Clues to the functions of mammalian sleep - PubMed (original) (raw)
Review
Clues to the functions of mammalian sleep
Jerome M Siegel. Nature. 2005.
Abstract
The functions of mammalian sleep remain unclear. Most theories suggest a role for non-rapid eye movement (NREM) sleep in energy conservation and in nervous system recuperation. Theories of REM sleep have suggested a role for this state in periodic brain activation during sleep, in localized recuperative processes and in emotional regulation. Across mammals, the amount and nature of sleep are correlated with age, body size and ecological variables, such as whether the animals live in a terrestrial or an aquatic environment, their diet and the safety of their sleeping site. Sleep may be an efficient time for the completion of a number of functions, but variations in sleep expression indicate that these functions may differ across species.
Conflict of interest statement
The author declares no competing interests.
Figures
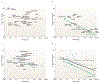
Figure 1 |. Distribution of some key sleep-regulating neuronal populations plotted on a sagittal section of a rat brain.
Circles indicate ‘REM sleep off’ neurons; purple represents serotonergic neurons (located on the midline), orange represents adrenergic or noradrenergic neurons, blue represents histaminergic neurons, red represents hypocretinergic (orexinergic) neurons. Squares indicate ‘sleep on’ neurons. The green star indicates ‘REM sleep on’ neurons. The area shaded in grey is both necessary and sufficient for REM sleep generation. The area shaded in yellow is both necessary and sufficient for NREM sleep generation. In the intact animal both REM sleep and NREM sleep involve interactions between brainstem and forebrain structures. Vlpo, ventrolateral preoptic area; Mpo, median preoptic.
Figure 2 |. Sleep time in mammals.
a, Carnivores are shown in dark red; b, herbivores are in green and c, omnivores in grey. Sleep times in carnivores, omnivores and herbivores differ significantly (P < 0.0002, F test, d.f. 2,68), with carnivore sleep amounts significantly greater than those of herbivores (P < 0.001, _t_-test, d.f. 24, 22). Sleep amount is an inverse function of body mass over all terrestrial mammals (black line). This function accounts for approximately 25% of the interspecies variance (d) in reported sleep amounts (regression of log weight against sleep amount, R = −0.5, P < 0.0001, n = 71). Herbivores are responsible for this relation because body mass and sleep time were significantly and inversely correlated in herbivores (R = −0.77, P < 0.001, d.f. 24), but were not in carnivores (R = −0.28, d.f. 24) or omnivores (R = −0.25, d.f. 25). It is not easy to quantify sleep parameters throughout the animal kingdom and as a result all desired parameters are rarely measured. For example, arousal thresholds are rarely systematically measured as part of a phylogenetic sleep study. Evidence for homeostatic regulation of sleep (sleep rebound) is seldom sought. Most species have not been implanted with electrodes for monitoring muscle tone and other variables. Instead, estimates are often based on visual observations, with the observer forced to intuit the differences between quiet waking and sleep. Other factors such as temperature, light cycle, food and noise conditions, which all affect sleep, have often not been controlled for. The age and health of the animals observed can vary, particularly depending on whether observations are made on animals in the wild or in the zoo. Often, observations of only one or two individuals are the source of the reported sleep amount of a given species. In animals observed in the wild, the weight of the subject is often not known, and in many cases the typical adult body weight, brain weight and other anatomical and physiological parameters cannot be or have not been precisely determined. Despite these sources of noise, significant relationships between weight, sleep time and diet are apparent. Source data for this figure were mainly from ref. .
Figure 3 |. Unihemispheric slow waves in cetaceans.
Top, photos of immature beluga, adult dolphin and section of adult dolphin brain. Electroencephalogram (EEG) of adult cetaceans, represented here by the beluga, during sleep are shown. All species of cetacean so far recorded have unihemispheric slow waves,,–. Top traces show left and right EEG activity. The spectral plots show 1–3-Hz power in the two hemispheres over a 12-hour period. The pattern in the cetaceans contrasts with the bilateral pattern of slow waves seen under normal conditions in all terrestrial mammals, represented here by the rat (bottom traces). The brain photograph is from the University of Wisconsin, Michigan State, and the National Museum of Health Comparative Mammalian Brain Collections.
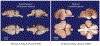
Figure 4 |. Size of the neocortex does not correlate positively with daily sleep amount.
Sleep amount is not proportional to the relative size of the cerebral cortex or to the degree of encephalization, as illustrated by these two examples. Brain photographs are from the University of Wisconsin, Michigan State, and the National Museum of Health Comparative Mammalian Brain Collections.
Similar articles
- The REM sleep-memory consolidation hypothesis.
Siegel JM. Siegel JM. Science. 2001 Nov 2;294(5544):1058-63. doi: 10.1126/science.1063049. Science. 2001. PMID: 11691984 Free PMC article. Review. - Sleep alterations in mammals: did aquatic conditions inhibit rapid eye movement sleep?
Madan V, Jha SK. Madan V, et al. Neurosci Bull. 2012 Dec;28(6):746-58. doi: 10.1007/s12264-012-1285-8. Epub 2012 Dec 7. Neurosci Bull. 2012. PMID: 23225315 Free PMC article. Review. - The ontogeny of mammalian sleep: a reappraisal of alternative hypotheses.
Frank MG, Heller HC. Frank MG, et al. J Sleep Res. 2003 Mar;12(1):25-34. doi: 10.1046/j.1365-2869.2003.00339.x. J Sleep Res. 2003. PMID: 12603784 Review. - The maturational trajectories of NREM and REM sleep durations differ across adolescence on both school-night and extended sleep.
Feinberg I, Davis NM, de Bie E, Grimm KJ, Campbell IG. Feinberg I, et al. Am J Physiol Regul Integr Comp Physiol. 2012 Mar 1;302(5):R533-40. doi: 10.1152/ajpregu.00532.2011. Epub 2011 Nov 23. Am J Physiol Regul Integr Comp Physiol. 2012. PMID: 22116514 Free PMC article. - Phylogenetic analysis of the ecology and evolution of mammalian sleep.
Capellini I, Barton RA, McNamara P, Preston BT, Nunn CL. Capellini I, et al. Evolution. 2008 Jul;62(7):1764-1776. doi: 10.1111/j.1558-5646.2008.00392.x. Evolution. 2008. PMID: 18384657 Free PMC article.
Cited by
- Impaired insulin signaling in human adipocytes after experimental sleep restriction: a randomized, crossover study.
Broussard JL, Ehrmann DA, Van Cauter E, Tasali E, Brady MJ. Broussard JL, et al. Ann Intern Med. 2012 Oct 16;157(8):549-57. doi: 10.7326/0003-4819-157-8-201210160-00005. Ann Intern Med. 2012. PMID: 23070488 Free PMC article. Clinical Trial. - Can Sleep and Resting Behaviours Be Used as Indicators of Welfare in Shelter Dogs (Canis lupus familiaris)?
Owczarczak-Garstecka SC, Burman OH. Owczarczak-Garstecka SC, et al. PLoS One. 2016 Oct 12;11(10):e0163620. doi: 10.1371/journal.pone.0163620. eCollection 2016. PLoS One. 2016. PMID: 27732667 Free PMC article. - Genetic ablation of hypocretin neurons alters behavioral state transitions in zebrafish.
Elbaz I, Yelin-Bekerman L, Nicenboim J, Vatine G, Appelbaum L. Elbaz I, et al. J Neurosci. 2012 Sep 12;32(37):12961-72. doi: 10.1523/JNEUROSCI.1284-12.2012. J Neurosci. 2012. PMID: 22973020 Free PMC article. - Manipulating the circadian and sleep cycles to protect against metabolic disease.
Nohara K, Yoo SH, Chen ZJ. Nohara K, et al. Front Endocrinol (Lausanne). 2015 Mar 23;6:35. doi: 10.3389/fendo.2015.00035. eCollection 2015. Front Endocrinol (Lausanne). 2015. PMID: 25852644 Free PMC article. Review. - Sleep in the rock hyrax, Procavia capensis.
Gravett N, Bhagwandin A, Lyamin OI, Siegel JM, Manger PR. Gravett N, et al. Brain Behav Evol. 2012;79(3):155-69. doi: 10.1159/000335342. Epub 2012 Jan 31. Brain Behav Evol. 2012. PMID: 22301688 Free PMC article.
References
- Dinges DF, Rogers NL & Baynard MD in Principles and Practice of Sleep Medicine Vol. 4 (eds Kryger MH, Roth T & Dement WC) 67–76 (Elsevier Saunders, Philadelphia, 2005).
- Huber R et al. Sleep homeostasis in Drosophila melanogaster. Sleep 27, 628–639 (2004). - PubMed
- Czeisler CA, Buxton O & Khalsa SBS in Principles and Practice of Sleep Medicine Vol. 4 (eds Kryger MH, Roth T & Dement WC) 375–394 (Elsevier Saunders, Philadelphia, 2005).
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources