The "perivascular pump" driven by arterial pulsation is a powerful mechanism for the distribution of therapeutic molecules within the brain - PubMed (original) (raw)
The "perivascular pump" driven by arterial pulsation is a powerful mechanism for the distribution of therapeutic molecules within the brain
Piotr Hadaczek et al. Mol Ther. 2006 Jul.
Abstract
We investigated the movement of interstitially infused macromolecules within the central nervous system (CNS) in rats with high and low blood pressure (BP)/heart rate and in rats euthanized immediately before infusion (no heart action). Adeno-associated virus 2 (AAV2), fluorescent liposomes, or bovine serum albumin was infused into rat striatum (six hemispheres per group) by convection-enhanced delivery (CED). After infusion, distribution volumes were evaluated. The rats with high BP/heart rate displayed a significantly larger distribution of the infused molecules within the injected site and more extensive transport of those molecules to the globus pallidus. This difference was particularly apparent for AAV2, for which a 16.5-fold greater distribution of viral capsids was observed in the rats with high BP/heart rate than in the rats with no heartbeat. Similar results were observed for liposomes, despite their larger diameter. The distribution of all infused molecules in all rats that had low or no blood flow was confined to the space around brain blood vessels. These findings show that fluid circulation within the CNS through the perivascular space is the primary mechanism by which viral particles and other therapeutic agents administered by CED are spread within the brain and that cardiac contractions power this process.
Figures
FIG. 1
Volumes of distribution (_V_d) of three substances infused into rat striatum (top) and distributed from there into the globus pallidus (bottom). Dark-shaded columns represent values for group H (high blood pressure/heart rate). Light-shaded columns represent values from group L (low blood pressure/heart rate). White columns represent values for group N (rats with no heart action). Standard deviation bars are shown on each column. Statistically significant differences in both the striatum and the globus pallidus among rats infused with the different molecules are shown as relative differences with *P < 0.009 and **P < 0.00005. Of interest is that the striatal _V_d of liposomes in group H was almost identical to that of bovine serum albumin. AAV2 distribution was significantly less due to entrapment of viral capsids by heparan sulfate proteoglycan receptors widely present on neurons [20].
FIG. 2
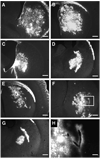
Photographs of brain sections stained for AAV2 capsids (antibody A20) in rats after intrastriatal infusion of AAV2 by convection-enhanced delivery. (A and B) Striatal sections from two different rats from group H (high blood pressure/heart rate); (C and D) brain sections from a more caudal part of the same brains showing positive signal in globus pallidus. For all rats from group H, the fluorescent signal was spread evenly within the hemisphere, forming a mostly uninterrupted distribution. (E and F) Brain sections from two rats with low blood pressure/heart rate when AAV2 was infused. The signal is restricted to the vicinity of infusion and the perivascular area surrounding nearby blood vessels. (G and H) Higher magnification views of E and F (original magnification ×20). Size bars: 500 (A through F) and 125 µm (G and H).
FIG. 3
Sections from rat brains infused with fluorescent liposomes. (A through D) Note the robust and widespread liposomal distribution within the striatum (A and B) and globus pallidus (C and D) of rats from group H (high blood pressure/heart rate). This signal pattern forms a fairly continuous area of fluorescence. (E and F) Striatal sections from rats from group L (low blood pressure/heart rate). Note the arborized pattern of spread suggesting perivascular pathways. (G) Brain section from a more caudal part of the brain from a rat from group L showing signal confined in the globus pallidus. (H) Higher magnification view of F (original magnification ×20). Size bars: 500 (A–G) and 125 µm (H).
Similar articles
- Effects of the perivascular space on convection-enhanced delivery of liposomes in primate putamen.
Krauze MT, Saito R, Noble C, Bringas J, Forsayeth J, McKnight TR, Park J, Bankiewicz KS. Krauze MT, et al. Exp Neurol. 2005 Nov;196(1):104-11. doi: 10.1016/j.expneurol.2005.07.009. Epub 2005 Aug 16. Exp Neurol. 2005. PMID: 16109410 - Real-time imaging of convection-enhanced delivery of viruses and virus-sized particles.
Szerlip NJ, Walbridge S, Yang L, Morrison PF, Degen JW, Jarrell ST, Kouri J, Kerr PB, Kotin R, Oldfield EH, Lonser RR. Szerlip NJ, et al. J Neurosurg. 2007 Sep;107(3):560-7. doi: 10.3171/JNS-07/09/0560. J Neurosurg. 2007. PMID: 17886556 - Extensive distribution of liposomes in rodent brains and brain tumors following convection-enhanced delivery.
Mamot C, Nguyen JB, Pourdehnad M, Hadaczek P, Saito R, Bringas JR, Drummond DC, Hong K, Kirpotin DB, McKnight T, Berger MS, Park JW, Bankiewicz KS. Mamot C, et al. J Neurooncol. 2004 May;68(1):1-9. doi: 10.1023/b:neon.0000024743.56415.4b. J Neurooncol. 2004. PMID: 15174514 - The use of convection-enhanced delivery with liposomal toxins in neurooncology.
Fiandaca MS, Berger MS, Bankiewicz KS. Fiandaca MS, et al. Toxins (Basel). 2011 Apr;3(4):369-97. doi: 10.3390/toxins3040369. Epub 2011 Mar 31. Toxins (Basel). 2011. PMID: 22069714 Free PMC article. Review. - Real-time imaging and quantification of brain delivery of liposomes.
Krauze MT, Forsayeth J, Park JW, Bankiewicz KS. Krauze MT, et al. Pharm Res. 2006 Nov;23(11):2493-504. doi: 10.1007/s11095-006-9103-5. Epub 2006 Sep 14. Pharm Res. 2006. PMID: 16972184 Review.
Cited by
- The glymphatic system: Current understanding and modeling.
Bohr T, Hjorth PG, Holst SC, Hrabětová S, Kiviniemi V, Lilius T, Lundgaard I, Mardal KA, Martens EA, Mori Y, Nägerl UV, Nicholson C, Tannenbaum A, Thomas JH, Tithof J, Benveniste H, Iliff JJ, Kelley DH, Nedergaard M. Bohr T, et al. iScience. 2022 Aug 20;25(9):104987. doi: 10.1016/j.isci.2022.104987. eCollection 2022 Sep 16. iScience. 2022. PMID: 36093063 Free PMC article. Review. - Fluid dynamics of cerebrospinal fluid flow in perivascular spaces.
Thomas JH. Thomas JH. J R Soc Interface. 2019 Oct 31;16(159):20190572. doi: 10.1098/rsif.2019.0572. Epub 2019 Oct 23. J R Soc Interface. 2019. PMID: 31640500 Free PMC article. - Glial Cell Line-Derived Neurotrophic Factor Gene Delivery in Parkinson's Disease: A Delicate Balance between Neuroprotection, Trophic Effects, and Unwanted Compensatory Mechanisms.
Tenenbaum L, Humbert-Claude M. Tenenbaum L, et al. Front Neuroanat. 2017 Apr 10;11:29. doi: 10.3389/fnana.2017.00029. eCollection 2017. Front Neuroanat. 2017. PMID: 28442998 Free PMC article. Review. - Widespread brain distribution and activity following i.c.v. infusion of anti-β-secretase (BACE1) in nonhuman primates.
Yadav DB, Maloney JA, Wildsmith KR, Fuji RN, Meilandt WJ, Solanoy H, Lu Y, Peng K, Wilson B, Chan P, Gadkar K, Kosky A, Goo M, Daugherty A, Couch JA, Keene T, Hayes K, Nikolas LJ, Lane D, Switzer R, Adams E, Watts RJ, Scearce-Levie K, Prabhu S, Shafer L, Thakker DR, Hildebrand K, Atwal JK. Yadav DB, et al. Br J Pharmacol. 2017 Nov;174(22):4173-4185. doi: 10.1111/bph.14021. Epub 2017 Oct 6. Br J Pharmacol. 2017. PMID: 28859225 Free PMC article. - Peristaltic flow in the glymphatic system.
Romanò F, Suresh V, Galie PA, Grotberg JB. Romanò F, et al. Sci Rep. 2020 Dec 3;10(1):21065. doi: 10.1038/s41598-020-77787-4. Sci Rep. 2020. PMID: 33273489 Free PMC article.
References
- Kaspar BK, et al. Targeted retrograde gene delivery for neuronal protection. Mol. Ther. 2002;5:50–56. - PubMed
- Tsiang H. Evidence for an intraaxonal transport of fixed and street rabies virus. J. Neuropathol. Exp. Neurol. 1979;38:286–299. - PubMed
- Lascano EF, Berria MI. Histological study of the progression of herpes simplex virus in mice. Arch. Virol. 1980;64:67–79. - PubMed
- Kristensson K, Nennesmo L, Persson L, Lycke E. Neuron to neuron transmission of herpes simplex virus: transport of virus from skin to brainstem nuclei. J. Neurol. Sci. 1982;54:149–156. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources