Activation of dendritic cells via inhibition of Jak2/STAT3 signaling - PubMed (original) (raw)
Activation of dendritic cells via inhibition of Jak2/STAT3 signaling
Yulia Nefedova et al. J Immunol. 2005.
Abstract
Signaling via Jak2/STAT3 is critically important for normal dendritic cell (DC) differentiation. In addition, we have previously demonstrated that hyperactivation of the Jak2/STAT3 pathway induced by tumor-derived factors (TDF) may be responsible for abnormal DC differentiation in cancer. In this study, using a novel selective inhibitor of Jak2/STAT3, JSI-124, we investigated the mechanism of the Jak2/STAT3 effect on DCs and the possibility of pharmacological regulation of DC differentiation in cancer. Our experiments have demonstrated that JSI-124 overcomes the differentiation block induced by TDF and promotes the differentiation of mature DCs and macrophages. Surprisingly, inhibition of Jak2/STAT3 signaling resulted in dramatic activation of immature DCs generated in the presence of TDF as well as in control medium. This activation manifested in up-regulation of MHC class II, costimulatory molecules, and a dramatic increase in the ability to stimulate allogeneic or Ag-specific T cells. Inhibition of Jak2/STAT3 signaling resulted in activation of the transcription factor NF-kappaB. This up-regulation was not due to a conventional pathway involving IkappaB alpha, but was probably due to a block of the dominant negative effect of STAT3. This indicates that Jak2/STAT3 play an important role in negative regulation of DC activation, and pharmacological inhibition of the Jak2/STAT3 pathway can be used to enhance DC function.
Figures
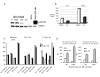
Figure 1.
JSI-124 decreases the levels of phospho-STAT3 and promotes DC differentiation. (A) DC were generated from HPCs in the presence of tumor cell (CT26) CM for 5 days. CD11c+ DCs were isolated and treated with different concentrations of JSI-124 for 24 hrs. Proteins were evaluated by Western Blotting. HeLa cells treated with IFN-α were used as positive control for STAT3 activation. (B) HPCs were grown in the presence of control (3T3) or tumor cell (CT26) CM for 5 days. CD11c+ DCs were isolated and treated with 0.5 μM JSI-124 or vc for 24 hrs. Intensity of specific bands was quantitated using ImageQuant software. Intensity of the signals from pSTAT3 was normalized to the one from total STAT3. (No differences in the level of total STAT3 between the cells were observed). (C) HPCs were isolated from mice BM and cultured in medium alone (No CM) or in the presence of 3T3 or CT26 CM. Cells were treated with JSI-124 or DMSO (vc) starting from day 3. On day 7 cells were collected. The proportions of different cell populations were calculated. Mean results of four experiments are shown. (D) CD11c+ cells from the experiment described in Fig. 1B were gated and the level of expression of MHC class II (IAd) and B7-2 (CD86) molecules was evaluated. Mean results of four experiments are shown.
Figure 2.
Jak/STAT3 pathway inhibitor JSI-124 activates DCs. DCs were generated from mice BM cells in the presence of control 3T3 or CT26 CM. CD11c+ DCs were isolated using magnetic beads separation technique. Cells were then treated with 0.5 μM JSI-124 or vc, and cultured again with 3T3 or CT26 CM. On day 4 cells were collected and their phenotype and function was evaluated. The level of surface markers expression was evaluated by flow cytometry. Mean results of three performed experiments (A) and typical example (B) are shown. _MFI_- mean fluorescence intensity. Dotted line represents CD11c+ DCs cultured for 4 days with DMSO, solid line − treated with JSI-124. Expression of MHC class II (IAd), CD86, and CD40 molecules was evaluated by flow cytometry as described in Materials and Methods. Cells were also used as stimulators of allogeneic T cells in MLR (C) and syngenic T cells in the presence of HA peptide (D). Two experiments with the same results were performed. (E) - DC ability to process antigen was evaluated by uptake of FITC-Dextran. _MFI_- mean fluorescence intensity. The level of FITC-dextran uptake at 4°C was subtracted from uptake at 37°C. Mean results of three performed experiments are shown. (F). DCs were generated from bone marrow HPC using GM-CSF and IL-4. CD11c+ cells were isolated and treated for 4 days with either 0.5 μM JSI-124 or DMSO (vc). After that time DCs were pulsed with 10 μg/ml of H2Kb matched peptide (IYSTVASSL). Naïve BALB/c mice were immunized s.c. with 2×105 DCs. Immunization was repeated once 10 days later. Seven days after the second immunization mice were sacrificed, splenocytes isolated and stimulated with either control (C.P.) or specific peptide (S.P.). The number of IFN-γ producing cells was measured in ELISPOT assay. Each group included 3 mice and each experiment was performed in quadruplicates. Naïve − non-immunized mice. Mean ± St. Dev. are shown. (G, H) Human DCs were generated in vitro from peripheral blood MNC with GM-CSF and IL-4 and purified by Metrizamide gradient centrifugation. Cells were then treated with 0.5 μM JSI-124 or DMSO (vc) for 5 days. (G) After that DCs were collected and labeled with PE-conjugated lineage cocktail antibody (Anti-CD3, CD14, CD19, CD56), PerCP-conjugated anti-HLA-DR antibody, and FITC-conjugted anti-CD86, CD40, CD83 antibodies. Cells were analyzed by flow cytometry (FACSCalibur). Fluorescence intensity was measured within the population of lin- HLA-DR+ DCs. _MFI_- mean fluorescence intensity. (H) DCs treated with JSI-124 or DMSO (vc) were mixed in triplicates in U-bottom 96-well plates at different ratios with 105 MNC from a different donor. Cells were incubated for 5 days. 3H-Thymidine was added 18 hr before cell harvesting. cpm − count per minute. Mean results of two experiments are shown. Spontaneous proliferation of MNC was less than 3,000 CPM.
Figure 3.
Activated by JSI-124 DCs exert their effect via direct cell-cell contact. (A) RANTES concentration was measured in triplicates in supernatants obtained after 48 hr treatment of CD11c+ DC with JSI-124 or VC as described in Material and Methods. (B) Splenocytes were isolated from syngeneic BALB/c mice and 2×105 cells were placed in the bottom chamber of U-bottom 96-well Transwell plates. Cells were stimulated with 0.5 μg/ml anti-CD3 antibody. DCs treated with DMSO (vc) or JSI-124 (JSI) were added either to the bottom chamber (2×103 cells per well) (mix) or to upper chamber (4×103 cells per well) (transwell). 3H-thymidine was added 18 hr prior to the cell harvest and its incorporation was measured on scintillation counter as described in Material and Methods. All experiments were performed in triplicates and repeated at least once to assure reproducibility of the results. (C) LN cells were isolated from HA-TCR transgenic mice and were placed into the bottom chamber of Transell wells (105 cells per well). Cells were stimulated with 12.5 μg/ml specific HA-derived peptide. DCs were placed into the bottom chamber (103 per well) or into the upper chamber (2×103) of the Transwell. After 4-day culture upper chamber was removed and cells were were pulsed for 1 hr with BrdU. The proportion of CD3+ T cells in S phase of cell cycle was evaluated by flow cytometry. Each experiment was performed in triplicates. Two experiments with the same results were performed.
Figure 4.
JSI-124 induces translocation of MHC class II to the surface of DCs. Murine BM HPCs were cultured with GM-CSF and IL-4 to generate DCs. After 5-day culture CD11c+ cells were isolated using magnetic beads separation technique and were then treated with 0.5 μM JSI-124 or DMSO (vc). (A) Total RNA was isolated from cells cultured for 24 and 48 hrs followed by RT-PCR as described in Material and Methods. (B) Cells were collected after 48 hrs in culture and Western blotting was performed using I-A/I-E antibody as described in Materials and Methods. (C) After 48 hrs in culture, cells were collected to cytospin slides. Slides were fixed, and stained with either isotype control IgG (control) or biotinylated anti-I-A/I-E antibody followed by streptavidin-FITC and slides were analyzed by confocal microscopy (x1000).
Figure 5.
Inhibition of Jak2/STAT3 signaling activate NF-κB in DCs. CD11c+DCs were generated as described in Fig. 4 and treated with either JSI-124 or DMSO (vc). (A) DCs were collected at time point indicated and EMSA with NF-κB specific probe was performed. (B) CD11c+ cells were co-transfected with NF-κB or control plasmid and plasmid carrying Renilla luciferase gene, and then treated with JSI-124 or DMSO (vc) for 36 hrs. Values of luciferase activity (LA) for specific and control plasmid were normalized to Renilla LA. Data presented is the fold increase of LA in cells transfected with plasmid containing NF-κB−responsive element over the LA in cells transfected with control plasmid. Mean results of three performed experiments are shown. (C) DCs were treated with 5 μg/ml LPS for 15-30 min. The level of IkBα was evaluated by Western blotting as described in Material and Methods; (D) - Level of IκBα in DCs after 30 min and 60 min of treatment with JSI-124; (E) - DCs were treated with JSI-124 or DMSO (vc) for 18 hr and 36 hr, whole cell lysates were prepared and the levels of different NF-κB subunits and IκBα was determined in Western blotting.
Figure 6.
Mechanism of JSI-124 induced activation of NF-κB in DCs.
(A) CD11c± DCs were generated as described above and cells were stimulated with 20 ng/ml TNFα for 40 min. Immunoprecipitation was performed with anti-STAT3 antibody or control rabbit IgG as described in Material and Methods. Membranes were then probed with antibodies against cRel and STAT3. (B) CD11c± DCs treated with JSI-124 or DMSO (VC) for 36 hrs were collected, cell lysates were prepared. STAT3 was precipitated using anti-STAT3 antibody. Membranes were then probed with antibodies against cRel, p65, and STAT3. (C) CD11c±DCs were generated as described in Fig. 4 and treated for 20 min with 20 ng/ml TNFα (T). In control (C) cells were cultured in medium alone. Nuclear extracts were prepared and EMSA was performed with NF-κB specific probe in the presence of 5 μg of rabbit IgG or rabbit polyclonal antibodies against p65 or cRel (Santa Cruz). Arrow points on the place of supershift. Cold inhibition − EMSA performed in the presence of x100 excess of unlabeled probe. Probe only − EMSA performed without nucleoproteins. (D). CD11c±DCs were treated with either JSI-124 or DMSO (VC) for 48 hr. Nuclear extracts were prepared and EMSA was performed as described above. Arrow points on the place of supershift. Cold inhibition − EMSA performed in the presence of x100 excess of unlabeled probe. Probe only − EMSA performed without nucleoproteins.
(E, F) DCs were generated from BM HPCs obtained from cRel-/- or control cRel+/+ mice. CD11c+ cells were isolated using magnetic beads separation technique and treated with 0.5 μM JSI-124 (solid line) or VC (dotted line) for 5 days. Cells then were collected and expression of MHC class II and co-stimulatory molecules was analyzed by flow cytometry. Typical example (E) and cumulative results (F) of three performed experiments are shown. In the bottom row labeled “cRel-/- LPS 24 hr”, DCs generated from cRel-/- HPCs were isolated using CD11c marker and were immediately activated for 24 hr with 5 μg/ml LPS. In that case solid line represent cells treated with LPS.
Similar articles
- Regulation of dendritic cell differentiation and antitumor immune response in cancer by pharmacologic-selective inhibition of the janus-activated kinase 2/signal transducers and activators of transcription 3 pathway.
Nefedova Y, Nagaraj S, Rosenbauer A, Muro-Cacho C, Sebti SM, Gabrilovich DI. Nefedova Y, et al. Cancer Res. 2005 Oct 15;65(20):9525-35. doi: 10.1158/0008-5472.CAN-05-0529. Cancer Res. 2005. PMID: 16230418 Free PMC article. - Hyperactivation of STAT3 is involved in abnormal differentiation of dendritic cells in cancer.
Nefedova Y, Huang M, Kusmartsev S, Bhattacharya R, Cheng P, Salup R, Jove R, Gabrilovich D. Nefedova Y, et al. J Immunol. 2004 Jan 1;172(1):464-74. doi: 10.4049/jimmunol.172.1.464. J Immunol. 2004. PMID: 14688356 - Mechanistic insights into impaired dendritic cell function by rapamycin: inhibition of Jak2/Stat4 signaling pathway.
Chiang PH, Wang L, Bonham CA, Liang X, Fung JJ, Lu L, Qian S. Chiang PH, et al. J Immunol. 2004 Feb 1;172(3):1355-63. doi: 10.4049/jimmunol.172.3.1355. J Immunol. 2004. PMID: 14734710 - Tumor-mediated inhibition of dendritic cell differentiation is mediated by down regulation of protein kinase C beta II expression.
Farren MR, Carlson LM, Lee KP. Farren MR, et al. Immunol Res. 2010 Mar;46(1-3):165-76. doi: 10.1007/s12026-009-8118-5. Immunol Res. 2010. PMID: 19756409 Review. - Metabolic Regulation of Dendritic Cell Differentiation.
He Z, Zhu X, Shi Z, Wu T, Wu L. He Z, et al. Front Immunol. 2019 Mar 13;10:410. doi: 10.3389/fimmu.2019.00410. eCollection 2019. Front Immunol. 2019. PMID: 30930893 Free PMC article. Review.
Cited by
- Electroporation-mediated novel albumin-fused Flt3L DNA delivery promotes cDC1-associated anticancer immunity.
Hu MH, Fan D, Tu HF, Tsai YC, He L, Zhou Z, Cheng M, Xing D, Wang S, Wu A, Wu TC, Hung CF. Hu MH, et al. Gene Ther. 2024 Oct 29. doi: 10.1038/s41434-024-00497-3. Online ahead of print. Gene Ther. 2024. PMID: 39472678 - Tumor-associated macrophages, dendritic cells, and neutrophils: biological roles, crosstalk, and therapeutic relevance.
Shen M, Du Y, Ye Y. Shen M, et al. Med Rev (2021). 2022 Feb 14;1(2):222-243. doi: 10.1515/mr-2021-0014. eCollection 2021 Dec. Med Rev (2021). 2022. PMID: 37724296 Free PMC article. Review. - Signaling pathways involved in the biological functions of dendritic cells and their implications for disease treatment.
Cheng H, Chen W, Lin Y, Zhang J, Song X, Zhang D. Cheng H, et al. Mol Biomed. 2023 May 15;4(1):15. doi: 10.1186/s43556-023-00125-3. Mol Biomed. 2023. PMID: 37183207 Free PMC article. Review. - Defective Induction of IL-27-Mediated Immunoregulation by Myeloid DCs in Multiple Sclerosis.
von Glehn F, Pochet N, Thapa B, Raheja R, Mazzola MA, Jangi S, Beynon V, Huang J, Farias AS, Paul A, Santos LMB, Gandhi R, Murugaiyan G, Weiner HL, Baecher-Allan CM. von Glehn F, et al. Int J Mol Sci. 2023 Apr 28;24(9):8000. doi: 10.3390/ijms24098000. Int J Mol Sci. 2023. PMID: 37175706 Free PMC article. - Novel strategies for cancer immunotherapy: counter-immunoediting therapy.
Liu S, Sun Q, Ren X. Liu S, et al. J Hematol Oncol. 2023 Apr 13;16(1):38. doi: 10.1186/s13045-023-01430-8. J Hematol Oncol. 2023. PMID: 37055849 Free PMC article. Review.
References
- Steinman RM. Some interfaces of dendritic cell biology. Apmis. 2003;111:675. - PubMed
- Banchereau J, Briere F, Caux C, Davoust J, Lebecque S, Liu YJ, Pulendran B, Palucka KA. Immunobiology of dendritic cells. Annu Rev Immunol. 2000;18:767. - PubMed
- Lanzavecchia A, Sallusto F. Regulation of T cell immunity by dendritic cells. Cell. 2001;106:263. - PubMed
- Gabrilovich D. The mechanisms and functional significance of tumour-induced dendritic-cell defects. Nat Rev Immunol. 2004;4:941. - PubMed
- Laouar Y, Welte T, Fu XY, Flavell RA. STAT3 Is Required for Flt3L-Dependent Dendritic Cell Differentiation. Immunity. 2003;19:903. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- F32CA103393/CA/NCI NIH HHS/United States
- 5P01CA78038/CA/NCI NIH HHS/United States
- R01 CA084488/CA/NCI NIH HHS/United States
- F32 CA103393/CA/NCI NIH HHS/United States
- CA100062/CA/NCI NIH HHS/United States
- R01 CA100062/CA/NCI NIH HHS/United States
- CA84488/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous