Kalirin-7 controls activity-dependent structural and functional plasticity of dendritic spines - PubMed (original) (raw)
Kalirin-7 controls activity-dependent structural and functional plasticity of dendritic spines
Zhong Xie et al. Neuron. 2007.
Abstract
Activity-dependent rapid structural and functional modifications of central excitatory synapses contribute to synapse maturation, experience-dependent plasticity, and learning and memory and are associated with neurodevelopmental and psychiatric disorders. However, the signal transduction mechanisms that link glutamate receptor activation to intracellular effectors that accomplish structural and functional plasticity are not well understood. Here we report that NMDA receptor activation in pyramidal neurons causes CaMKII-dependent phosphorylation of the guanine-nucleotide exchange factor (GEF) kalirin-7 at residue threonine 95, regulating its GEF activity, leading to activation of small GTPase Rac1 and rapid enlargement of existing spines. Kalirin-7 also interacts with AMPA receptors and controls their synaptic expression. By demonstrating that kalirin expression and spine localization are required for activity-dependent spine enlargement and enhancement of AMPAR-mediated synaptic transmission, our study identifies a signaling pathway that controls structural and functional spine plasticity.
Figures
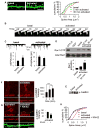
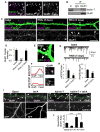
Figure 1. NMDA receptor-dependent spine enlargement is associated with Rac1 and CaMKII activation
(A) Quantification of spine areas of cultured cortical pyramidal neurons in basal and activated conditions: 5-min and 30-min activated are significantly different from basal, p<0.005. (B) Time-lapse imaging of single spine before (basal) and after (activated) NMDAR activation. (C) Traces show AMPA-mediated mEPSC recordings from neurons in basal and activated (30 min) conditions, *p<0.05. (D) Quantification of integrated intensities of Rac1-GTP bands in control (basal) and activated neurons, normalized for protein loading, **p<0.005. (E) Western blot of extracts of control and activated neurons with an anti-phospho-Thr286-CaMKII antibody. (F) Time-dependent phosphorylation of CaMKIIα/β in dendrites of cortical neurons upon NMDAR activation; immunostaining with anti phospho-CaMKIIα/β antibody, ***p<0.001. (G) Effects of KN-62 (10 μM) on NMDAR activation-dependent CaMKII phosphorylation and spine enlargement, **p<0.005. (H) Quantification of (G): activated is significantly different from basal, p<0.001. Scale bars: 10 μm.
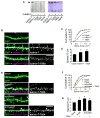
Figure 2. CaMKII interacts with and phosphorylates kalirin-7
(A) Coimmunoprecipitation of kalirin-7 with CaMKII and PSD-95 from rat forebrain homogenate; negative controls were IgG and myc antibodies. (B) Colocalization of CaMKII and kalirin-7 in spines of pyramidal neurons. (C) Western blot for phospho-Thr (p-Thr): constitutively active CaMKIIα (CaMKII-CA) phopshorylates kalirin-7, dependent on the presence of phosphatase inhibitors. Kalirin-7 was immunoprecipitated from hEK293 cells overexpressing myc-kalirin-7 with or without CaMKII-CA. D) Phosphorylation of kalirin-7 by wild-type CaMKII is blocked by KN-62. Cells were preincubated with or without 10 μM KN-62 before immunoprecipitation; Western blot with p-Thr and myc antibodies; quantification of integrated intensities of p-Thr bands, normalized for protein loading, *p<0.05. (E) NMDAR activation-dependent phosphorylation of kalirin-7 in neurons; kalirin was immunoprecipitated and analyzed by Western blotting for p-Thr. (F) Epitope-tagged kalirin-7 or truncated forms were overexpressed in hEK293 cells with CaMKII-CA; kalirin was immunoprecipitated and phosphoproteins were analyzed by Western blotting with a p-Thr antibody. (G) Purified CaMKII/calmodulin directly phosphorylates immunopurified kalirin-7 in vitro, in presence of purified CaMKII/calmodulin and Y-[32P]-ATP; arrowhead: phospho-kalirin-7 band, asterisk: non-specific band. Scale bar: 10 μm.
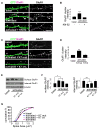
Figure 3. Identification of the phosphorylated site of kalirin-7
(A) Kalirin-7 is directly phosphorylated by CaMKII, but kalirin mutant T95A is not; arrowhead: phospho-kalirin-7 band. Kalirin-T95A (T95A) was immunopurified and analyzed as in Figure 2G. (B) Thr-95 is a key regulatory site for ability of kalirin-7 to induce spine enlargement. Spine morphology was analyzed on neurons (div 28) transfected with plasmids expressing GFP alone, or together with wild-type kalirin-7 or kalirin-7-T95A. (C). Quantification of spine areas in (B), p<0.001. (D). Quantification of linear spine densities in (B): *p<0.05. (E) Thr-95 is key regulatory site for activity-dependent spine enlargement. Spine morphology was analyzed as in (B), in activated or untreated control neurons. (F). Quantification of spine areas in (E), p<0.001. (G) Quantification of spine densities in (E), p<0.05. Scale bars: 10 μm.
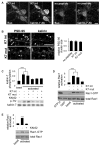
Figure 4. CaMKII and spine targeting are required for activity-dependent kalirin-7 phosphorylation and Rac1 activation
(A) A kalirin-7 interfering peptide is able to penetrate neurons: neurons were incubated with a fluorescein-tagged kalirin-7 interfering peptide (K7 int), a control peptide (K7 mut), or left untreated. Retained peptide was detected based on its tag (fluo) or using a kalirin-7 antibody. (B) K7 int (arrowheads) but not K7 mut displaces kalirin-7 from spines. PSD-95 is not displaced (arrows). (C) Activity-dependent phosphorylation of kalirin-7 in neurons requires its association with the postsynaptic density (PSD) and active CaMKII. Neurons were left untreated or preincubated with K7 int, K7 mut, or KN-62, and subjected to APV withdrawal; Western blot with p-Thr and kalirin antibodies: quantification of normalized p-Thr integrated intensities, ***p<0.001. (D) and (E) Neurons were left untreated or were preincubated with K7 int, K7 mut (D), or KN-62 (E), and subjected to APV withdrawal; quantification of normalized integrated intensities of Rac1-GTP bands,***p<0.001. Scale bars: 10 μm.
Figure 5. Kalirin-7 expression and spine localization are required for maintenance of mature spine morphology
(A) Expression of kalirin-7 in rat cerebral cortex during postnatal development. (B) Effects of kalirin knockdown on spine number in cortical pyramidal neurons. Neurons (div 24) were transfected with empty vector pGsuper (basal), pGsuper-RNAi (RNAi), pGsuper-RNAi together with a rescue plasmid (RNAi+rescue K7), and a mutated RNAi with 2 point mutations, for 5 days. (C) Quantification of spine linear densities in (B), ***p<0.00. (D) Time-dependent (3–5 days) effect of kalirin knockdown on spine areas and linear density. (E) Quantification of spine linear densities in (D), ***p<0.001. (F) Quantification of spine areas in (D), p<0.05. Quantification of K7 int and K7 mut on the areas of dendritic spines after incubation for 2 hours (G) and 2 days (H). p<0.05. Scale bar: 10 μm.
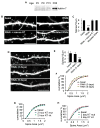
Figure 6. Kalirin-7 controls AMPA receptor maintenance in spines
(A) Kalirin-7 colocalizes with GluR1 in spines of pyramidal neurons (div 28). (B) Kalirin-7 coimmunoprecipitates with GluR1 from rat forebrain homogenate, while control IgG does not. (C) Effect of kalirin knockdown (3–5 days) on the maintenance of GluR1 clusters, quantified in (D), *p<0.001. (E) Partial knockdown of kalirin (3 days) causes altered localization of GluR1 clusters; in RNAi-treated neurons GluR1 clusters are often in spine neck/base (arrowheads). (F) Line scans of GluR1 immunofluorescence across representative spines and the adjacent dendritic shaft in basal (top) and RNAi-treated (bottom) neurons. (G) Effect of kalirin knockdown (5 days) on AMPAR-mediated basal synaptic transmission. Traces show representative recordings of AMPAR-mediated mEPSCs. (H) Quantification of mEPSC frequencies and amplitudes in (G), *p<0.05, **p<0.005. (I) Elevated GluR1 spine expression induced by kalirin-7 overexpression (2 days) is blocked by actin polymerization inhibitor latrunculin A (LAT-A, 5 μM, 24 hr). (J) Integrated intensities of GluR1 immunofluorescence in the dendritic spine and dendritic shaft were quantified and the spine/shaft ratios were plotted. Dashed lines: dendritic shafts; arrows: GluR1 cluster in the shaft, arrowheads: GluR1 cluster in spines; ***p<0.001. Scale bars: 10 μm (A, C, I); 1 μm (E, F).
Figure 7. NMDAR-dependent spine delivery of AMPA receptors depends on CaMKII and kalirin spine targeting
(A) Preincubation of neurons with KN-62 (activated+KN-62) blocks activity-induced (activated) delivery of GluR1 to spines. (B) Quantification of average integrated GluR1 cluster immunofluorescence intensities, normalized to basal, ***p<0.001. (C) Interfering peptide K7 int blocks the increase in GluR1 content in spine heads following activation of NMDARs, while control K7 mut peptide does not. (D) Quantification of GluR1 cluster integrated intensities (C), **p<0.005. (E) NMDAR activation increases cell-surface GluR1; incubation with K7 int interferes with the activity-dependent increase of surface GluR1 expression, K7 mut does not. Total amount of GluR1 expressed on cell surface was determined by cross-linking surface receptors. (F) Quantification of normalized GluR1 surface expression in cortical neurons, *p<0.05. (G) Quantification of spine areas in (F), p<0.001. Scale bars, 10 μm.
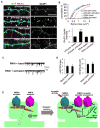
Figure 8. NMDAR-dependent spine enlargement and enhancement of AMPA receptor-mediated synaptic transmission depend on kalirin expression
(A) Partial kalirin knockdown (3 days) prevents NMDAR activation-induced spine enlargement and GluR1 spine delivery (RNAi activated), compared to neurons expressing control plasmid and subjected to APV withdrawal (pGsuper activated); basal spine morphologies and GluR1 cluster intensities were similar in unstimulated neurons expressing control plasmid (pGsuper basal) or RNAi (RNAi basal). (B) Quantification of spine areas and intensities in (A), ***p<0.001. (C) Effect of kalirin knockdown (3 days) on NMDAR activation-dependent enhancement of AMPAR-mediated synaptic transmission. Traces show representative recordings of AMPAR-mediated mEPSCs. (D) Quantification of AMPA mEPSC frequencies and amplitudes in (C). (E) Model of the regulation of activity-dependent synapse structural and functional plasticity by the NMDAR/CaMKII/kalirin-7/Rac1 pathway. Scale bars, 10 μm.
Similar articles
- Coordination of synaptic adhesion with dendritic spine remodeling by AF-6 and kalirin-7.
Xie Z, Photowala H, Cahill ME, Srivastava DP, Woolfrey KM, Shum CY, Huganir RL, Penzes P. Xie Z, et al. J Neurosci. 2008 Jun 11;28(24):6079-91. doi: 10.1523/JNEUROSCI.1170-08.2008. J Neurosci. 2008. PMID: 18550750 Free PMC article. - Scaffold protein X11α interacts with kalirin-7 in dendrites and recruits it to Golgi outposts.
Jones KA, Eng AG, Raval P, Srivastava DP, Penzes P. Jones KA, et al. J Biol Chem. 2014 Dec 19;289(51):35517-29. doi: 10.1074/jbc.M114.587709. Epub 2014 Nov 5. J Biol Chem. 2014. PMID: 25378388 Free PMC article. - The guanine nucleotide exchange factor (GEF) Asef2 promotes dendritic spine formation via Rac activation and spinophilin-dependent targeting.
Evans JC, Robinson CM, Shi M, Webb DJ. Evans JC, et al. J Biol Chem. 2015 Apr 17;290(16):10295-308. doi: 10.1074/jbc.M114.605543. Epub 2015 Mar 6. J Biol Chem. 2015. PMID: 25750125 Free PMC article. - Regulation of neuronal PKA signaling through AKAP targeting dynamics.
Dell'Acqua ML, Smith KE, Gorski JA, Horne EA, Gibson ES, Gomez LL. Dell'Acqua ML, et al. Eur J Cell Biol. 2006 Jul;85(7):627-33. doi: 10.1016/j.ejcb.2006.01.010. Epub 2006 Feb 28. Eur J Cell Biol. 2006. PMID: 16504338 Review. - Excitatory amino acid involvement in dendritic spine formation, maintenance and remodelling.
McKinney RA. McKinney RA. J Physiol. 2010 Jan 1;588(Pt 1):107-16. doi: 10.1113/jphysiol.2009.178905. Epub 2009 Nov 23. J Physiol. 2010. PMID: 19933758 Free PMC article. Review.
Cited by
- PAK1 protein expression in the auditory cortex of schizophrenia subjects.
Deo AJ, Goldszer IM, Li S, DiBitetto JV, Henteleff R, Sampson A, Lewis DA, Penzes P, Sweet RA. Deo AJ, et al. PLoS One. 2013 Apr 22;8(4):e59458. doi: 10.1371/journal.pone.0059458. Print 2013. PLoS One. 2013. PMID: 23613712 Free PMC article. - Rac GEF Dock4 interacts with cortactin to regulate dendritic spine formation.
Ueda S, Negishi M, Katoh H. Ueda S, et al. Mol Biol Cell. 2013 May;24(10):1602-13. doi: 10.1091/mbc.E12-11-0782. Epub 2013 Mar 27. Mol Biol Cell. 2013. PMID: 23536706 Free PMC article. - Developmental vulnerability of synapses and circuits associated with neuropsychiatric disorders.
Penzes P, Buonanno A, Passafaro M, Sala C, Sweet RA. Penzes P, et al. J Neurochem. 2013 Jul;126(2):165-82. doi: 10.1111/jnc.12261. Epub 2013 May 22. J Neurochem. 2013. PMID: 23574039 Free PMC article. Review. - Differential CaMKII regulation by voltage-gated calcium channels in the striatum.
Pasek JG, Wang X, Colbran RJ. Pasek JG, et al. Mol Cell Neurosci. 2015 Sep;68:234-43. doi: 10.1016/j.mcn.2015.08.003. Epub 2015 Aug 5. Mol Cell Neurosci. 2015. PMID: 26255006 Free PMC article. - The Rac-GAP alpha2-chimaerin regulates hippocampal dendrite and spine morphogenesis.
Valdez CM, Murphy GG, Beg AA. Valdez CM, et al. Mol Cell Neurosci. 2016 Sep;75:14-26. doi: 10.1016/j.mcn.2016.06.002. Epub 2016 Jun 11. Mol Cell Neurosci. 2016. PMID: 27297944 Free PMC article.
References
- Connor S, Williams PT, Armstrong B, Petit TL, Ivanco TL, Weeks AC. Long-term potentiation is associated with changes in synaptic ultrastructure in the rat neocortex. Synapse. 2006;59:378–382. - PubMed
- Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature. 2001;411:494–498. - PubMed
- Engert F, Bonhoeffer T. Dendritic spine changes associated with hippocampal long-term synaptic plasticity. Nature. 1999;399:66–70. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R56 MH071316/MH/NIMH NIH HHS/United States
- R01 MH071316-02S1/MH/NIMH NIH HHS/United States
- R01 MH071316-01A1/MH/NIMH NIH HHS/United States
- R01 MH071316-02/MH/NIMH NIH HHS/United States
- R01 MH071316-03/MH/NIMH NIH HHS/United States
- MH071316/MH/NIMH NIH HHS/United States
- R01 MH071316/MH/NIMH NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials