Bidirectional transport of amino acids regulates mTOR and autophagy - PubMed (original) (raw)
. 2009 Feb 6;136(3):521-34.
doi: 10.1016/j.cell.2008.11.044.
Philip Bergman, Bailin Zhang, Ellen Triantafellow, Henry Wang, Beat Nyfeler, Haidi Yang, Marc Hild, Charles Kung, Christopher Wilson, Vic E Myer, Jeffrey P MacKeigan, Jeffrey A Porter, Y Karen Wang, Lewis C Cantley, Peter M Finan, Leon O Murphy
Affiliations
- PMID: 19203585
- PMCID: PMC3733119
- DOI: 10.1016/j.cell.2008.11.044
Bidirectional transport of amino acids regulates mTOR and autophagy
Paul Nicklin et al. Cell. 2009.
Abstract
Amino acids are required for activation of the mammalian target of rapamycin (mTOR) kinase which regulates protein translation, cell growth, and autophagy. Cell surface transporters that allow amino acids to enter the cell and signal to mTOR are unknown. We show that cellular uptake of L-glutamine and its subsequent rapid efflux in the presence of essential amino acids (EAA) is the rate-limiting step that activates mTOR. L-glutamine uptake is regulated by SLC1A5 and loss of SLC1A5 function inhibits cell growth and activates autophagy. The molecular basis for L-glutamine sensitivity is due to SLC7A5/SLC3A2, a bidirectional transporter that regulates the simultaneous efflux of L-glutamine out of cells and transport of L-leucine/EAA into cells. Certain tumor cell lines with high basal cellular levels of L-glutamine bypass the need for L-glutamine uptake and are primed for mTOR activation. Thus, L-glutamine flux regulates mTOR, translation and autophagy to coordinate cell growth and proliferation.
Figures
Figure 1
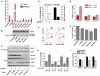
Glutamine-Dependent Regulation of mTORC1 Effector Signaling (A) HeLa cells deprived of growth factors and nutrients (starve) were treated with DMEM or EAA for 60 min. The phosphorylation of S6K1Thr389 and S6Ser235/236 and S6240/244, and total levels of S6K1 (70 kDa cytoplasmic and 85 kDa nuclear isoforms are indicated) were assessed using western blotting. (B) HeLa cells were starved as in (A) and then treated with the indicated concentrations of EAA and/or L-glutamine for 60 min. EAA concentration (x-fold) was based on final concentration in DMEM (1×). Phosphorylation of S6Ser235/236 was quantitated using the in-cell western assay and expressed as fold-increase compared to starved cells. (C) Starved HeLa cells were incubated with DMEM or L-glutamine-deficient DMEM (DMEM-GLN) for 60 min and the phosphorylation of S6Ser235/236 was quantitated. (D) Starved HeLa cells were treated for 60 min as indicated and the phosphorylation of S6Ser235/236 was quantitated. 1 mM L- or D-glutamine was used. (E) Starved HeLa cells were treated as indicated and the phosphorylation of S6K1Thr389 was quantitated as described in the supplement. (F) Starved HeLa cells were treated as indicated for 60 min and levels of 4EBP1 and eIF4G associated with m7 GTP beads analyzed using western blotting. (G) S2 or S2-R Drosophila cells starved of serum and amino acids were then treated with amino acids in the presence or absence of L-glutamine. Data shown are representative of three independent experiments. Data with error bars represent mean ± SD.
Figure 2
L-Glutamine Integrates EAA and Growth Factor Signaling to mTORC1 and Regulates the Temporal Dynamics of S6K1 (A) Starved HeLa cells were preincubated with L-glutamine (GLN preload) or starve medium (–) for 1 hr prior to adding EAA or EAA/GLN for the times indicated. Phosphorylation of S6K1 was analyzed by western blotting. (B) Cells were pretreated with either L-glutamine (G) or EAA (E) for 1 hr and incubated with EAA, L-glutamine or EAA/GLN for 15 min prior to lysis. (C) Cells were treated as in (A) except that insulin (100 nM) or EGF (25 ng/mL) were included as indicated. (D) Cells pretreated with either L-glutamine or starve medium for 1 hr were then incubated with EAA or EAA/GLN, respectively, for the indicated times. (E) Cells pretreated for 1 hr with 10 mM L-glutamine, L-glutamic acid (GLU) or α-ketoglutaric acid (αKG), were washed and then incubated with starve or EAA-containing medium for 15 min. (F) Cells pretreated with either 10 mM L-glutamine or 13 nonessential amino acids (NEAA) for 1 hr, were washed and then incubated with starve or EAA-containing medium for 15 min. (G) Starved HeLa cells were treated as indicated for 1 hr and lysed. All data is representative of at least three independent experiments.
Figure 3
Pharmacological Modulation of SLC1A5 and SLC7A5 Inhibits mTOR Regulation (A) Starved HeLa cells were treated as indicated in the presence of increasing concentrations of GPNA (1 hr). (B) Starved cells were treated for 60 min and the cap translation complex analyzed. 10 mM GPNA was used. (C) Cells were pretreated (preload) for 60 min with L-glutamine in the presence or absence of GPNA prior to treatment with EAA for 15 min in the presence or absence of GPNA. (D and E) Starved HeLa cells were treated with EAA containing 1 mML-glutamine (EAA/GLN) and 100 nM insulin in the absence or presence of BCH for 60 min. A nonspecific reactive band (*) is indicated. (F) The phosphorylation of S6Ser235/236 was quantitated following treatment with the indicated concentrations (X) of EAA in the presence of 1mML-glutamine and D-phenylalanine. (G) Starved HeLa cells were treated as indicated in the presence and absence of 50 mM D-phenylalanine (D-Phe). (H) Cells were pretreated for 60 min as indicated and then treated with EAA for 15 min in the presence or absence of D-Phe. Data with error bars represent mean ± SD.
Figure 4
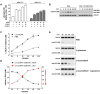
Bidirectional Transport of L-Glutamine and EAA Is Upstream of mTOR Activation (A) Starved cells were preincubated with L-glutamine or starve medium (–) for 1 hr prior to adding EAA or starve medium (15 min). Conditioned medium (CM) was removed and added to naive starved cells for 1 hr (lanes 1-5) and S6K1 phosphorylation analyzed. Phosphorylation of S6K1 in an extract from EAA-treated L-glutamine primed cells is shown (lane 6). (B) Conditioned medium was prepared as in (A) except collection was 2.5, 5, 10, and 15 min after adding EAA. (C) LC/MS/MS ion chromatograms of L-glutamine in conditioned medium taken at 2.5 min from cells pretreated with starve medium (PBS) prior to EAA (left trace), pretreated with L-glutamine prior to PBS (middle trace) or pretreated with L-glutamine prior to EAA (right trace). (D) Starved HeLa cells were pretreated with labeled L-glutamine for 1 hr, washed, and then treated with EAA containing 0.4 mM labeled L-leucine (L-Leu/EAA) for various times (minutes). The absolute levels of labeled L-glutamine in conditioned medium (black symbols) and cell extracts (red symbols) was quantitated using LC/MS/MS. (E) Starved cells were treated as in (C) and the L-glutamine efflux (fold-change in medium concentration) after treatment with PBS, EAA or insulin for 2.5 min quantitated. (F) Cells were treated as in (D) and after 2.5 min the absolute levels of effluxed labeled L-glutamine (black) and labeled L-leucine in cells (red) was quantitated. (G) Cells pretreated with 10 mM labeled L-glutamine or labeled L-glutamic acid for 1 hr were washed, treated with EAA containing labeled L-leucine for 2.5 min and extracted. (H) Starved cells were treated with labeled L-glutamine for 1 hr in the absence or presence of 10 mM GPNA. The absolute levels of L-glutamine in cell extracts were quantitated. (I) Cells were treated with labeled L-glutamine for 1 hr, washed and incubated with 0.4 mM labeled L-leucine in the absence and presence of BCH or D-Phe for 2.5 min. Data with error bars represent mean ± SD.
Figure 5
RNAi-Mediated Downregulation of SLC1A5, SLC3A2, or SLC7A5 Inhibits Amino Acid Transport and mTOR Pathway Activity (A) HeLa cells transfected with the indicated siRNAs were amino acid starved (open bars) and treated for 60 min with EAA containing 1 mM L-glutamine (red bars). Phosphorylation of S6Ser235/236 was quantitated using the Delfia readout. (B) After siRNA transfection (96 hr) S6K1 phosphorylation was analyzed. (C) Cells transfected with siRNAs were treated as indicated (1 hr) and the cap translation complex analyzed. (D) Cells transfected with scramble or SLC1A5_1 siRNA were starved and preincubated with labeled L-glutamine (1 hr). Cells were extracted for quantitation of labeled L-glutamine uptake (open bars) or treated with labeled L-leucine/EAA (2.5 min) for quantitation of effluxed labeled L-glutamine (filled bars). (E) Cells were transfected with the indicated siRNAs, starved and pretreated with labeled L-glutamine (hatched bars) or starve medium (filled bars) for 1 hr, washed and then treated with 0.4 mM labeled L-leucine for 2.5 min. Cell extracts were processed for absolute quantitation of L-leucine. (F) HeLa cells were transfected with siRNAs (nonsilencing, NS) for 72 hr and collected for cell size quantitation. (G) Cell size data from three independent experiments (mean ± SEM) is shown. (H) HeLa cells were transfected with scramble, mTOR, SLC1A5 or SLC7A5 siRNAs in the presence or absence of a TSC2 siRNA and processed for cell size quantitation. (I) Vector-expressing (mock) or RNAi-insensitive GFP-SLC1A5-expressing HeLa cells (clones L1 and L2) were transfected with siRNAs and processed for cell size quantitation. The data shown (mean ± SD) represent three independent experiments.
Figure 6
Efflux of Endogenous L-Glutamine Regulates S6K1 Activation in MCF7 Cells (A) MCF7 cells were amino acid starved, treated as indicated (1 hr) and processed for quantitation of S6 phosphorylation. (B) Starved MCF7 cells were treated with EAA in the absence or presence of 50 mM D-Phe for various times. (C) Starved MCF7 cells were treated with labeled L-leucine/EAA (filled circles) or starve medium (stars) for various times and conditioned medium processed for quantitation of L-glutamine. (D) MCF7 cells were treated as in (C) and extracted at the times indicated for quantitation of L-glutamine (black circles) or labeled L-leucine (red circles). (E) Starved MCF7 cells were treated as indicated and S6K1 activation analyzed using western blotting. Data with error bars represent mean ± SD.
Figure 7
L-Glutamine and SLC1A5 Regulate Autophagy (A) Confocal images of mCherry-eGFP-LC3a distribution before (+) and after (−) L-glutamine starvation are shown. (B) Quantitation of autophagy flux in RT112 cells. (C) RT112 cells transfected with the individual siRNAs for 72 hr were serum starved for 24 hr and processed for autophagy flux quantitation. Rapamycin was added to scrambled siRNA transfected cells during the starvation period. (D) EAA regulation of mTOR signaling, cell growth and suppression of autophagy requires intracellular uptake and subsequent efflux of L-glutamine (SLC3A2 dimerized with SLC7A5 is shown in yellow). All data (mean ± SD) are representative of at least three independent experiments.
Comment in
- An amino acid shuffle activates mTORC1.
Cohen A, Hall MN. Cohen A, et al. Cell. 2009 Feb 6;136(3):399-400. doi: 10.1016/j.cell.2009.01.021. Cell. 2009. PMID: 19203575
Similar articles
- An amino acid shuffle activates mTORC1.
Cohen A, Hall MN. Cohen A, et al. Cell. 2009 Feb 6;136(3):399-400. doi: 10.1016/j.cell.2009.01.021. Cell. 2009. PMID: 19203575 - The glutamine transporter ASCT2 (SLC1A5) promotes tumor growth independently of the amino acid transporter LAT1 (SLC7A5).
Cormerais Y, Massard PA, Vucetic M, Giuliano S, Tambutté E, Durivault J, Vial V, Endou H, Wempe MF, Parks SK, Pouyssegur J. Cormerais Y, et al. J Biol Chem. 2018 Feb 23;293(8):2877-2887. doi: 10.1074/jbc.RA117.001342. Epub 2018 Jan 11. J Biol Chem. 2018. PMID: 29326164 Free PMC article. - SLC1A5 glutamine transporter is a target of MYC and mediates reduced mTORC1 signaling and increased fatty acid oxidation in long-lived Myc hypomorphic mice.
Zhao X, Petrashen AP, Sanders JA, Peterson AL, Sedivy JM. Zhao X, et al. Aging Cell. 2019 Jun;18(3):e12947. doi: 10.1111/acel.12947. Epub 2019 Mar 25. Aging Cell. 2019. PMID: 30909319 Free PMC article. - Targeting SLC1A5 and SLC3A2/SLC7A5 as a Potential Strategy to Strengthen Anti-Tumor Immunity in the Tumor Microenvironment.
Nachef M, Ali AK, Almutairi SM, Lee SH. Nachef M, et al. Front Immunol. 2021 Apr 19;12:624324. doi: 10.3389/fimmu.2021.624324. eCollection 2021. Front Immunol. 2021. PMID: 33953707 Free PMC article. Review. - Regulation of autophagy by amino acids and MTOR-dependent signal transduction.
Meijer AJ, Lorin S, Blommaart EF, Codogno P. Meijer AJ, et al. Amino Acids. 2015 Oct;47(10):2037-63. doi: 10.1007/s00726-014-1765-4. Epub 2014 Jun 1. Amino Acids. 2015. PMID: 24880909 Free PMC article. Review.
Cited by
- Investigating the role of GLUL as a survival factor in cellular adaptation to glutamine depletion via targeted stable isotope resolved metabolomics.
Bayram Ș, Razzaque YS, Geisberger S, Pietzke M, Fürst S, Vechiatto C, Forbes M, Mastrobuoni G, Kempa S. Bayram Ș, et al. Front Mol Biosci. 2022 Aug 12;9:859787. doi: 10.3389/fmolb.2022.859787. eCollection 2022. Front Mol Biosci. 2022. PMID: 36032676 Free PMC article. - Inhibition of glutaminolysis in combination with other therapies to improve cancer treatment.
Shen YA, Chen CL, Huang YH, Evans EE, Cheng CC, Chuang YJ, Zhang C, Le A. Shen YA, et al. Curr Opin Chem Biol. 2021 Jun;62:64-81. doi: 10.1016/j.cbpa.2021.01.006. Epub 2021 Mar 12. Curr Opin Chem Biol. 2021. PMID: 33721588 Free PMC article. Review. - Deregulated Myc requires MondoA/Mlx for metabolic reprogramming and tumorigenesis.
Carroll PA, Diolaiti D, McFerrin L, Gu H, Djukovic D, Du J, Cheng PF, Anderson S, Ulrich M, Hurley JB, Raftery D, Ayer DE, Eisenman RN. Carroll PA, et al. Cancer Cell. 2015 Feb 9;27(2):271-85. doi: 10.1016/j.ccell.2014.11.024. Epub 2015 Jan 29. Cancer Cell. 2015. PMID: 25640402 Free PMC article. - T cell-specific deletion of Pgam1 reveals a critical role for glycolysis in T cell responses.
Toriyama K, Kuwahara M, Kondoh H, Mikawa T, Takemori N, Konishi A, Yorozuya T, Yamada T, Soga T, Shiraishi A, Yamashita M. Toriyama K, et al. Commun Biol. 2020 Jul 24;3(1):394. doi: 10.1038/s42003-020-01122-w. Commun Biol. 2020. PMID: 32709928 Free PMC article. - Regulatory T cell metabolism at the intersection between autoimmune diseases and cancer.
Kurniawan H, Soriano-Baguet L, Brenner D. Kurniawan H, et al. Eur J Immunol. 2020 Nov;50(11):1626-1642. doi: 10.1002/eji.201948470. Epub 2020 Oct 26. Eur J Immunol. 2020. PMID: 33067808 Free PMC article. Review.
References
- Anjum R, Blenis J. The RSK family of kinases: emerging roles in cellular signalling. Nat. Rev. Mol. Cell Biol. 2008;9:747–758. - PubMed
- Arsham AM, Neufeld TP. Thinking globally and acting locally with TOR. Curr. Opin. Cell Biol. 2006;18:589–597. - PubMed
- Blommaart EF, Luiken JJ, Blommaart PJ, van Woerkom GM, Meijer AJ. Phosphorylation of ribosomal protein S6 is inhibitory for autophagy in isolated rat hepatocytes. J. Biol. Chem. 1995;270:2320–2326. - PubMed
- Bodine SC, Stitt TN, Gonzalez M, Kline WO, Stover GL, Bauerlein R, Zlotchenko E, Scrimgeour A, Lawrence JC, Glass DJ, et al. Akt/mTOR pathway is a crucial regulator of skeletal muscle hypertrophy and can prevent muscle atrophy in vivo. Nat. Cell Biol. 2001;3:1014–1019. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous