouse model of Rett syndrome - PubMed (original) (raw)
Reduced cortical activity due to a shift in the balance between excitation and inhibition in a mouse model of Rett syndrome
Vardhan S Dani et al. Proc Natl Acad Sci U S A. 2005.
Abstract
Rett Syndrome (RTT) is a devastating neurological disorder that is caused by mutations in the MECP2 gene. Mecp2-mutant mice have been used as a model system to study the disease mechanism. Our previous work has suggested that MeCP2 malfunction in neurons is the primary cause of RTT in the mouse. However, the neurophysiological consequences of MeCP2 malfunction remain obscure. Using whole-cell patch-clamp recordings in cortical slices, we show that spontaneous activity of pyramidal neurons is reduced in Mecp2-mutant mice. This decrease is not caused by a change in the intrinsic properties of the recorded neurons. Instead, the balance between cortical excitation and inhibition is shifted to favor inhibition over excitation. Moreover, analysis of the miniature excitatory postsynaptic currents (mEPSCs)/inhibitory postsynaptic currents (mIPSCs) in the Mecp2-mutant cortex reveals a reduction in mEPSC amplitudes, without significant change in the average mIPSC amplitude or frequency. These findings provide the first detailed electrophysiological analysis of Mecp2-mutant mice and provide a framework for understanding the pathophysiology of the disease and tools for studying the underlying disease mechanisms.
Figures
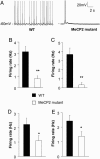
Fig. 1.
Spontaneous firing of L5 pyramidal neurons in S1 is reduced in Mecp2 mutants, compared with WT controls. (A) Representative spontaneous firing recordings at room temperature (25 ± 1°C) from a WT (Left) and _Mecp2_-mutant cell (Right). (B) Average spontaneous firing rate of WT and mutants. The ≈4-fold difference in mean firing rate of WT and mutant was statistically significant (Student's t test, P < 0.001). (C) Recordings performed at closer to physiological temperature (31-33°C) also showed a decreased firing rate in mutants. (D and E) Smaller reductions in spontaneous firing rate at presymptomatic ages. Mean firing rate was reduced ≈2-fold in presymptomatic mice at P 14-P 16 (D), or at P 21-P 24 (E). Statistical significance is indicated by * for P < 0.05 and ** for P < 0.01.
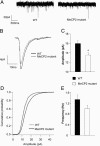
Fig. 2.
Intrinsic excitability of L5 neurons from Mecp2 mutants is unchanged. All recordings are from 4- to 5-week-old mice. Slices were continuously perfused in standard ACSF (at room temperature) containing APV, DNQX, and bicuculine. (A) Sample traces of spikes evoked by a depolarizing current step of 130 pA in a WT (Left) and a mutant (Right)L5 cell. (B) Mean firing rate vs. injected current amplitude (F-I curve). The mutant and WT F-I curves are almost identical. The mean first spike threshold (C) and input resistance (D) of mutant cells (n = 12) were not significantly different from WT cells (n = 12).
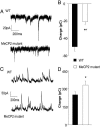
Fig. 3.
The balance between excitation and inhibition onto L5 pyramidal neurons is altered in Mecp2 mutants. Recordings were made in the presence of ongoing spontaneous activity. Total excitatory and inhibitory synaptic charge was calculated by integrating baseline subtracted spontaneous synaptic current. (A) Representative recordings of spontaneous excitatory postsynaptic currents (EPSCs) (recorded at the chloride-reversal potential) from L5 pyramidal neurons in slices from 4- to 5-week-old WT and mutant mice in modified ACSF. (B) The average excitatory charge is reduced in mutant mice (n = 12 for WT and mutants; P < 0.01, Student's t test) Representative recordings of spontaneous inhibitory postsynaptic currents (IPSCs) (recorded at the reversal potential for spontaneous EPSCs. (C) Representative recordings of spontaneous IPSCs (recorded at the reversal potential for spontaneous EPSCs. (D) The average inhibitory charge recorded from the same cells as in C is increased, compared with WT controls (n = 12 for WT and mutant, P < 0.05, t test).
Fig. 4.
Reduced amplitude of excitatory quantal transmission in L5 pyramids of 4- to 5-week-old _Mecp2_-mutant mice. mEPSCs recorded from L5 pyramidal cells in the presence of APV, bicuculine, and TTX. (A) Sample traces from WT and mutant cells voltage clamped at -70 mV. (B) Averaged mEPSC from 12 WT and 12 mutant cells. (C) mEPSC amplitude was reduced by 15%, and this difference was statistically significant (P < 0.01, t test). (D) Cumulative histograms of 300 events from each of 12 WT cells and 12 mutant cells showed a significant leftward shift in the mEPSC amplitude for mutant cells. (E) a small reduction in the mean mEPSC frequency was observed in mutants was not statistically significant (P = 0.12, t test).
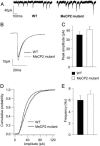
Fig. 5.
Subtle differences in unitary IPSCs in L5 pyramids of 4- to 5-week-old Mecp2 mutant mice. mIPSCs recorded from L5 pyramidal cells in the presence of APV, DNQX, and TTX. (A) Sample traces from WT and mutant cells voltage-clamped at -70 mV. (B) Averaged mIPSC from 17 WT and 17 mutant cells. The slightly increased mIPSC amplitude in mutant cells was not statistically significant across cells (C) (P = 0.23, t test), although the cumulative histograms of events from WT and mutant cells (D) showed a skew toward larger amplitudes that was highly significant. There was no significant change in the mean mIPSC frequency (E) and a small but significant change in the mean decay time constant (Table 1).
Similar articles
- Excitation/inhibition imbalance and impaired synaptic inhibition in hippocampal area CA3 of Mecp2 knockout mice.
Calfa G, Li W, Rutherford JM, Pozzo-Miller L. Calfa G, et al. Hippocampus. 2015 Feb;25(2):159-68. doi: 10.1002/hipo.22360. Epub 2014 Sep 25. Hippocampus. 2015. PMID: 25209930 Free PMC article. - Defective GABAergic neurotransmission in the nucleus tractus solitarius in Mecp2-null mice, a model of Rett syndrome.
Chen CY, Di Lucente J, Lin YC, Lien CC, Rogawski MA, Maezawa I, Jin LW. Chen CY, et al. Neurobiol Dis. 2018 Jan;109(Pt A):25-32. doi: 10.1016/j.nbd.2017.09.006. Epub 2017 Sep 18. Neurobiol Dis. 2018. PMID: 28927958 Free PMC article. - Jointly reduced inhibition and excitation underlies circuit-wide changes in cortical processing in Rett syndrome.
Banerjee A, Rikhye RV, Breton-Provencher V, Tang X, Li C, Li K, Runyan CA, Fu Z, Jaenisch R, Sur M. Banerjee A, et al. Proc Natl Acad Sci U S A. 2016 Nov 15;113(46):E7287-E7296. doi: 10.1073/pnas.1615330113. Epub 2016 Nov 1. Proc Natl Acad Sci U S A. 2016. PMID: 27803317 Free PMC article. - MeCP2 in neurons: closing in on the causes of Rett syndrome.
Caballero IM, Hendrich B. Caballero IM, et al. Hum Mol Genet. 2005 Apr 15;14 Spec No 1:R19-26. doi: 10.1093/hmg/ddi102. Hum Mol Genet. 2005. PMID: 15809268 Review. - Rett syndrome: of girls and mice--lessons for regression in autism.
Glaze DG. Glaze DG. Ment Retard Dev Disabil Res Rev. 2004;10(2):154-8. doi: 10.1002/mrdd.20030. Ment Retard Dev Disabil Res Rev. 2004. PMID: 15362175 Review.
Cited by
- Modeling psychiatric disorders at the cellular and network levels.
Brennand KJ, Simone A, Tran N, Gage FH. Brennand KJ, et al. Mol Psychiatry. 2012 Dec;17(12):1239-53. doi: 10.1038/mp.2012.20. Epub 2012 Apr 3. Mol Psychiatry. 2012. PMID: 22472874 Free PMC article. Review. - Rett syndrome: genes, synapses, circuits, and therapeutics.
Banerjee A, Castro J, Sur M. Banerjee A, et al. Front Psychiatry. 2012 May 8;3:34. doi: 10.3389/fpsyt.2012.00034. eCollection 2012. Front Psychiatry. 2012. PMID: 22586411 Free PMC article. - The enhancement of activity rescues the establishment of Mecp2 null neuronal phenotypes.
Scaramuzza L, De Rocco G, Desiato G, Cobolli Gigli C, Chiacchiaretta M, Mirabella F, Pozzi D, De Simone M, Conforti P, Pagani M, Benfenati F, Cesca F, Bedogni F, Landsberger N. Scaramuzza L, et al. EMBO Mol Med. 2021 Apr 9;13(4):e12433. doi: 10.15252/emmm.202012433. Epub 2021 Mar 5. EMBO Mol Med. 2021. PMID: 33665914 Free PMC article. - A mouse model for MeCP2 duplication syndrome: MeCP2 overexpression impairs learning and memory and synaptic transmission.
Na ES, Nelson ED, Adachi M, Autry AE, Mahgoub MA, Kavalali ET, Monteggia LM. Na ES, et al. J Neurosci. 2012 Feb 29;32(9):3109-17. doi: 10.1523/JNEUROSCI.6000-11.2012. J Neurosci. 2012. PMID: 22378884 Free PMC article. - Presynaptic GABA(B) receptors decrease neurotransmitter release in vestibular nuclei neurons during vestibular compensation.
Shao M, Reddaway R, Hirsch JC, Peusner KD. Shao M, et al. Neuroscience. 2012 Oct 25;223:333-54. doi: 10.1016/j.neuroscience.2012.07.061. Epub 2012 Aug 4. Neuroscience. 2012. PMID: 22871524 Free PMC article.
References
- Hagberg, B. (1997) Eur. Child Adolesc. Psychiatry 6, Suppl. 1, 2-4. - PubMed
- Amir, R. E., Van den Veyver, I. B., Wan, M., Tran, C. Q., Francke, U. & Zoghbi, H. Y. (1999) Nat. Genet. 23, 185-188. - PubMed
- Chen, R. Z., Akbarian, S., Tudor, M. & Jaenisch, R. (2001) Nat. Genet. 27, 327-331. - PubMed
- Guy, J., Hendrich, B., Holmes, M., Martin, J. E. & Bird, A. (2001) Nat. Genet. 27, 322-326. - PubMed
- Shahbazian, M., Young, J., Yuva-Paylor, L., Spencer, C., Antalffy, B., Noebels, J., Armstrong, D., Paylor, R. & Zoghbi, H. (2002) Neuron 35, 243-254. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases