The evolution of mutation rate in finite asexual populations - PubMed (original) (raw)
The evolution of mutation rate in finite asexual populations
Jean-Baptiste André et al. Genetics. 2006 Jan.
Abstract
In this article, we model analytically the evolution of mutation rate in asexual organisms. Three selective forces are present. First, everything else being equal, individuals with higher mutation rate have a larger fitness, thanks to the energy and time saved by not replicating DNA accurately. Second, as a flip side, the genome of these individuals is replicated with errors that may negatively affect fitness. Third, and conversely, replication errors have a potential benefit if beneficial mutations are to be generated. Our model describes the fate of modifiers of mutation rate under the three forces and allows us to predict the long-term evolutionary trajectory of mutation rate. We obtain three major results. First, in asexuals, the needs for both adaptation and genome preservation are not evolutionary forces that can stabilize mutation rate at an intermediate optimum. When adaptation has a significant role, it primarily destabilizes mutation rate and yields the emergence of strong-effect mutators. Second, in contrast to what is usually believed, the appearance of modifiers with large mutation rate is more likely when the fitness cost of each deleterious mutation is weak, because the cost of replication errors is then paid after a delay. Third, in small populations, and even if adaptations are needed, mutation rate is always blocked at the minimum attainable level, because the rate of adaptation is too slow to play a significant role. Only populations whose size is above a critical mass see their mutation rate affected by the need for adaptation.
Figures
Figure 1.
The average modifier's frequency, plotted as a function of the number of generations after appearance, according to analytical models (thin lines) and to stochastic individual-based simulations (thick lines). The generation of deleterious mutations affects the modifier's frequency; the cost of accuracy and the existence of adaptive mutations are not considered. Simulations are performed with a population size and are averaged over
runs. They are performed in two cases with different costs of deleterious mutations:
(thick solid line) and
(thick shaded line). The result of the analytical model neglecting the cost delay (Equation 14) is given by the dashed line. The results of the analytical model with delay (Equation 13) are given by the two thin lines, for each cost of deleterious mutation (
and 0.01, respectively, thin solid and thin shaded lines). Initial modifier's frequency is
; wild-type mutation rate is
; modifier's effect is
; and the deleterious genome has size
.
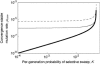
Figure 2.
Convergence stable mutation rate as a function of the fitness cost of each deleterious mutation in model A, under hypothesis 1 (advantageous mutations stronger than deleterious mutations, Equation 22). The size of the deleterious genome is , the frequency of selective sweeps is
, and the cost of replication accuracy is absent (
).
Figure 3.
Convergence stable mutation rate as a function of the rate of selective sweep in model A, under hypothesis 1 (advantageous mutations stronger than deleterious mutations, Equation 22). The size of the deleterious genome is ; the cost of deleterious mutations is
. The cost of replication accuracy is characterized by
(no cost of accuracy, thick line),
(thin line), or
(dashed line).
Figure 4.
Convergence stable mutation rate as a function of population size, in model B (Equation 26). The size of the deleterious genome is ; the size of the adaptive genome is
; the probability of fixation of adaptive mutations is
; the rate of environmental changes is
; and the cost of deleterious mutations is
. (a) The cost of accuracy is neglected (
); the population size has a threshold effect on convergence stable (CS) mutation rate. (b and c) The cost of accuracy is taken into account with
and
, respectively; the population size has a continuous and weak relative effect on CS mutation rate. Note that a, b, and c have different scales.
References
- Bachtrog, D., and I. Gordo, 2004. Adaptive evolution of asexual populations under Muller's ratchet. Evolution 58**:** 1403–1413. -PubMed
- Dawson, K. J., 1998. Evolutionarily stable mutation rates. J. Theor. Biol. 194**:** 143–157. -PubMed
- Dawson, K. J., 1999. The dynamics of infinitesimally rare alleles, applied to the evolution of mutation rates and the expression of deleterious mutations. Theor. Popul. Biol. 55**:** 1–22. -PubMed
- Dieckmann, U., 1997. Can adaptive dynamics invade? Trends Ecol. Evol. 12**:** 128–131. -PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources