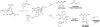Sir2 protein deacetylases: evidence for chemical intermediates and functions of a conserved histidine - PubMed (original) (raw)
Sir2 protein deacetylases: evidence for chemical intermediates and functions of a conserved histidine
Brian C Smith et al. Biochemistry. 2006.
Abstract
Sir2 NAD+-dependent protein deacetylases are implicated in a variety of cellular processes such as apoptosis, gene silencing, life-span regulation, and fatty acid metabolism. Despite this, there have been relatively few investigations into the detailed chemical mechanism. Sir2 proteins (sirtuins) catalyze the chemical conversion of NAD+ and acetylated lysine to nicotinamide, deacetylated lysine, and 2'-O-acetyl-ADP-ribose (OAADPr). In this study, Sir2-catalyzed reactions are shown to transfer an 18O label from the peptide acetyl group to the ribose 1'-position of OAADPr, providing direct evidence for the formation of a covalent alpha-1'-O-alkylamidate, whose existence is further supported by the observed methanolysis of the alpha-1'-O-alkylamidate intermediate to yield beta-1'-O-methyl-ADP-ribose in a Sir2 histidine-to-alanine mutant. This conserved histidine (His-135 in HST2) activates the ribose 2'-hydroxyl for attack on the alpha-1'-O-alkylamidate. The histidine mutant is stalled at the intermediate, allowing water and other alcohols to compete kinetically with the attacking 2'-hydroxyl. Measurement of the pH dependence of kcat and kcat/Km values for both wild-type and histidine-to-alanine mutant enzymes confirms roles of this residue in NAD+ binding and in general-base activation of the 2'-hydroxyl. Also, transfer of an 18O label from water to the carbonyl oxygen of the acetyl group in OAADPr is consistent with water addition to the proposed 1',2'-cyclic intermediate formed after 2'-hydroxyl attack on the alpha-1'-O-alkylamidate. The effect of pH and of solvent viscosity on the kcat values suggests that final product release is rate-limiting in the wild-type enzyme. Implications of this new evidence on the mechanisms of deacetylation and possible ADP-ribosylation catalyzed by Sir2 deacetylases are discussed.
Figures
Scheme 1
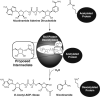
General reaction catalyzed by Sir2 protein deacetylases.
Scheme 2
Proposed mechanism of formation of both ADPr and _O_AADPr in HST2 H135A.
Figure 1
(A) 18O-labelling experiments with HST2 WT. Reactions contained 1 mM DTT, 200 _μ_M18O-AcH3, 185 _μ_M NAD+, 10 _μ_M HST2, in natural abundance water or 90% 18OH2, and 20 mM pyridine buffer at pH 7. Each reaction was split into three aliquots, two of which were lyophilized and exchanged in 10% formic acid and 90% natural abundance OH2 or 18OH2. 18O incorporation was determined by examining the relative peak heights at ∼600.5, ∼602.5, and ∼604.5 m/z corresponding to zero, one, or two 18O atoms. (B) The rate of non-enzymatic exchange of an 18O-label from water into _O_AADPr. Purified _O_AADPr was diluted to 16.5 _μ_M in 20 mM pyridine buffer and 90% 18OH2 at pH 7. Timepoints were injected directly into the mass spectrometer. The y-intercept, which represents the zero-timepoint before exposing _O_AADPr to 18O-water, was calculated as 13.3%, and is consistent with the natural distribution of heavy isotopes across the entire _O_AADPr molecule. A rate of non-enzymatic incorporation of 0.15% per minute was calculated, which corresponds to a maximum of 4.5% non-enzymatic incorporation for the 30-minute reaction time used in our assays.
Figure 1
(A) 18O-labelling experiments with HST2 WT. Reactions contained 1 mM DTT, 200 _μ_M18O-AcH3, 185 _μ_M NAD+, 10 _μ_M HST2, in natural abundance water or 90% 18OH2, and 20 mM pyridine buffer at pH 7. Each reaction was split into three aliquots, two of which were lyophilized and exchanged in 10% formic acid and 90% natural abundance OH2 or 18OH2. 18O incorporation was determined by examining the relative peak heights at ∼600.5, ∼602.5, and ∼604.5 m/z corresponding to zero, one, or two 18O atoms. (B) The rate of non-enzymatic exchange of an 18O-label from water into _O_AADPr. Purified _O_AADPr was diluted to 16.5 _μ_M in 20 mM pyridine buffer and 90% 18OH2 at pH 7. Timepoints were injected directly into the mass spectrometer. The y-intercept, which represents the zero-timepoint before exposing _O_AADPr to 18O-water, was calculated as 13.3%, and is consistent with the natural distribution of heavy isotopes across the entire _O_AADPr molecule. A rate of non-enzymatic incorporation of 0.15% per minute was calculated, which corresponds to a maximum of 4.5% non-enzymatic incorporation for the 30-minute reaction time used in our assays.
Figure 2
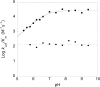
pH rate profiles. Reaction rates were determined based on _O_AADPr formation. Reactions for HST2 WT contained 150 _μ_M 3H-AcH3, 1 mM DTT, 0.02−2 _μ_M HST2 WT, and varying concentrations of NAD+ from 5−320 _μ_M in TBA buffer. Reaction for HST2 H135A contained 2−3 mM 3H-AcH3, 1 mM DTT, 2−5 _μ_M HST2 H135A, and varying concentrations of NAD+ from 5−1280 _μ_M in TBA buffer. (A) Effect of pH on kcat/Km varying NAD+ for HST2 WT (squares) and HST2 H135A (circles). HST2 WT displayed a critical ionization with a pKa of 6.77 ± 0.06, while HST2 H135A exhibited no effect over the pH range tested. (B) Effect of pH on kcat for HST2 WT (squares) and HST2 H135A (circles). HST2 H135A displayed a critical ionization with a pKa of 7.43 ± 0.13 while HST2 WT showed no critical ionizations over the pH range tested.
Figure 2
pH rate profiles. Reaction rates were determined based on _O_AADPr formation. Reactions for HST2 WT contained 150 _μ_M 3H-AcH3, 1 mM DTT, 0.02−2 _μ_M HST2 WT, and varying concentrations of NAD+ from 5−320 _μ_M in TBA buffer. Reaction for HST2 H135A contained 2−3 mM 3H-AcH3, 1 mM DTT, 2−5 _μ_M HST2 H135A, and varying concentrations of NAD+ from 5−1280 _μ_M in TBA buffer. (A) Effect of pH on kcat/Km varying NAD+ for HST2 WT (squares) and HST2 H135A (circles). HST2 WT displayed a critical ionization with a pKa of 6.77 ± 0.06, while HST2 H135A exhibited no effect over the pH range tested. (B) Effect of pH on kcat for HST2 WT (squares) and HST2 H135A (circles). HST2 H135A displayed a critical ionization with a pKa of 7.43 ± 0.13 while HST2 WT showed no critical ionizations over the pH range tested.
Figure 3
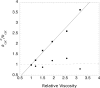
Effect of viscosity on kcat. Reaction rates were determined based on _O_AADPr formation. k cat 0/kcat is the kcat the absence of sucrose divided by the kcat measured at each concentration of sucrose. HST2 WT (squares) reactions contained 500 _μ_M NAD+, 2−64 _μ_M AcH3 (11-mer) from, 1 mM DTT, 0−34% w/v sucrose, and 30−40 nM HST2 WT in TBA buffer at pH 8. HST2 H135A (circles) reactions contained 2 mM NAD+, 150−2400 _μ_M AcH3 (11-mer), 1 mM DTT, 0−34% w/v sucrose, and 30−40 nM HST2 WT in TBA buffer at pH 8. Linear regression analysis showed a slope of 1.15 ± 0.07 for HST2 WT and 0.02 ± 0.12 for HST2 H135A.
Figure 4
The relative formation of ADPr and _O_AADPr. Reactions contained 400−600 _μ_M AcH3 (11-mer), 1 mM DTT, 500−1000 _μ_M 32P-NAD+, 20−200 _μ_M HST2 H135A or 0.5−50 _μ_M HST2 WT, and 20−50 mM Tris-Cl buffer at pH 7.5 and 25 °C. The ADPr observed from HST2 WT was at background levels and its formation did not increase over time. Each column was normalized to the total product formed a each timepoint. Error bars represent standard deviations.
Figure 5
18O-labelling experiments with HST2 H135A. Reactions contained 1 mM DTT, 400 _μ_M AcH3 (11-mer), 500 _μ_M NAD+, 50 _μ_M HST2 H135A, 84% 18OH2, and 20 mM pyridine buffer adjusted at pH 7 were reacted for 30 minutes at 25 °C. Reactions were split into three aliquots, two of which were lyophilized and then exchanged in 10% formic acid and 90% natural abundance OH2 or 18OH2. 18O incorporation was determined by examining the relative peak heights at ∼600.5, ∼602.5, and ∼604.5 m/z corresponding to zero, one, or two 18O atoms.
Figure 6
(A) ESI-MS of the 30 minute reaction of HST2 H135A with 1 mM DTT, 400 _μ_M AcH3 (11-mer), 500 _μ_M NAD+, 50 _μ_M HST2 H135A, 20% v/v MeOH, and 20 mM pyridine buffer at pH 7 at 25 °C. HST2 H135A forms ADPr, 1’-_O_-methyl-ADPr, and _O_AADPr under these conditions. (B) ESI-MS of the 30 minute control reaction of HST2 WT with 1 mM DTT, 400 _μ_M AcH3 (11-mer), 500 _μ_M NAD+, 21 _μ_M HST2 WT, 20% v/v MeOH, and 20 mM pyridine buffer at pH 7 at 25 °C. HST2 WT forms no detectable 1’-_O_-methyl-ADPr under these reaction conditions. (C) ESI-MS of the 30 minute non-enzymatic incorporation of 20% v/v MeOH into 500 _μ_M ADPr with 1 mM DTT, 400 _μ_M AcH3 (11-mer), and 20 mM pyridine buffer at pH 7 at 25 °C. There was no detectable 1’-_O_-methyl-ADPr in this control. Therefore, the 1’-_O_-methyl-ADPr observed with HST2 H135A was due to enzymatic activity inherent in the mutant enzyme only.
Similar articles
- Structural basis for nicotinamide cleavage and ADP-ribose transfer by NAD(+)-dependent Sir2 histone/protein deacetylases.
Zhao K, Harshaw R, Chai X, Marmorstein R. Zhao K, et al. Proc Natl Acad Sci U S A. 2004 Jun 8;101(23):8563-8. doi: 10.1073/pnas.0401057101. Epub 2004 May 18. Proc Natl Acad Sci U S A. 2004. PMID: 15150415 Free PMC article. - Substrate specificity and kinetic mechanism of the Sir2 family of NAD+-dependent histone/protein deacetylases.
Borra MT, Langer MR, Slama JT, Denu JM. Borra MT, et al. Biochemistry. 2004 Aug 3;43(30):9877-87. doi: 10.1021/bi049592e. Biochemistry. 2004. PMID: 15274642 - Sir2 regulation by nicotinamide results from switching between base exchange and deacetylation chemistry.
Sauve AA, Schramm VL. Sauve AA, et al. Biochemistry. 2003 Aug 12;42(31):9249-56. doi: 10.1021/bi034959l. Biochemistry. 2003. PMID: 12899610 - SIR2: the biochemical mechanism of NAD(+)-dependent protein deacetylation and ADP-ribosyl enzyme intermediates.
Sauve AA, Schramm VL. Sauve AA, et al. Curr Med Chem. 2004 Apr;11(7):807-26. doi: 10.2174/0929867043455675. Curr Med Chem. 2004. PMID: 15078167 Review. - The Sir 2 family of protein deacetylases.
Denu JM. Denu JM. Curr Opin Chem Biol. 2005 Oct;9(5):431-40. doi: 10.1016/j.cbpa.2005.08.010. Curr Opin Chem Biol. 2005. PMID: 16122969 Review.
Cited by
- Entamoeba histolytica sirtuin EhSir2a deacetylates tubulin and regulates the number of microtubular assemblies during the cell cycle.
Dam S, Lohia A. Dam S, et al. Cell Microbiol. 2010 Jul;12(7):1002-14. doi: 10.1111/j.1462-5822.2010.01449.x. Epub 2010 Feb 9. Cell Microbiol. 2010. PMID: 20148900 Free PMC article. - Nuclear ADP-ribosylation reactions in mammalian cells: where are we today and where are we going?
Hassa PO, Haenni SS, Elser M, Hottiger MO. Hassa PO, et al. Microbiol Mol Biol Rev. 2006 Sep;70(3):789-829. doi: 10.1128/MMBR.00040-05. Microbiol Mol Biol Rev. 2006. PMID: 16959969 Free PMC article. Review. - Mechanisms and molecular probes of sirtuins.
Smith BC, Hallows WC, Denu JM. Smith BC, et al. Chem Biol. 2008 Oct 20;15(10):1002-13. doi: 10.1016/j.chembiol.2008.09.009. Chem Biol. 2008. PMID: 18940661 Free PMC article. Review. - Structure and biochemical functions of SIRT6.
Pan PW, Feldman JL, Devries MK, Dong A, Edwards AM, Denu JM. Pan PW, et al. J Biol Chem. 2011 Apr 22;286(16):14575-87. doi: 10.1074/jbc.M111.218990. Epub 2011 Mar 1. J Biol Chem. 2011. PMID: 21362626 Free PMC article.
References
- Rusche LN, Kirchmaier AL, Rine J. The establishment, inheritance, and function of silenced chromatin in Saccharomyces cerevisiae. Annu Rev Biochem. 2003;72:481–516. - PubMed
- Gasser SM, Cockell MM. The molecular biology of the SIR proteins. Gene. 2001;279:1–16. - PubMed
- Starai VJ, Celic I, Cole RN, Boeke JD, Escalante-Semerena JC. Sir2-dependent activation of acetyl-CoA synthetase by deacetylation of active lysine. Science. 2002;298:2390–2392. - PubMed
- Vaziri H, Dessain SK, Ng Eaton E, Imai SI, Frye RA, Pandita TK, Guarente L, Weinberg RA. hSIR2(SIRT1) functions as an NAD-dependent p53 deacetylase. Cell. 2001;107:149–159. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- GM065386/GM/NIGMS NIH HHS/United States
- R01 GM065386/GM/NIGMS NIH HHS/United States
- T32 GM008349/GM/NIGMS NIH HHS/United States
- 5 T32 GM08349/GM/NIGMS NIH HHS/United States
- R01 GM065386-05S1/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources