Tissue factor: a link between C5a and neutrophil activation in antiphospholipid antibody induced fetal injury - PubMed (original) (raw)
Tissue factor: a link between C5a and neutrophil activation in antiphospholipid antibody induced fetal injury
Patricia Redecha et al. Blood. 2007.
Abstract
Fetal loss in patients with antiphospholipid (aPL) antibodies has been ascribed to thrombosis of placental vessels. However, we have shown that inflammation, specifically activation of complement with generation of the anaphylotoxin C5a, is an essential trigger of fetal injury. In this study, we analyzed the role of the procoagulant molecule tissue factor (TF) in a mouse model of aPL antibody-induced pregnancy loss. We found that either blockade of TF with a monoclonal antibody in wild-type mice or a genetic reduction of TF prevented aPL antibody-induced inflammation and pregnancy loss. In response to aPL antibody-generated C5a, neutrophils express TF potentiating inflammation in the deciduas and leading to miscarriages. Importantly, we showed that TF in myeloid cells but not fetal-derived cells (trophoblasts) was associated with fetal injury, suggesting that the site for pathologic TF expression is neutrophils. We found that TF expression in neutrophils contributes to respiratory burst and subsequent trophoblast injury and pregnancy loss induced by aPL antibodies. The identification of TF as an important mediator of C5a-induced oxidative burst in neutrophils in aPL-induced fetal injury provides a new target for therapy to prevent pregnancy loss in the antiphospholipid syndrome.
Figures
Figure 1
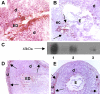
Expression of TF in decidual tissue of aPL-treated mice. (A,B,D,E) Pregnant Balb/c mice were treated on day 8 with aPL-IgG or IgG from a nonautoimmune individual (NH-IgG) and killed 2 hours later. Uteri were dissected and decidua sections were cut. (A,B) Decidua sections stained with an anti–mouse TF antibody. The chromogen was DAB (brown) and the counterstain was hematoxylin. In aPL-treated mice (A), there was extensive TF staining (brown color) in deciduas (d) and embryo debris (ED). In contrast, the decidual tissue from NH-IgG–treated mice showed minimal staining for TF (B) at the ectoplacental cone (ec) (arrows) and intact embryo (E). Original magnification × 40. (C) TF levels in the uterine contents of aPL- and NH-IgG–treated mice were measured by Western blotting. Lane 1 shows purified mouse TF standard (3 μg); lane 2, uterine content of an aPL-treated mouse; lane 3, uterine content of a NH-IgG–treated mouse. (D,E) Immunohistochemical analysis of fibrin in sections of uteri from aPL-treated (D) and NH-IgG–treated (e) mice. Fibrin was detected only at the decidua-uterine wall interface (arrows), and no difference in the staining intensity was observed between the 2 treatments. Original magnification × 10. d indicates decidua; u, uterine wall.
Figure 2
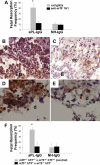
Inhibition of TF activity protects embryos from aPL-induced inflammation and embryonic death. (A) Pregnant female mice treated on days 8 and 12 with aPL-IgG received either an anti-TF mAb (1H1) or a rat IgG2a. On day 15 of pregnancy, mice were killed, uteri were dissected, and fetal resorption rates calculated (number of resorptions/number of fetuses + number of resorptions). There were 6 to 8 mice in each group. Mice that received aPL-IgG had a high frequency of fetal resorption compared with those that received normal human IgG (P < .001). Treatment with anti-TF mAb 1H1 led to a significant reduction in the frequency of fetal resorption compared with those mice receiving aPL-IgG (P < .01). Error bars here and in panel F are SD. (B,C) Immunohistochemical analysis of C3 in sections of deciduas from mice treated with either aPL-IgG + IgG2a or aPL-IgG + anti-TF antibody. (B) In deciduas from aPL-IgG + IgG2a–treated mice, C3 deposits (brown) were present throughout decidual tissue surrounding the necrotic residual embryonic debris (ED). (C) In contrast, in mice treated with aPL-IgG plus anti-TF antibody, C3 deposition was less intense and limited to the ectoplacental cone (ec) and the embryos remained intact (E). (D,E) Immunohistochemical analysis for neutrophils in sections of deciduas from aPL-IgG and aPL-IgG + anti-TF mAb 1H1. (D) Intense staining for neutrophils (brown) was observed in deciduas from mice treated with aPL-IgG plus IgG2a. In contrast, less neutrophil infiltration was observed in deciduas from mice that had received aPL-IgG + anti-TF mAb 1H1 (E). Counterstain: hematoxylin. Original magnification × 500. (F) Pregnant mice expressing low levels of TF (mTF−/−,hTF+) were treated on days 8 and 12 with aPL-IgG or NH-IgG. On day 15, fetal resorption rates were calculated. Approximately 40% of the embryos in control mice (mTF+/−,hTF+) treated with aPL-IgG were resorbed. Mice expressing low activity of TF showed a reduction in aPL-induced fetal resorption frequency compared with control mice (*P < .001).
Figure 3
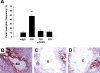
Only aPL antibodies that activate complement induce an increase in TF and fetal death in pregnant mice. (A) Pregnant mice were treated on days 8 and 12 of pregnancy with 2 different mouse monoclonal antibodies that recognize phospholipids on trophoblast cells. FB1 is an IgG2bκ that activates complement via classical pathway and FD1 is an IgG1κ that does not activate complement. A group of mice was treated with FB1 and the complement inhibitor Crry. On day 15, mice were killed and the frequency of fetal resorption was calculated (n = 6-7 mice/group). Mice that received FB1 had a high frequency of fetal resorption compared with those that received mIgG (P < .001). Administration of Crry protected mice from fetal death induced by FB1. Mice treated with FD1 did not show an increase in fetal resorption frequency that was observed with FB1. Error bars are SD. (B-D) Immunohistochemical analysis of decidual tissue from day 8 of pregnancy. Decidual tissue was processed for TF expression as described in “Materials and methods, Determination of TF functional activity in deciduas.” In FB1-treated mice (B), there was extensive TF deposition (brown) and embryo debris (ED). Treatment with Crry prevented the increase in TF expression in deciduas from FB1-treated mice (C). The decidual tissue from FD1-treated mice (D) showed minimal staining for TF in the ectoplacental cone (asterisk) and intact embryo comparable with mice treated with mIgG (not shown). Original magnification × 100.
Figure 4
C5aR interaction and neutrophils are required for the increase in TF expression in aPL-induced fetal damage. (A) Pregnant C3aR−/− and C3aR+/+ mice were treated with aPL-IgG or NH-IgG on days 8 and 12 of pregnancy. Fetal resorption frequency was calculated as described 19in “Materials and methods, Murine aPL-induced fetal loss model.” Treatment with aPL-IgG caused an increase in fetal resorptions in wild-type mice (*P < .001 versus NH-IgG). C3aR−/− mice were not protected from fetal loss induced by aPL-IgG. Error bars here and in panel E are SD. (B-D,F-H) Immunohistochemical analysis of decidual tissue of day 8 of pregnancy. Decidual tissue was processed for TF as described in legend for Figure 1. (B) Deciduas from C3aR−/− mice treated with aPL-IgG showed extensive TF staining and embryo debris (ED) comparable with wild-type mice treated with aPL-IgG (not shown). (C) Deciduas from C5−/− mice showed less TF staining limited to the ectoplacental cone (ec) and intact embryos (E), while in deciduas from C5+/+ mice (D) there was extensive TF deposition and embryonic debris (ED). (E) Pregnant C6+/+ and C6−/− mice were treated with aPL-IgG or NH-IgG on days 8 and 12 of pregnancy. Fetal resorption frequency was calculated as described 19in “Materials and methods, Murine aPL-induced fetal loss model.” Treatment of C6+/+ with aPL-IgG caused an increase in fetal resorptions (*P < .005 versus NH-IgG). C6−/− mice were not protected from fetal loss and showed an increase in TF expression induced by aPL-IgG (F). (G) Deciduas from C5aR−/− mice treated with aPL-IgG showed limited TF staining and intact embryos (E). (H) TF staining was reduced in deciduas from wild-type mice that had received anti-Gr before aPL-IgG treatment.
Figure 5
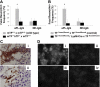
TF expression on neutrophils from aPL-treated mice depends on C5aR. (A) FACS analysis of TF expression on whole blood neutrophils from aPL-treated mice. The number of TF-positive neutrophils increased in aPL-treated C5aR+/+ mice in comparison with untreated mice (39% ± 16% vs 4% ± 2%, P < .005). The number of TF-positive neutrophils did not increase in aPL-treated C5aR−/− mice (9% ± 4%). (B) Immunohistochemical detection of TF on neutrophils from C5aR+/+ mice treated with aPL-IgG (i), C5aR−/− mice treated with aPL-IgG (ii), and untreated mice (iii). (C) FACS analysis of TF expression on whole blood neutrophils from aPL-treated mice. The number of TF-positive neutrophils increased in aPL-treated TFfloxed/floxed mice in comparison with untreated mice (26% ± 9% vs 7% ± 1%, P < .001). In contrast, the number of TF-positive neutrophils did not increase in TFfloxed/floxed/LysM-Cre mice treated with aPL-IgG (6% ± 2%). (D) Immunohistochemical detection of TF on neutrophils from TFfloxed/floxed mice treated with aPL-IgG (i), TFfloxed/floxed/LysM-Cre mice treated with aPL-IgG (ii), and untreated mice (iii). (E) FACS analysis of ROS production in neutrophils from aPL-treated mice. The number of ROS-positive neutrophils increased in aPL-treated TFfloxed/floxed mice in comparison with untreated mice (29% ± 9% vs 5% ± 1%, P < .01). The number of ROS-positive neutrophils did not increase TFfloxed/floxed/LysM-Cre mice treated with aPL-IgG (10% ± 3%).
Figure 6
TF expression by myeloid cells but not fetal-derived cells contributes to aPL-induced fetal loss. (A) Low TF female mice (mTF−/−,hTF+) mated with wild-type males (mTF+/+) and wild-type female mice mated with wild-type males (mTF+/+ × mTF+/+) were treated on days 8 and 12 with aPL-IgG or NH-IgG. On day 15, fetal resorption rates were calculated as described 19in “Materials and methods, Murine aPL-induced fetal loss model.” Approximately 40% of the embryos in wild-type matings treated with aPL-IgG were resorbed. Low TF female mice mated with wild-type males showed a reduction in aPL-induced fetal resorption frequency compared with wild-type mice (P < .005). Error bars here and in panel B are SD. (B) Pregnant TFfloxed/floxed and TFfloxed/floxed/LysM-Cre mice were treated with aPL-IgG or NH-IgG on days 8 and 12 of pregnancy. Fetal resorption frequency was calculated as described in “Materials and methods, Murine aPL-induced fetal loss model.” Treatment with aPL-IgG caused an increase in fetal resorptions in TFfloxed/floxed mice (*P < .001 versus NH-IgG). TFfloxed/floxed/LysM-Cre mice were protected from fetal loss induced by aPL-IgG. Fetal resorption frequency in these mice was comparable with TFfloxed/floxed mice treated with NH-IgG. (C) Immunohistochemical analysis for neutrophils in sections of deciduas from aPL-IgG–treated mice. Intense staining for neutrophils (brown color) was observed in deciduas from TFfloxed/floxed mice treated with aPL-IgG (Ci). In contrast, less neutrophil infiltration was observed in deciduas from TFfloxed/floxed/LysMCre mice that had received aPL-IgG (Cii). Counterstain: hematoxylin. Original magnification × 500. (D) Superoxide (O2−) generation in decidual tissue was determined using dihydroethidium fluorescence. aPL-induced O2− formation (Di) is attenuated in TFfloxed/floxed/LysM-Cre mice (Dii) and low TF mice (Diii) to a similar extent to NH-IgG–treated mice (Div). Original magnification × 800.
Figure 7
Mechanism of aPL-induced TF increase and fetal death. APL antibodies are preferentially targeted to the placenta where they activate complement leading to the generation of potent anaphylatoxin C5a. C5a attracts and activates neutrophils. As a result of C5a-C5aR interaction, neutrophils express TF. TF on neutrophils contribute to oxidative burst and subsequent trophoblast injury and ultimately fetal death.
Similar articles
- Tissue factor in antiphospholipid antibody-induced pregnancy loss: a pro-inflammatory molecule.
Girardi G, Mackman N. Girardi G, et al. Lupus. 2008 Oct;17(10):931-6. doi: 10.1177/0961203308094994. Lupus. 2008. PMID: 18827058 Free PMC article. Review. - Pravastatin prevents miscarriages in antiphospholipid antibody-treated mice.
Girardi G. Girardi G. J Reprod Immunol. 2009 Nov;82(2):126-31. doi: 10.1016/j.jri.2009.03.003. Epub 2009 Jun 24. J Reprod Immunol. 2009. PMID: 19556010 - Neutrophil activation by the tissue factor/Factor VIIa/PAR2 axis mediates fetal death in a mouse model of antiphospholipid syndrome.
Redecha P, Franzke CW, Ruf W, Mackman N, Girardi G. Redecha P, et al. J Clin Invest. 2008 Oct;118(10):3453-61. doi: 10.1172/JCI36089. J Clin Invest. 2008. PMID: 18802482 Free PMC article. - Role of tissue factor in the maternal immunological attack of the embryo in the antiphospholipid syndrome.
Girardi G. Girardi G. Clin Rev Allergy Immunol. 2010 Dec;39(3):160-5. doi: 10.1007/s12016-009-8187-1. Clin Rev Allergy Immunol. 2010. PMID: 19921475 Review. - Complement inhibition keeps mothers calm and avoids fetal rejection.
Girardi G. Girardi G. Immunol Invest. 2008;37(5):645-59. doi: 10.1080/08820130802191615. Immunol Invest. 2008. PMID: 18716942 Review.
Cited by
- How We Interpret Thrombosis with Thrombocytopenia Syndrome?
Yamada S, Asakura H. Yamada S, et al. Int J Mol Sci. 2024 May 1;25(9):4956. doi: 10.3390/ijms25094956. Int J Mol Sci. 2024. PMID: 38732176 Free PMC article. Review. - Complement biomarkers in the antiphospholipid syndrome - Approaches to quantification and implications for clinical management.
Cole MA, Gerber GF, Chaturvedi S. Cole MA, et al. Clin Immunol. 2023 Dec;257:109828. doi: 10.1016/j.clim.2023.109828. Epub 2023 Oct 31. Clin Immunol. 2023. PMID: 37913840 Review. - ADP: the missing link between thrombosis and hemolysis.
Gerber GF, Brodsky RA. Gerber GF, et al. Blood Adv. 2023 Oct 24;7(20):6364-6366. doi: 10.1182/bloodadvances.2023011186. Blood Adv. 2023. PMID: 37874560 Free PMC article. No abstract available. - The pathogenesis of obstetric APS: a 2023 update.
Mineo C, Shaul PW, Bermas BL. Mineo C, et al. Clin Immunol. 2023 Oct;255:109745. doi: 10.1016/j.clim.2023.109745. Epub 2023 Aug 23. Clin Immunol. 2023. PMID: 37625670 Free PMC article. Review. - Dysregulated low-density granulocyte contributes to early spontaneous abortion.
Ye H, Li L, Dong Y, Zheng Q, Sha Y, Li L, Yang P, Jia Y, Gu J. Ye H, et al. Front Immunol. 2023 Feb 23;14:1119756. doi: 10.3389/fimmu.2023.1119756. eCollection 2023. Front Immunol. 2023. PMID: 36911722 Free PMC article.
References
- Esmon CT. The interactions between inflammation and coagulation. Br J Haematol. 2005;131:417–430. - PubMed
- Chu AJ. Tissue factor upregulation drives a thrombosis-inflammation circuit in relation to cardiovascular complications. Cell Biochem Funct. 2006;24:173–192. - PubMed
- Strukova S. Blood coagulation-dependent inflammation: coagulation-dependent inflammation and inflammation-dependent thrombosis. Front Biosci. 2006;11:59–80. - PubMed
- Ruf Dorfleutner A, Riewald M. Specificity of coagulation factor signaling. J Thromb Haemost. 2003;1:1495–1503. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous