Organizational principles of human visual cortex revealed by receptor mapping - PubMed (original) (raw)
Organizational principles of human visual cortex revealed by receptor mapping
Simon B Eickhoff et al. Cereb Cortex. 2008 Nov.
Abstract
This receptorarchitectonic study of the human visual cortex investigated interareal differences in mean receptor concentrations and laminar distribution patterns of 16 neurotransmitter receptors in the dorsal and ventral parts of areas V1, V2, V3 as well as in adjoining areas V4 (ventrally) and V3A (dorsally). Both the functional hierarchy of these areas and a distinction between dorsal and ventral visual cortices were reflected by significant receptorarchitectonic differences. The observation that dorso-ventral differences existed in all extrastriate areas (including V2) is particularly important for the discussion about the relationship between dorsal and ventral V3 as it indicates that a receptorarchitectonic distinction between the ventral and dorsal visual cortices is present in but not specific to V3. This molecular specificity is mirrored by previously reported differences in retinal microstructure and functional differences as revealed in behavioral experiments demonstrating differential advantages for stimulus processing in the upper and lower visual fields. We argue that these anatomical and functional differences may be regarded as the result of an evolutionary optimization adapting to the processing of the most relevant stimuli occurring in the upper and lower visual fields.
Figures
Figure 1.
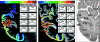
Contrast-enhanced, color-coded autoradiographs for the muscarinic M2 receptor (left) and the benzodiazepine binding sites (middle). Note that the calcarine section of V1 has not been included in the color coding and smoothing due to its obvious distortions caused by tangentional sectioning of a cortical folding. The location of cytoarchitectonic borders is shown for comparison in panel (right). Cortical areas were defined by receptorarchitectonic differences and cytoarchitectonic examination of adjacent cell body stained sections using quantitative methods (Schleicher et al. 2005) and established anatomical criteria (Amunts et al. 2000; Rottschy et al. 2007). White lines denote the borders between visual areas, whereas the boxes indicate the ROIs, from which receptor density profiles were extracted for all 16 analyzed receptors (Eickhoff, Schleicher, et al. 2007). For illustration, these profiles extracted from this particular section for the adrenergic α2 receptor are shown in the smaller inserts. Further details and additional examples for this approach are provided by Eickhoff et al. (2007) and Eickhoff, Rottschy, et al. (2007).
Figure 2.
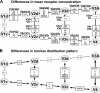
Significant (P < 0,05, family-wise error corrected) differences in mean receptor concentration (A) or laminar receptor distribution pattern (B) between visual areas, which are either adjacent or corresponding to each other in the retinotopic organization of early visual cortex.
Figure 3.
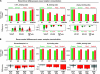
Synopsis of the mean concentrations for those receptors, which showed consistent differences reflecting cortical hierarchy (A) or visual stream distinction (B). All bars represent grand mean concentrations obtained from averaging the mean (across ROIs) concentrations of the 4 individual cases. The lower rows give the mean difference between hierarchically adjacent (A, separately by visual stream), respectively, corresponding areas (B). Significant differences are shown by colored bars, nonsignificant ones in gray. Lines denote the minimum and maximum difference observed in individual cases. Because the statistical analysis was carried out as a conjunction requiring significant effects in each case, these lines do not implicate statistical significance. Confidence intervals, however, would contradict the logic of multisubject conjunction studies needed to analyze the small sample sizes available for autoradiographic data.
Figure 4.
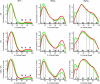
Laminar distribution patterns in ventral/dorsal V2 and V3 as well as V4/V3A for those receptors, showing consistent laminar differences between both visual streams (cf. Fig. 2). All displayed profiles are grand mean profiles obtained from averaging the mean (across ROIs) profiles of the 4 individual cases analyzed in this study.
Similar articles
- The retinotopic organization of primate dorsal V4 and surrounding areas: A functional magnetic resonance imaging study in awake monkeys.
Fize D, Vanduffel W, Nelissen K, Denys K, Chef d'Hotel C, Faugeras O, Orban GA. Fize D, et al. J Neurosci. 2003 Aug 13;23(19):7395-406. doi: 10.1523/JNEUROSCI.23-19-07395.2003. J Neurosci. 2003. PMID: 12917375 Free PMC article. - The projections from striate cortex (V1) to areas V2 and V3 in the macaque monkey: asymmetries, areal boundaries, and patchy connections.
Van Essen DC, Newsome WT, Maunsell JH, Bixby JL. Van Essen DC, et al. J Comp Neurol. 1986 Feb 22;244(4):451-80. doi: 10.1002/cne.902440405. J Comp Neurol. 1986. PMID: 3958238 - Reappraisal of DL/V4 boundaries based on connectivity patterns of dorsolateral visual cortex in macaques.
Stepniewska I, Collins CE, Kaas JH. Stepniewska I, et al. Cereb Cortex. 2005 Jun;15(6):809-22. doi: 10.1093/cercor/bhh182. Epub 2004 Sep 30. Cereb Cortex. 2005. PMID: 15459077 - Resolving the organization of the territory of the third visual area: a new proposal.
Kaas JH, Roe AW, Baldwin MK, Lyon DC. Kaas JH, et al. Vis Neurosci. 2015 Jan;32:E016. doi: 10.1017/S0952523815000152. Vis Neurosci. 2015. PMID: 26241553 Review. - Controversies about the visual areas located at the anterior border of area V2 in primates.
Gattass R, Lima B, Soares JG, Ungerleider LG. Gattass R, et al. Vis Neurosci. 2015;32:E019. doi: 10.1017/S0952523815000188. Vis Neurosci. 2015. PMID: 26581040 Free PMC article. Review.
Cited by
- The Neurotransmitter Receptor Architecture of the Mouse Olfactory System.
Lothmann K, Amunts K, Herold C. Lothmann K, et al. Front Neuroanat. 2021 Apr 23;15:632549. doi: 10.3389/fnana.2021.632549. eCollection 2021. Front Neuroanat. 2021. PMID: 33967704 Free PMC article. - Computational model links normalization to chemoarchitecture in the human visual system.
Aqil M, Knapen T, Dumoulin SO. Aqil M, et al. Sci Adv. 2024 Jan 5;10(1):eadj6102. doi: 10.1126/sciadv.adj6102. Epub 2024 Jan 3. Sci Adv. 2024. PMID: 38170784 Free PMC article. - Connectomics and new approaches for analyzing human brain functional connectivity.
Craddock RC, Tungaraza RL, Milham MP. Craddock RC, et al. Gigascience. 2015 Mar 25;4:13. doi: 10.1186/s13742-015-0045-x. eCollection 2015. Gigascience. 2015. PMID: 25810900 Free PMC article. Review. - Receptor architecture of macaque and human early visual areas: not equal, but comparable.
Rapan L, Niu M, Zhao L, Funck T, Amunts K, Zilles K, Palomero-Gallagher N. Rapan L, et al. Brain Struct Funct. 2022 May;227(4):1247-1263. doi: 10.1007/s00429-021-02437-y. Epub 2021 Dec 20. Brain Struct Funct. 2022. PMID: 34931262 Free PMC article. - Ant Colony Clustering for ROI Identification in Functional Magnetic Resonance Imaging.
Veloz A, Weinstein A, Pszczolkowski S, Hernández-García L, Olivares R, Muñoz R, Taramasco C. Veloz A, et al. Comput Intell Neurosci. 2019 Dec 26;2019:5259643. doi: 10.1155/2019/5259643. eCollection 2019. Comput Intell Neurosci. 2019. PMID: 32082371 Free PMC article.
References
- Alonso JM. Neural connections and receptive field properties in the primary visual cortex. Neuroscientist. 2002;8:443–456. - PubMed
- Amunts K, Malikovic A, Mohlberg H, Schormann T, Zilles K. Brodmann's areas 17 and 18 brought into stereotaxic space—where and how variable? Neuroimage. 2000;11:66–84. - PubMed
- Andrade da Costa BL, Hokoc JN. Photoreceptor topography of the retina in the New World monkey Cebus apella. Vision Res. 2000;40:2395–2409. - PubMed
- Callaway EM. Local circuits in primary visual cortex of the macaque monkey. Annu Rev Neurosci. 1998;21:47–74. - PubMed