Spatiotemporal asymmetry of associative synaptic plasticity in fear conditioning pathways - PubMed (original) (raw)
Spatiotemporal asymmetry of associative synaptic plasticity in fear conditioning pathways
Ryong-Moon Shin et al. Neuron. 2006.
Abstract
Input-specific long-term potentiation (LTP) in afferent inputs to the amygdala serves an essential function in the acquisition of fear memory. Factors underlying input specificity of synaptic modifications implicated in information transfer in fear conditioning pathways remain unclear. Here we show that the strength of naive synapses in two auditory inputs converging on a single neuron in the lateral nucleus of the amygdala (LA) is only modified when a postsynaptic action potential closely follows a synaptic response. The stronger inhibitory drive in thalamic pathway, as compared with cortical input, hampers the induction of LTP at thalamo-amygdala synapses, contributing to the spatial specificity of LTP in convergent inputs. These results indicate that spike timing-dependent synaptic plasticity in afferent projections to the LA is both temporarily and spatially asymmetric, thus providing a mechanism for the conditioned stimulus discrimination during fear behavior.
Figures
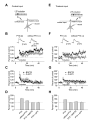
Figure 1
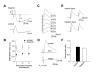
Properties of Spike Timing-Dependent LTP at the Cortico-Amygdala and Thalamo-Amygdala Synapses. (A) A schematic representation of the experimental design when cortical input was individually activated, showing the position of the recording and stimulation electrodes. (B) Summary graphs of the LTP experiments in cortical input in the presence of PTX (100 μM, mean ±SEM; n = 12) and without PTX (n = 9) in the bath solution. To induce LTP, 80 EPSPs were evoked at a frequency of 2 Hz; each EPSP was paired with action potential induced in a recorded neuron with 4-8 ms delay from the onset of the EPSP by short depolarizing current injections through the recording electrode. Insets show the average of 10 EPSPs recorded in individual experiments before and 35 min after the LTP-inducing stimulation (arrow) with PTX (left) or without PTX (right) in the bath solution. (C) LTP in cortical input is blocked by D-APV (50 μM, n = 4) in the external solution or when a high concentration of Ca2+ chelator BAPTA (10mM) is included in the recording pipette solution (n = 4). (D) Summary of LTP experiments (mean ±SEM) at cortico-amygdala synapses. (E) A schematic representation of the experimental design in thalamic pathway. (F) Summary graphs of the LTP experiments in thalamic input in the presence of PTX (n = 6) and without PTX (n = 5) in the bath solution. Insets show the average of 10 thalamo-amygdala EPSPs recorded in individual experiments before and 35 min after the LTP-inducing stimulation with PTX (left) or without PTX (right) in the bath solution. (G) LTP in thalamic input is blocked by D-APV (50 μM, n = 6) in the bath solution or by BAPTA (10mM) in the recording pipette solution (n = 10). (H) Summary of LTP experiments (mean ±SEM) at thalamo-amygdala synapses.
Figure 2
Time Window for the Induction of Spike Timing-Dependent Plasticity in the LA Is Asymmetric. (A) Summary of the percentage change in the amplitude of the cortico-amygdala EPSP 35-40 min after correlated pre- and postsynaptic activation when either the EPSP preceded action potential in a postsynaptic neuron (x-axis, positive time intervals) or AP preceded the EPSP (x-axis, negative time intervals), with or without PTX in the bath solution. Time intervals were determined between the onset of the EPSP and the peak of the postsynaptic AP. Each data point represents an individual experiment. (B) Summary of LTP experiments in cortical input as in (A) using positive time intervals of 0 to +8 ms, greater than +8 to +15 and greater than +15 to +30 ms or negative time intervals of 0 to -8 ms, greater than -8 ms to -15 ms and greater than -15 ms to -30 ms. The number of experiments is indicated for each time window. (C) Summary of the percentage change in the amplitude of the thalamo-amygdala EPSP 35-40 min after correlated pre- and postsynaptic activation (same experimental procedure as in A), with or without PTX in the bath solution. (D) Summary of LTP experiments in thalamic input as in (C) using positive time intervals of 0 to +8 ms, greater than +8 to +15 and greater than +15 to +30 ms or negative time intervals of 0 to -8 ms, greater than -8 ms to -15 ms and greater than -15 ms to -30 ms. (E) Summary graphs of all experiments in cortical input when AP preceded the EPSP (negative time intervals) in the presence of PTX (100 μM, mean ±SEM; n = 12) and without PTX (n = 12) in the bath solution. Traces are averages of 10 EPSPs recorded in individual experiments before and 30 min after the repetitive AP-EPSP pairing (arrow) with PTX (left) or without PTX (right) in the bath solution. (F) Summary graphs of all experiments in thalamic input when AP preceded the EPSP in the presence of PTX (n = 9) and without PTX (n = 11) in the bath solution. (G) Effects of the repetitive AP-EPSP paring with interpulse delays of -5 to -15 ms on the amplitude of the EPSP, expressed as the percentage change relative to the baseline value, when same number of the AP-EPSP pairs (80) was induced at four different frequencies (0.2 Hz, 0.5 Hz, 1 Hz, 2 Hz; x-axis, logarithmic scale) in cortical and thalamic inputs with PTX in the external solution.
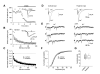
Figure 3
Quantal Parameters of Glutamatergic Synaptic Transmission at Cortical and Thalamic Pathways. (A) Evoked cortico-amygdala EPSCs were blocked by bath application of 20 μM CNQX at a holding potential of -70 mV. (B) The progressive block by MK-801 (40 μM) of the NMDA receptor EPSC recorded in the presence of CNQX at a holding potential of -40 mV. MK-801 was applied to the slice in the absence of presynaptic stimulation for 10 min. To measure rate of MK-801 block, the external capsule was stimulated at 0.1 Hz frequency. Inset shows the baseline NMDAR EPSC (1) and its block at the end of presynaptic stimulation in the presence of MK-801 (2). (C) Summary graphs of the experiments with MK-801 protocol in cortical (n = 9) and thalamic (n = 9) inputs. In each individual experiment, EPSC amplitudes were normalized by the first EPSC. Data points show mean ±SEM. (D) The amplitude of the AMPAR EPSC recorded either in cortical (left) or thalamic (right) inputs at a holding potential of -70 mV was significantly reduced, when Sr2+ was substituted for extracellular Ca2+. (E) Representative traces of the asynchronous quantal EPSCs evoked by stimulation (at arrow) of cortical (left) or thalamic (right) inputs. (F) Cumulative amplitude histograms of asynchronous quantal events recorded either at cortical (n = 10) or thalamic (n = 12) pathways. (G) Summary box plots of asynchronous EPSC data in cortical and thalamic inputs. The line inside the boxes marks the median, and the box boundaries indicate the 25th and 75th percentiles. The error bars indicate the 10th and 90th percentiles.
Figure 4
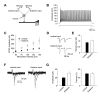
Convergent Cortical and Thalamic Inputs Do Not Differ in Subunit Composition of Synaptic NMDA Receptors. (A) Application of ifenprodil (10 μM) moderately depressed the isolated NMDAR EPSC recorded in the presence of CNQX at a holding potential of +50 mV in cortical input. Subsequent addition of D-APV (50 μM) completely blocked the EPSC. Insets show the averages of 10 NMDAR EPSCs recorded under baseline conditions (1), during ifenprodil-induced depression (2) and after D-APV application (3). (B) Averaged graphs of the experiments involving ifenprodil application as in (A) in cortical input (n = 5) and thalamic input (n = 5). Graphs were obtained by normalizing data in 60 s epochs to the mean value of the baseline (pre-drug) NMDAR EPSC (mean ±SEM). (C) Summary of the ifenprodil effects on the NMDAR EPSC at cortical and thalamic pathways. (D) Application of NVP-AAM077 (0.5 μM) significantly depressed the isolated NMDAR EPSC recorded at a holding potential of +50 mV in cortical input. Insets show the averages of 10 NMDAR EPSCs recorded under baseline conditions (1), during NVP-AAM077-induced depression (2) and after D-APV application (3). NVP-AAAM077 was a generous gift from Novartis Pharma AG (Switzerland). (E) Averaged graphs of the experiments involving NVP-AAM077 application as in (D) in cortical input (n = 8) and thalamic input (n = 7). (F) Summary of the NVP-AAM077 effects on the NMDAR EPSC at cortical and thalamic pathways (mean ±SEM).
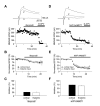
Figure 5
Evidence that Activation of Synaptic NR2B-Containing NMDA Receptors Accounts for the Induction of Spike Timing-Dependent LTP in Cortical and Thalamic Inputs. (A) LTP at cortico-amygdala pathway induced by the repetitive EPSP-AP paring using positive time intervals of +4 to +6 ms between the onset of the EPSP and the AP peak (n = 5) with PTX in the bath solution. Insets show the average of 10 EPSPs recorded before (1) and 35 min after (2) the EPSP-AP pairing procedure. (B) Effects of ifenprodil (10 μM, n = 9) and NVP-AAM077 (0.5 μM, n = 6) on spike timing-dependent LTP in cortical input. (C) Summary of LTP experiments in cortical input shown in (A) and (B) (mean ±SEM). (D) LTP at thalamo-amygdala pathway (n = 8) induced with PTX in the bath solution. Insets show the average of 10 EPSPs recorded before (1) and 35 min after (2) the EPSP-AP pairing procedure. (E) Effects of ifenprodil (n = 5) and NVP-AAM077 (n = 6) on spike timing-dependent LTP in thalamic input. (F) Summary of LTP experiments in thalamic input shown in (D) and (E) (mean ±SEM).
Figure 6
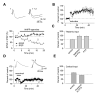
The EPSP/IPSP Sequences in the Cortical and Thalamic Inputs to the LA neuron. (A) Response of a cell in the LA to prolonged depolarizing current injection. Significant spike frequency accommodation observed in the recorded cell indicates that it is a pyramidal neuron. (B) Biphasic synaptic response recorded under current-clamp conditions in the LA neuron at -55 mV (control) in response to stimulation of cortical input. It consisted of the initial EPSP that was followed by the PTX-sensitive IPSP. A second trace (PTX) depicts an isolated EPSP when the IPSP was blocked with PTX. (C) The ESPS/IPSP sequence (control) was blocked by CNQX (20 μM), indicating the IPSP is disynaptic. (D) A schematic representation of the neural circuit for the EPSP/IPSP sequences in the LA. IN, interneuron; PN, principal neuron. (E) The EPSP/IPSP sequences recorded in cortical input over a range of membrane potentials (from -110 mV to -50 mV). Each trace represents the average of 10 responses. Dashed line marks the peak amplitude of the IPSP. (F) Dependence of the IPSP amplitude on membrane potential when responses were evoked either at cortical (n = 6) or thalamic pathway (n = 6). Values for the graph were obtained by normalizing the peak IPSP amplitude at each membrane potential to the amplitude of the IPSP recorded at -50 mV. Reversal potential of the IPSP was -71.3 ±4.0 mV (n = 6) and -72.1 ±3.7 mV (n = 6) in cortical and thalamic inputs, respectively (no significant difference between inputs, p = 0.89).
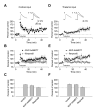
Figure 7
Feed-Forward GABAA Receptor-Mediated Inhibition of Principal Neurons Is Stronger In Thalamic Input. (A) Examples of the EPSP/IPSP sequences recorded at a membrane potential of -55 mV in convergent cortical and thalamic pathways. Traces show average of 10 responses. (B) Input-output curves for the monosynaptic AMPAR EPSP and disynaptic GABAAR IPSP recorded at convergent cortical and thalamic pathways presented as an IPSP/EPSP index. The IPSP amplitude was normalized by the EPSP amplitude for both pathways in each individual experiment (n = 9). Feed-forward IPSP is enhanced in thalamic input compared to cortical input. The intensity of stimulation at both pathways was gradually increased from the threshold stimulus required to elicit IPSP, that was determined in each individual experiment, with an increment of 50 μA to produce synaptic responses of increasing amplitude. First points represent responses evoked by the stimuli at the threshold + 50 μA. (C) Biphasic synaptic currents recorded at holding potentials of -70 mV to +30 mV in cortical input. (D) Disynaptic GABAAR IPSCs recorded with the cesium-based intrapipette solution at a holding potential of 0mV. Under these conditions, the AMPAR EPSC did not make a detectable contribution to the evoked response, since it was blocked by PTX (50 μM). The IPSC was also blocked by CNQX (20 μM, lower trace) providing evidence of its disynaptic origin. (E) Examples of the GABAAR ISPC pairs induced in cortical input with a 70-ms interstimulus interval. Traces show average of 10 responses. (F) Summary plot of paired-pulse ratio (PPR) data for the IPSCs recorded in cortical (n = 17) or thalamic (n = 15) inputs (mean ±SEM). PPR was calculated by dividing the second IPSC amplitude by the first IPSC amplitude
Figure 8
Enhanced Excitatory Drive to Interneurons in Thalamic Pathway Mediates Input Specific Differences in Inhibition. (A) A schematic representation of the experimental design when cortical and thalamic inputs to the same interneuron were alternately activated. (B) The recorded cell was identified as interneuron based on non-accommodating firing pattern in response to prolonged depolarizing current injection. (C) Input-output curves for the AMPAR EPSC recorded in interneurons at a holding potential of -70 mV in the presence of PTX (50 μM) at convergent cortical and thalamic pathways (n = 5). A leftward shift in the input-output curves obtained in thalamic input, as compared to cortical pathway, indicates a stronger inhibitory drive in thalamic input. 10-15 EPSCs were recorded and averaged for each stimulation intensity. (D) Examples of the AMPAR EPSCs pairs induced with a 50-ms interstimulus interval in convergent cortical (top) and thalamic (bottom) inputs. Traces show average of 10 responses. (E) Summary plot of paired-pulse ratio (PPR) data for the cortico-amygdala and thalamo-amygdala EPSCs recorded in cortical or thalamic inputs (n= 4, mean ±SEM). PPR was calculated by dividing the second EPSC amplitude by the first EPSC amplitude. (F) Superimposed traces of unitary EPSCs recorded in interneuron at -70 mV in response to stimulation of cortical (left) and thalamic (right) inputs. (G) Summary plot (mean ± SEM) of mean amplitude (left, n = 6) and failure rate (right, n = 6) estimates for unitary EPSCs in cortical and thalamic inputs to interneurons.
Figure 9
Input Specific Differences in Inhibition Account for Spatial Specificity of LTP at Convergent Inputs in the LA. (A) Effects of NHPP-spermine on biphasic synaptic responses elicited by stimulation of thalamic input and recorded under current-clamp conditions in the LA neuron at -55 mV. NHPP-spermine (5 μM, n = 4) produced significant depression of feed-forward disynaptic IPSP (open symbols), with a little effect on monosynaptic EPSP (closed symbols). Graphs were obtained by normalizing data in 60 s epochs to the mean value of the baseline (pre-drug) responses (mean ±SEM). Insets show the averages of 8 biphasic responses recorded under baseline conditions (1) and during NHPP-spermine-induced depression (2). (B) LTP at thalamo-amygdala pathway induced in the presence of NHPP-spermine (5 μM) by the repetitive EPSP-AP paring using positive time intervals of +4 to +6 ms between the onset of the EPSP and the AP peak without PTX in the bath solution (n = 4). (C) Summary of LTP experiments. LTP in thalamic input was induced either without (control, n = 5) or with 100 μM PTX (n = 6; same data as in 1H) and in the presence of spermine (n = 4; mean ±SEM). (D) Induction of LTP at cortico-amygdala pathway by the repetitive EPSP-AP paring is prevented in the presence of muscimol (2 μM). Insets show the averages of 8 responses recorded under baseline conditions (1) and after pairing (2). (E) Summary of LTP experiments in cortical input as in (D). LTP was induced either under control conditions (control: no PTX, n = 9; same data as in 1D) or in the presence of muscimol (n = 5; mean ±SEM).
Similar articles
- Fear conditioning occludes LTP-induced presynaptic enhancement of synaptic transmission in the cortical pathway to the lateral amygdala.
Tsvetkov E, Carlezon WA, Benes FM, Kandel ER, Bolshakov VY. Tsvetkov E, et al. Neuron. 2002 Apr 11;34(2):289-300. doi: 10.1016/s0896-6273(02)00645-1. Neuron. 2002. PMID: 11970870 - Fear memories induce a switch in stimulus response and signaling mechanisms for long-term potentiation in the lateral amygdala.
Schroeder BW, Shinnick-Gallagher P. Schroeder BW, et al. Eur J Neurosci. 2004 Jul;20(2):549-56. doi: 10.1111/j.1460-9568.2004.03517.x. Eur J Neurosci. 2004. PMID: 15233764 - Postsynaptic BDNF signalling regulates long-term potentiation at thalamo-amygdala afferents.
Meis S, Endres T, Lessmann V. Meis S, et al. J Physiol. 2012 Jan 1;590(1):193-208. doi: 10.1113/jphysiol.2011.220434. Epub 2011 Nov 14. J Physiol. 2012. PMID: 22083603 Free PMC article. - Synaptic mechanisms of associative memory in the amygdala.
Maren S. Maren S. Neuron. 2005 Sep 15;47(6):783-6. doi: 10.1016/j.neuron.2005.08.009. Neuron. 2005. PMID: 16157273 Review. - Synaptic transmission and plasticity in the amygdala. An emerging physiology of fear conditioning circuits.
Maren S. Maren S. Mol Neurobiol. 1996 Aug;13(1):1-22. doi: 10.1007/BF02740749. Mol Neurobiol. 1996. PMID: 8892333 Review.
Cited by
- Multimodal and Site-Specific Plasticity of Amygdala Parvalbumin Interneurons after Fear Learning.
Lucas EK, Jegarl AM, Morishita H, Clem RL. Lucas EK, et al. Neuron. 2016 Aug 3;91(3):629-43. doi: 10.1016/j.neuron.2016.06.032. Epub 2016 Jul 14. Neuron. 2016. PMID: 27427462 Free PMC article. - Hierarchical order of coexisting pre- and postsynaptic forms of long-term potentiation at synapses in amygdala.
Shin RM, Tully K, Li Y, Cho JH, Higuchi M, Suhara T, Bolshakov VY. Shin RM, et al. Proc Natl Acad Sci U S A. 2010 Nov 2;107(44):19073-8. doi: 10.1073/pnas.1009803107. Epub 2010 Oct 18. Proc Natl Acad Sci U S A. 2010. PMID: 20956319 Free PMC article. - Norepinephrine enables the induction of associative long-term potentiation at thalamo-amygdala synapses.
Tully K, Li Y, Tsvetkov E, Bolshakov VY. Tully K, et al. Proc Natl Acad Sci U S A. 2007 Aug 28;104(35):14146-50. doi: 10.1073/pnas.0704621104. Epub 2007 Aug 20. Proc Natl Acad Sci U S A. 2007. PMID: 17709755 Free PMC article. - Input-specific synaptic plasticity in the amygdala is regulated by neuroligin-1 via postsynaptic NMDA receptors.
Jung SY, Kim J, Kwon OB, Jung JH, An K, Jeong AY, Lee CJ, Choi YB, Bailey CH, Kandel ER, Kim JH. Jung SY, et al. Proc Natl Acad Sci U S A. 2010 Mar 9;107(10):4710-5. doi: 10.1073/pnas.1001084107. Epub 2010 Feb 22. Proc Natl Acad Sci U S A. 2010. PMID: 20176955 Free PMC article. - Enhanced cortico-amygdala efficacy and suppressed fear in absence of Rap1.
Pan BX, Vautier F, Ito W, Bolshakov VY, Morozov A. Pan BX, et al. J Neurosci. 2008 Feb 27;28(9):2089-98. doi: 10.1523/JNEUROSCI.5156-07.2008. J Neurosci. 2008. PMID: 18305243 Free PMC article.
References
- Bauer EP, LeDoux JP, Nader K. Fear conditioning and LTP in the lateral amygdala are sensitive to the same stimulus contingencies. Nat. Neurosci. 2001;4:687–688. - PubMed
- Bi G, Poo M. Synaptic modification by correlated activity: Hebb’s postulate revisited. Annu. Rev. Neurosci. 2001;24:139–166. - PubMed
- Bi G, Rubin J. Timing in synaptic plasticity: from detection to integration. Trends in Neurosci. 2005;28:222–228. - PubMed
- Bissiere S, Humeau Y, Luthi A. Dopamine gates LTP induction in lateral amygdala by suppressing feedforward inhibition. Nat. Neurosci. 2003;6:587–592. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 NS045625/NS/NINDS NIH HHS/United States
- DA15098/DA/NIDA NIH HHS/United States
- R03 DA015098/DA/NIDA NIH HHS/United States
- R01 NS044185/NS/NINDS NIH HHS/United States
- NS44185/NS/NINDS NIH HHS/United States
- NS045625/NS/NINDS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical