Antioxidants protect PINK1-dependent dopaminergic neurons in Drosophila - PubMed (original) (raw)
Antioxidants protect PINK1-dependent dopaminergic neurons in Drosophila
Danling Wang et al. Proc Natl Acad Sci U S A. 2006.
Abstract
Parkinson's disease (PD) is the most frequent neurodegenerative movement disorder. Mutations in the PINK1 gene are linked to the autosomal recessive early onset familial form of PD. The physiological function of PINK1 and pathological abnormality of PD-associated PINK1 mutants are largely unknown. We here show that inactivation of Drosophila PINK1 (dPINK1) using RNAi results in progressive loss of dopaminergic neurons and in ommatidial degeneration of the compound eye, which is rescued by expression of human PINK1 (hPINK1). Expression of human SOD1 suppresses neurodegeneration induced by dPINK1 inactivation. Moreover, treatment of dPINK1 RNAi flies with the antioxidants SOD and vitamin E significantly inhibits ommatidial degeneration. Thus, dPINK1 plays an essential role in maintaining neuronal survival by preventing neurons from undergoing oxidative stress, thereby suggesting a potential mechanism by which a reduction in PINK1 function leads to PD-associated neurodegeneration.
Conflict of interest statement
Conflict of interest statement: No conflicts declared.
Figures
Fig. 1.
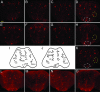
Knockdown of dPINK1 results in age-dependent loss of DA neurons, but not serotonergic neurons, in Drosophila. Brains dissected from flies expressing _elav_-GAL4 driver alone (A, E, and L), _elav_-GAL4/UAS-lac Z (B, F, and M), _elav_-GAL4/UAS-hPINK1 (C, G, and N), _elav_-GAL4/UAS-dPINK1 RNAi (D, H, and O), and _elav_-GAL4/UAS-dPINK1 RNAi/UAS-hPINK1 (K) aged 1 day (A–D) or 10 days (E–G, K, and L–O) were immunostained with either anti-Drosophila TH antibody or anti-5HT antibody followed by an Alexa Fluor 594-labeled secondary antibody to identify DA (A–H and K) or serotonergic (L–O) neurons, respectively. Representative pictures shown were collected by using confocal microscopy. Localization of Drosophila DA and serotonergic neurons are illustrated in I and J, respectively. Note that changes of DA neurons in VUM (white circle) and PPL2 (yellow circle) regions in 1-day (D) and 10-day (H) fly brains with dPINK1 knockdown as well as 10-day fly brains (K) with dPINK1 knockdown rescued by hPINK1 are indicated. Serotonergic neurons remain similar among all groups (L–O). (Scale bar, 50 μm.)
Fig. 2.
Knockdown of dPINK1 induces degeneration of ommatidia and retinal neurons in Drosophila. External eye phenotypes of flies aged 1 day expressing _GMR_-GAL4 driver alone (A, _A_′, and _A_″), _GMR_-GAL4/UAS-lac Z (B, _B_′, and _B_″), _GMR_-GAL4/UAS-dPINK1 RNAi (C, _C_′, _C_″), _GMR_-GAL4/UAS-hPINK1 (D and _D_′), _GMR_-GAL4/UAS-_hPINK1_G309D (_D_″), _GMR_-GAL4/UAS-dPINK1 RNAi/UAS-hPINK1 (E and _E_′), and _GMR_-GAL4/UAS-dPINK1 RNAi/UAS-hPINK1 G309D (_E_″) under light microscopy (A_–_E and _A_″–_E_″) and electronic microscopy (_A_′–_E_′). Magnification of light microscopic images and electronic microscopic images is ×25 and ×400, respectively. Arrows indicate lesions of ommatidial degeneration (C, _C_′, _C_″, and _E_″). Microscopic images of DLG staining (F_–_I) as well as phalloidin and elav double staining of 44 h AFP retina dissected from flies expressing _GMR_-GAL4 driver alone (F and J), _GMR_-GAL4/UAS-lac Z (G and K), and _GMR_-GAL4/UAS-dPINK1 RNAi (H, I, L, and M). Images from two independent lines expressing _GMR_-GAL4/UAS-dPINK1 RNAi are shown (H and L are from one line, whereas I and M are from another independent line). Note that dPINK1 knockdown induces cellular disorganization (H and I) and loss of neurons (L and M; yellow arrows). (Scale bar, 10 μm.)
Fig. 3.
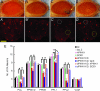
Expression of hSOD1 suppresses degeneration of ommatidia and DA neurons. Representative images of external eye phenotypes of 3-day-old flies expressing _GMR_-GAL4 driver alone (A), _GMR_-GAL4/UAS-lac Z (B), _GMR_-GAL4/UAS-dPINK1 RNAi (C), and _GMR_-GAL4/UAS-dPINK1 RNAi/UAS-hSOD1 (D), as well as images of DA neurons identified by anti-Drosophila TH staining of 10-day-old flies expressing _elav_-GAL4 driver alone (_A_′), _elav_-GAL4/UAS-lac Z (_B_′), _elav_-GAL4/UAS-dPINK1 RNAi (_C_′), and _elav_-GAL4/UAS-dPINK1 RNAi/UAS-hSOD1 (_D_′). Note that ommatidial degeneration (C, yellow arrow) and loss (_C_′) and rescue (_D_′) of DA neurons in PPM1/2 (white circle) and PPL2 (yellow circle) are indicated. (Scale bar, 50 μm.) (E) Quantification of DA neurons is shown for 10-day-old flies expressing _elav_-GAL4 driver alone (WT), _elav_-GAL4/UAS-lac Z (lac Z), _elav_-GAL4/UAS-hPINK1 (hPINK1), _elav_-GAL4/UAS-hSOD1 (SOD1), _elav_-GAL4/UAS-dPINK1 RNAi (dPINK1KD), _elav_-GAL4/UAS-dPINK1 RNAi/UAS-lac Z (dPINK1KD, lacZ), _elav_-GAL4/UAS-dPINK1 RNAi/UAS-hPINK1 (dPINK1KD, hPINK1), and _elav_-GAL4/UAS-dPINK1 RNAi/UAS-hSOD1 (dPINK1KD, SOD1). DA neurons in six brain regions, including PAL, PPM1/2, PPM3, PPL1, PPL2, and VUM, were quantitated, and differences were statistically analyzed. ∗, P < 0.05. ns, no statistical significance.
Fig. 4.
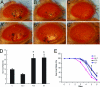
Inhibition of ommatidial degeneration by the antioxidants SOD1 and vitamin E. Flies (7 days old) with dPINK1 knockdown driven by _GMR_-GAL4 (_GMR_-GAL4/UAS-dPINK1 RNAi) were treated without (A) or with 100 units/ml (B) and 1,000 units/ml (C) SOD, or without (_A_′) or with 20 μg/ml (_B_′) and 200 μg/ml (_C_′) vitamin E. Representative images of external eye phenotype of each group are shown. (Magnification, ×25.) Flies overexpressing hPINK1 were treated with either 20 mM paraquat for 24 h (D) or 1% H2O2 over 6 days (E) and were quantitated for survival rates. Results from lines expressing _Da_-GAL4 driver alone (Da-GAL4) or _Da_-GAL4/UAS-lac Z (lac Z) and two independent lines expressing _Da_-GAL4/UAS-hPINK1 (hPINK1FC2 and hPINK1TC1) are shown. ∗∗, P < 0.001.
Comment in
- Antioxidants put Parkinson flies back in the PINK.
Bier E. Bier E. Proc Natl Acad Sci U S A. 2006 Sep 5;103(36):13269-70. doi: 10.1073/pnas.0606288103. Epub 2006 Aug 28. Proc Natl Acad Sci U S A. 2006. PMID: 16938856 Free PMC article. No abstract available.
Similar articles
- Antioxidants put Parkinson flies back in the PINK.
Bier E. Bier E. Proc Natl Acad Sci U S A. 2006 Sep 5;103(36):13269-70. doi: 10.1073/pnas.0606288103. Epub 2006 Aug 28. Proc Natl Acad Sci U S A. 2006. PMID: 16938856 Free PMC article. No abstract available. - Mitochondrial pathology and muscle and dopaminergic neuron degeneration caused by inactivation of Drosophila Pink1 is rescued by Parkin.
Yang Y, Gehrke S, Imai Y, Huang Z, Ouyang Y, Wang JW, Yang L, Beal MF, Vogel H, Lu B. Yang Y, et al. Proc Natl Acad Sci U S A. 2006 Jul 11;103(28):10793-8. doi: 10.1073/pnas.0602493103. Epub 2006 Jul 3. Proc Natl Acad Sci U S A. 2006. PMID: 16818890 Free PMC article. - Pink1 suppresses alpha-synuclein-induced phenotypes in a Drosophila model of Parkinson's disease.
Todd AM, Staveley BE. Todd AM, et al. Genome. 2008 Dec;51(12):1040-6. doi: 10.1139/G08-085. Genome. 2008. PMID: 19088817 - Genetic mutations and functions of PINK1.
Kawajiri S, Saiki S, Sato S, Hattori N. Kawajiri S, et al. Trends Pharmacol Sci. 2011 Oct;32(10):573-80. doi: 10.1016/j.tips.2011.06.001. Epub 2011 Jul 23. Trends Pharmacol Sci. 2011. PMID: 21784538 Review. - PINK1 mediates neuronal survival in monkey.
Sun Z, Ye J, Yuan J. Sun Z, et al. Protein Cell. 2022 Jan;13(1):4-5. doi: 10.1007/s13238-021-00889-w. Epub 2021 Nov 23. Protein Cell. 2022. PMID: 34813054 Free PMC article. Review. No abstract available.
Cited by
- PINK1 protects against oxidative stress by phosphorylating mitochondrial chaperone TRAP1.
Pridgeon JW, Olzmann JA, Chin LS, Li L. Pridgeon JW, et al. PLoS Biol. 2007 Jul;5(7):e172. doi: 10.1371/journal.pbio.0050172. Epub 2007 Jun 19. PLoS Biol. 2007. PMID: 17579517 Free PMC article. - Neurodegenerative models in Drosophila: polyglutamine disorders, Parkinson disease, and amyotrophic lateral sclerosis.
Ambegaokar SS, Roy B, Jackson GR. Ambegaokar SS, et al. Neurobiol Dis. 2010 Oct;40(1):29-39. doi: 10.1016/j.nbd.2010.05.026. Epub 2010 May 31. Neurobiol Dis. 2010. PMID: 20561920 Free PMC article. Review. - Drosophila models of Parkinson's disease: discovering relevant pathways and novel therapeutic strategies.
Muñoz-Soriano V, Paricio N. Muñoz-Soriano V, et al. Parkinsons Dis. 2011 Mar 3;2011:520640. doi: 10.4061/2011/520640. Parkinsons Dis. 2011. PMID: 21512585 Free PMC article. - FOXO3a-dependent regulation of Pink1 (Park6) mediates survival signaling in response to cytokine deprivation.
Mei Y, Zhang Y, Yamamoto K, Xie W, Mak TW, You H. Mei Y, et al. Proc Natl Acad Sci U S A. 2009 Mar 31;106(13):5153-8. doi: 10.1073/pnas.0901104106. Epub 2009 Mar 10. Proc Natl Acad Sci U S A. 2009. PMID: 19276113 Free PMC article. - Developmental nicotine exposure affects larval brain size and the adult dopaminergic system of Drosophila melanogaster.
Morris M, Shaw A, Lambert M, Perry HH, Lowenstein E, Valenzuela D, Velazquez-Ulloa NA. Morris M, et al. BMC Dev Biol. 2018 Jun 14;18(1):13. doi: 10.1186/s12861-018-0172-6. BMC Dev Biol. 2018. PMID: 29898654 Free PMC article.
References
- Lang A. E., Lozano A. M. N. Engl. J. Med. 1998;339:1130–1143. - PubMed
- Lang A. E., Lozano A. M. N. Engl. J. Med. 1998;339:1044–1053. - PubMed
- Dawson T. M., Dawson V. L. Science. 2003;302:819–822. - PubMed
- Valente E. M., Abou-Sleiman P. M., Caputo V., Muqit M. M., Harvey K., Gispert S., Ali Z., Del Turco D., Bentivoglio A. R., Healy D. G., et al. Science. 2004;304:1158–1160. - PubMed
- Paisan-Ruiz C., Jain S., Evans E. W., Gilks W. P., Simon J., van der Brug M., Lopez de Munain A., Aparicio S., Gil A. M., Khan N., et al. Neuron. 2004;44:595–600. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous