A causal role for brain-derived neurotrophic factor in the homeostatic regulation of sleep - PubMed (original) (raw)
A causal role for brain-derived neurotrophic factor in the homeostatic regulation of sleep
Ugo Faraguna et al. J Neurosci. 2008.
Abstract
Slow-wave activity (SWA), the EEG power between 0.5 and 4 Hz during non-rapid eye movement (NREM) sleep, is one of the best characterized markers of sleep need, because it increases as a function of preceding waking duration and decreases during sleep, but the underlying mechanisms remain unknown. We hypothesized that SWA is high at sleep onset because it reflects the occurrence, during the previous waking period, of widespread synaptic potentiation in cortical and subcortical areas. Consistent with this hypothesis, we recently showed that the more rats explore, the stronger is the cortical expression of BDNF during wakefulness, and the larger is the increase in SWA during the subsequent sleep period. There is compelling evidence that BDNF plays a causal role in synaptic potentiation, and exogenous application of BDNF in vivo is sufficient to induce long-term increases in synaptic strength. We therefore performed cortical unilateral microinjections of BDNF in awake rats and measured SWA during the subsequent sleep period. SWA during NREM sleep was higher in the injected hemisphere relative to the contralateral one. The effect was reversible within 2 h, and did not occur during wakefulness or rapid eye movement sleep. Asymmetries in NREM SWA did not occur after vehicle injections. Furthermore, microinjections, during wakefulness, of a polyclonal anti-BDNF antibody or K252a, an inhibitor of BDNF TrkB receptors, led to a local SWA decrease during the following sleep period. These effects were also reversible and specific for NREM sleep. These results show a causal link between BDNF expression during wakefulness and subsequent sleep regulation.
Figures
Figure 1.
Experimental protocol. A, Schematic of a rat brain coronal section [B, +2.70 according to Paxinos and Watson (2005)] showing the position of the two frontal microinjection cannula/bipolar wire electrodes. B, ABC staining of the diffusion of BDNF (right) and anti-BDNF (left) ∼30 and 120 min after the injections, respectively. CC, Corpus callosum; I–II, cortical layers I and II; V–VI, cortical layers V and VI. C, Experimental design for the injection of BDNF and its vehicle. D, Experimental design for the injection of BDNF blockers and their corresponding control. BSL, Baseline day (24 h before the injection); EXP, experimental day. Every day the animals were gently handled for 30 min after light onset. The day of the injection, rats were first handled as usual, injected soon afterward, and then kept awake by exposure to novel objects for either 1 h (C) or 4 h (D). The light and dark bars indicate the light and dark period, respectively. Rats are nocturnal animals and therefore tend to sleep mostly during the day and stay awake during the night.
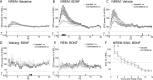
Figure 2.
Effects of BDNF on the EEG power spectrum. EEG power spectrum during NREM sleep in baseline (A) and after injection of BDNF (B) or vehicle (C). All data are mean ± SEM for the first 2 h after sleep onset. For each frequency bin (0.25 Hz), values are expressed as percentage of the NREM EEG power spectrum during the first 6 h of baseline (BSL), normalized for the mean power density in the 15–20 Hz range within each interval. The bottom panels show the statistical significance for each frequency bin, comparing the injected with the contralateral hemisphere (p < 0.05, Wilcoxon's nonparametric test). Note that the higher EEG power in the low frequencies after vehicle injections relative to baseline is expected, because rats were awake longer. D, EEG power spectrum during waking over the first 3 h after the injection of BDNF. E, EEG power spectrum during REM sleep during the first 2 h of sleep after the injection of BDNF. F, Time course of the changes in SWA (0.5–4 Hz) during NREM sleep after the injection of BDNF (Bonferroni's post hoc between injected and contralateral hemispheres, *p < 0.01, for the first two 1 h intervals, after two-way rANOVA; F(11,66) = 3.37; p = 0.000098). SWA is expressed as the mean value of relative NREM EEG power across frequencies ranging from 0.5 through 4 Hz. Data in all panels refer to n = 7 rats.
Figure 3.
Effects of K252a on the EEG power spectrum. EEG power spectrum during NREM sleep in baseline (A) and after injection of K252a (B) or vehicle (C). All data are mean ± SEM for the first 2 h after sleep onset. For each frequency bin (0.25 Hz), values are expressed as percentage of the NREM EEG power spectrum during the first 6 h of baseline (BSL), normalized for the mean power density in the 15–20 Hz range within each interval. The bottom panels show the statistical significance for each frequency bin, comparing the injected with the contralateral hemisphere (p < 0.05, Wilcoxon's nonparametric test). Note that the higher EEG power in the low frequencies after vehicle injections relative to baseline is expected, because rats were awake longer. D, EEG power spectrum during waking for the first 3 h after the injection of K252a. E, EEG power spectrum during REM sleep during the first 2 h of sleep after injection of K252a. F, Time course of the changes in SWA (0.5–4 Hz) during NREM sleep after the injection of K252a (Bonferroni's post hoc between injected and contralateral hemispheres, *p < 0.01, for the first four 1 h intervals, after two-way rANOVA; F(11,55) = 11.79; p < 0.00001). SWA is expressed as the mean value of relative normalized NREM EEG power across frequencies ranging from 0.5 through 4 Hz. Data in all panels refer to n = 6 rats.
Figure 4.
Effects of the anti-BDNF antibody on the EEG power spectrum. EEG power spectrum during NREM sleep in baseline (A) and after injection of anti-BDNF (B) or anti-NT3 (C). All data are mean ± SEM for the first 2 h after sleep onset. For each frequency bin (0.25 Hz), values are expressed as percentage of the NREM EEG power spectrum during the first 6 h of baseline (BSL), normalized for the mean power density in the 15–20 Hz range within each interval. The bottom panels show the statistical significance for each frequency bin, comparing the injected with the contralateral hemisphere (p < 0.05, Wilcoxon's nonparametric test). Note that the higher EEG power in the low frequencies after vehicle injections relative to baseline is expected, because rats were awake longer. D, EEG power spectrum during waking for the first 3 h after the injection of anti-BDNF. E, EEG power spectrum during REM sleep during the first 2 h of sleep after the injection of anti-BDNF. F, Time course of the changes in NREM SWA after the injection of anti-BDNF (Bonferroni's post hoc between injected and contralateral hemispheres, *p < 0.01, for the first four 1 h intervals after two-way rANOVA; F(11,66) = 7.3; p < 0.00001). SWA is expressed as the mean value of relative normalized NREM EEG power across frequencies ranging from 0.5 through 4 Hz. Data in all panels refer to n = 7 rats.
Similar articles
- Exploratory behavior, cortical BDNF expression, and sleep homeostasis.
Huber R, Tononi G, Cirelli C. Huber R, et al. Sleep. 2007 Feb;30(2):129-39. doi: 10.1093/sleep/30.2.129. Sleep. 2007. PMID: 17326538 - Unilateral cortical spreading depression affects sleep need and induces molecular and electrophysiological signs of synaptic potentiation in vivo.
Faraguna U, Nelson A, Vyazovskiy VV, Cirelli C, Tononi G. Faraguna U, et al. Cereb Cortex. 2010 Dec;20(12):2939-47. doi: 10.1093/cercor/bhq041. Epub 2010 Mar 26. Cereb Cortex. 2010. PMID: 20348156 Free PMC article. - Synaptic potentiation and sleep need: clues from molecular and electrophysiological studies.
Hanlon EC, Vyazovskiy VV, Faraguna U, Tononi G, Cirelli C. Hanlon EC, et al. Curr Top Med Chem. 2011;11(19):2472-82. doi: 10.2174/156802611797470312. Curr Top Med Chem. 2011. PMID: 21906017 Review. - Electrophysiological correlates of sleep homeostasis in freely behaving rats.
Vyazovskiy VV, Cirelli C, Tononi G. Vyazovskiy VV, et al. Prog Brain Res. 2011;193:17-38. doi: 10.1016/B978-0-444-53839-0.00002-8. Prog Brain Res. 2011. PMID: 21854953 Free PMC article. Review.
Cited by
- Plasma and cerebrospinal fluid proteomic signatures of acutely sleep-deprived humans: an exploratory study.
Vaquer-Alicea A, Yu J, Liu H, Lucey BP. Vaquer-Alicea A, et al. Sleep Adv. 2023 Nov 14;4(1):zpad047. doi: 10.1093/sleepadvances/zpad047. eCollection 2023. Sleep Adv. 2023. PMID: 38046221 Free PMC article. - Neurogenesis in the rat neonate's hippocampus with maternal short-term REM sleep deprivation restores by royal jelly treatment.
Khodaverdiloo A, Farhadi M, Jameie M, Jameie SB, Pirhajati V. Khodaverdiloo A, et al. Brain Behav. 2021 Dec;11(12):e2423. doi: 10.1002/brb3.2423. Epub 2021 Nov 22. Brain Behav. 2021. PMID: 34807519 Free PMC article. - Delta wave power: an independent sleep phenotype or epiphenomenon?
Davis CJ, Clinton JM, Jewett KA, Zielinski MR, Krueger JM. Davis CJ, et al. J Clin Sleep Med. 2011 Oct 15;7(5 Suppl):S16-8. doi: 10.5664/JCSM.1346. J Clin Sleep Med. 2011. PMID: 22003323 Free PMC article. Review. - Variations in COMT and NTRK2 Influence Symptom Burden in Women Undergoing Breast Cancer Treatment.
Young EE, Kelly DL, Shim I, Baumbauer KM, Starkweather A, Lyon DE. Young EE, et al. Biol Res Nurs. 2017 May;19(3):318-328. doi: 10.1177/1099800417692877. Epub 2017 Feb 16. Biol Res Nurs. 2017. PMID: 28205449 Free PMC article. Clinical Trial. - Time to be SHY? Some comments on sleep and synaptic homeostasis.
Tononi G, Cirelli C. Tononi G, et al. Neural Plast. 2012;2012:415250. doi: 10.1155/2012/415250. Epub 2012 Apr 29. Neural Plast. 2012. PMID: 22619736 Free PMC article.
References
- Achermann P, Borbely AA. Mathematical models of sleep regulation. Front Biosci. 2003;8:s683–s693. - PubMed
- Baker-Herman TL, Fuller DD, Bavis RW, Zabka AG, Golder FJ, Doperalski NJ, Johnson RA, Watters JJ, Mitchell GS. BDNF is necessary and sufficient for spinal respiratory plasticity following intermittent hypoxia. Nat Neurosci. 2004;7:48–55. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources