Tcof1/Treacle is required for neural crest cell formation and proliferation deficiencies that cause craniofacial abnormalities - PubMed (original) (raw)
Comparative Study
. 2006 Sep 5;103(36):13403-8.
doi: 10.1073/pnas.0603730103. Epub 2006 Aug 28.
Affiliations
- PMID: 16938878
- PMCID: PMC1557391
- DOI: 10.1073/pnas.0603730103
Comparative Study
Tcof1/Treacle is required for neural crest cell formation and proliferation deficiencies that cause craniofacial abnormalities
Jill Dixon et al. Proc Natl Acad Sci U S A. 2006.
Abstract
Neural crest cells are a migratory cell population that give rise to the majority of the cartilage, bone, connective tissue, and sensory ganglia in the head. Abnormalities in the formation, proliferation, migration, and differentiation phases of the neural crest cell life cycle can lead to craniofacial malformations, which constitute one-third of all congenital birth defects. Treacher Collins syndrome (TCS) is characterized by hypoplasia of the facial bones, cleft palate, and middle and external ear defects. Although TCS results from autosomal dominant mutations of the gene TCOF1, the mechanistic origins of the abnormalities observed in this condition are unknown, and the function of Treacle, the protein encoded by TCOF1, remains poorly understood. To investigate the developmental basis of TCS we generated a mouse model through germ-line mutation of Tcof1. Haploinsufficiency of Tcof1 leads to a deficiency in migrating neural crest cells, which results in severe craniofacial malformations. We demonstrate that Tcof1/Treacle is required cell-autonomously for the formation and proliferation of neural crest cells. Tcof1/Treacle regulates proliferation by controlling the production of mature ribosomes. Therefore, Tcof1/Treacle is a unique spatiotemporal regulator of ribosome biogenesis, a deficiency that disrupts neural crest cell formation and proliferation, causing the hypoplasia characteristic of TCS craniofacial anomalies.
Conflict of interest statement
Conflict of interest statement: No conflicts declared.
Figures
Fig. 1.
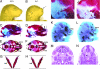
Analysis of DBA × C57BL/6 Tcof1+/− embryos. Comparison of wild-type (A) and mutant (B) newborn pups. Tcof1+/− mutant mice exhibit shortened and domed-shaped heads with frontonasal dysplasia (arrow). Skeletal staining of E17.5 wild-type (C, E, G, and I) and Tcof1+/− (D, F, H, and J) embryos reveals hypoplasia of numerous craniofacial bones including the nasal (n), frontal (f), premaxillary (pmx), maxillary (mx), mandibular (md), and temporal bones (t) together with cleft palate (arrow) in mutant embryos. In comparisons of wild-type (K) and mutant (L) embryo skeletal preparations, cranioskeletal hypoplasia is evident at E15.5 in the frontal, premaxillary, and maxillary elements. Transverse histological sections of E14.5 wild-type (M) and mutant (N) embryos highlight the absence of complete midline fusion (arrowhead) and the lack of a nasal septum (ns) and conchae (c) in Tcof1+/− embryos.
Fig. 2.
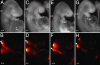
Neural crest cells at the level of rhombomere 2 (r2) and rhombomere 4 (r4) in wild-type (A, B, E, and F) and Tcof1+/− (C, D, G, and H) embryos were labeled with DiI (red) at E8.25 and cultured for 24 h. Fewer migrating neural crest cells from r2 and r4 populating the first branchial arch (ba1) (B and D) and second branchial arch (ba2) (F and H), respectively, in E9.5 Tcof1+/− (D and H) embryos were observed.
Fig. 3.
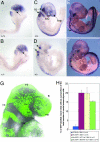
Neural crest cell quantification in Tcof1+/− embryos. Sox10 in situ hybridization in E8.5 (A and B), E9.5 (C and D), and E10.5 (E and F) wild-type (A, C, and E) and Tcof1+/− (B, D, and F) embryos reveals the deficiency in cranial neural crest cell contribution to the frontonasal prominence (fnp), first and second branchial arches (ba1 and ba2; arrows in A and B), cranial ganglia (cg; arrows in C and D) such as the trigeminal ganglion (t in E), and to the hypoglossopharyngeal and vagal ganglia posterior to the otic vesicle (double arrows in E and F). (G) E9.0 wild-type embryo obtained by intercrossing C57BL/6 Pax3GFP and DBA Tcof1+/− mice to quantify the number of migrating cranial neural crest cells in flow cytometry analyses. Neural crest cells (GFP-labeled) contribute to the frontonasal process (fnp), the mesenchyme surrounding the optic placode (op), and the maxillary (mx) and mandibular (md) prominences of the first branchial arch (ba1), as well as the more caudal branchial arches. fb, forebrain; mb, midbrain; hb, hindbrain. (H) Histogram demonstrating a 22.3% reduction in the relative proportion of GFP-labeled migrating neural crest cells to cranial mesenchyme cells in Tcof1+/− mutant (green bar) as compared with wild-type littermate (yellow bar) and positive control (red bar) embryos. The blue bar represents a negative control for GFP in wild-type DBA × C57BL/6 embryos.
Fig. 4.
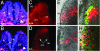
Neuroepithelial-specific apoptosis in Tcof1+/− embryos. TUNEL staining for apoptosis (green) in E8.25 wild-type (A) and Tcof1+/− (B) embryos and in a cultured Tcof1+/− embryo (C) in which forebrain-derived (fb), midbrain-derived (mb), and hindbrain-derived (hb) neural crest cells were labeled with DiI (red). Double-stained cells (yellow) in the neural plate (np) and not in migrating neural crest cells (red) highlight the specificity of apoptosis in the neuroepithelium of Tcof1+/− embryos. Caspase3 immunohistochemistry of parasagittal cranial sections from E8.75 wild-type (D) and Tcof1+/− (E) embryos revealed enhanced caspase3 activity in the neuroepithelium (arrows) of Tcof1+/− mutants.
Fig. 5.
Cellular analysis of Tcof1+/− embryos. (A_–_D) BrdU incorporation (pink in A and B) and Y10B immunostaining (red in C and D) of transverse sections of E8.75 wild-type (A and C) and Tcof1+/− (B and D) embryos. A reduction in cell proliferation (arrows) in the neuroepithelium (ne) and cranial mesenchyme (cm) corresponds with diminished production of mature 28S rRNA (arrows in D) in Tcof1+/− embryos. No proliferation anomalies were observed in the endoderm (arrowheads in A and B) or surface ectoderm (se). (E_–_H) TUNEL-stained embryos after homotopic hindbrain neural plate (np) transplantations of DiI-labeled (red) wild-type neuroepithelium in cultured E8.25 wild-type (E) and Tcof1+/− (F) embryos. Transplanted cells exhibit minimal apoptosis and generate migrating neural crest cells. (G and H) E8.75–E9.0 TUNEL-stained embryos after homotopic transplantations of DiI-labeled Tcof1+/− hindbrain neuroepithelium in cultured E8.25 wild-type (G) and Tcof1+/− (H) embryos. Transplanted cells exhibit significant apoptosis (arrows in G and H).
Similar articles
- Face off against ROS: Tcof1/Treacle safeguards neuroepithelial cells and progenitor neural crest cells from oxidative stress during craniofacial development.
Sakai D, Trainor PA. Sakai D, et al. Dev Growth Differ. 2016 Sep;58(7):577-85. doi: 10.1111/dgd.12305. Epub 2016 Aug 2. Dev Growth Differ. 2016. PMID: 27481486 Free PMC article. Review. - Treacher Collins syndrome: unmasking the role of Tcof1/treacle.
Sakai D, Trainor PA. Sakai D, et al. Int J Biochem Cell Biol. 2009 Jun;41(6):1229-32. doi: 10.1016/j.biocel.2008.10.026. Epub 2008 Nov 5. Int J Biochem Cell Biol. 2009. PMID: 19027870 Free PMC article. Review. - Treacher Collins syndrome.
Dixon J, Trainor P, Dixon MJ. Dixon J, et al. Orthod Craniofac Res. 2007 May;10(2):88-95. doi: 10.1111/j.1601-6343.2007.00388.x. Orthod Craniofac Res. 2007. PMID: 17552945 Review. - Increased levels of apoptosis in the prefusion neural folds underlie the craniofacial disorder, Treacher Collins syndrome.
Dixon J, Brakebusch C, Fässler R, Dixon MJ. Dixon J, et al. Hum Mol Genet. 2000 Jun 12;9(10):1473-80. doi: 10.1093/hmg/9.10.1473. Hum Mol Genet. 2000. PMID: 10888597
Cited by
- Re-focusing on Agnathia-Otocephaly complex.
Dubucs C, Chassaing N, Sergi C, Aubert-Mucca M, Attié-Bitach T, Lacombe D, Thauvin-Robinet C, Arpin S, Perez MJ, Cabrol C, Chen CP, Aziza J, Colin E, Martinovic J, Calvas P, Plaisancié J. Dubucs C, et al. Clin Oral Investig. 2021 Mar;25(3):1353-1362. doi: 10.1007/s00784-020-03443-w. Epub 2020 Jul 9. Clin Oral Investig. 2021. PMID: 32643087 Review. - Large deletions encompassing the TCOF1 and CAMK2A genes are responsible for Treacher Collins syndrome with intellectual disability.
Vincent M, Collet C, Verloes A, Lambert L, Herlin C, Blanchet C, Sanchez E, Drunat S, Vigneron J, Laplanche JL, Puechberty J, Sarda P, Geneviève D. Vincent M, et al. Eur J Hum Genet. 2014 Jan;22(1):52-6. doi: 10.1038/ejhg.2013.98. Epub 2013 May 22. Eur J Hum Genet. 2014. PMID: 23695276 Free PMC article. - A common cellular response to broad splicing perturbations is characterized by metabolic transcript downregulation driven by the Mdm2-p53 axis.
Varineau JE, Calo E. Varineau JE, et al. Dis Model Mech. 2024 Feb 1;17(2):dmm050356. doi: 10.1242/dmm.050356. Epub 2024 Mar 1. Dis Model Mech. 2024. PMID: 38426258 Free PMC article. - When ribosomes go bad: diseases of ribosome biogenesis.
Freed EF, Bleichert F, Dutca LM, Baserga SJ. Freed EF, et al. Mol Biosyst. 2010 Mar;6(3):481-93. doi: 10.1039/b919670f. Epub 2010 Jan 11. Mol Biosyst. 2010. PMID: 20174677 Free PMC article. Review. - Facial dysostoses: Etiology, pathogenesis and management.
Trainor PA, Andrews BT. Trainor PA, et al. Am J Med Genet C Semin Med Genet. 2013 Nov;163C(4):283-94. doi: 10.1002/ajmg.c.31375. Epub 2013 Oct 4. Am J Med Genet C Semin Med Genet. 2013. PMID: 24123981 Free PMC article. Review.
References
- Trainor P., Bronner-Fraser M., Krumlauf R. In: Handbook of Stem Cells: Embryonic Stem Cells. Lanza G., Weissman I., Thomson J., Pedersen R., Hogan B., Gearhart J., Blau H., Melton D., Moore M., Verfaillie C., et al., editors. Boston: Academic; 2004. pp. 219–232.
- Selleck M. A., Bronner-Fraser M. Development (Cambridge, U.K.) 1995;121:525–538. - PubMed
- Bronner-Fraser M., Fraser S. Neuron. 1989;3:755–766. - PubMed
- Moury J. D., Jacobson A. G. Dev. Biol. 1990;141:243–253. - PubMed
- Rollhauser-ter Horst J. Anat. Embryol. 1977;151:309–316. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 DE016082-05/DE/NIDCR NIH HHS/United States
- P50 DE 016215/DE/NIDCR NIH HHS/United States
- R01 DE 016082-01/DE/NIDCR NIH HHS/United States
- G0400264/MRC_/Medical Research Council/United Kingdom
- R01 DE016082-02/DE/NIDCR NIH HHS/United States
- R01 DE016082-03/DE/NIDCR NIH HHS/United States
- P50 DE016215/DE/NIDCR NIH HHS/United States
- R01 DE016082-01/DE/NIDCR NIH HHS/United States
- R01 DE016082-04/DE/NIDCR NIH HHS/United States
- R01 DE016082-06/DE/NIDCR NIH HHS/United States
- R01 DE016082/DE/NIDCR NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases