Specificity of sensory-motor connections encoded by Sema3e-Plxnd1 recognition - PubMed (original) (raw)
Specificity of sensory-motor connections encoded by Sema3e-Plxnd1 recognition
Eline Pecho-Vrieseling et al. Nature. 2009.
Abstract
Spinal reflexes are mediated by synaptic connections between sensory afferents and motor neurons. The organization of these circuits shows several levels of specificity. Only certain classes of proprioceptive sensory neurons make direct, monosynaptic connections with motor neurons. Those that do are bound by rules of motor pool specificity: they form strong connections with motor neurons supplying the same muscle, but avoid motor pools supplying antagonistic muscles. This pattern of connectivity is initially accurate and is maintained in the absence of activity, implying that wiring specificity relies on the matching of recognition molecules on the surface of sensory and motor neurons. However, determinants of fine synaptic specificity here, as in most regions of the central nervous system, have yet to be defined. To address the origins of synaptic specificity in these reflex circuits we have used molecular genetic methods to manipulate recognition proteins expressed by subsets of sensory and motor neurons. We show here that a recognition system involving expression of the class 3 semaphorin Sema3e by selected motor neuron pools, and its high-affinity receptor plexin D1 (Plxnd1) by proprioceptive sensory neurons, is a critical determinant of synaptic specificity in sensory-motor circuits in mice. Changing the profile of Sema3e-Plxnd1 signalling in sensory or motor neurons results in functional and anatomical rewiring of monosynaptic connections, but does not alter motor pool specificity. Our findings indicate that patterns of monosynaptic connectivity in this prototypic central nervous system circuit are constructed through a recognition program based on repellent signalling.
Figures
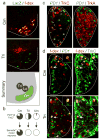
Figure 1. Sema3e marks Cm motor neurons and PlexinD1 proprioceptive neurons
(a) Sema3e directed LacZ in p0 Sema3enlz spinal cords after retrograde f-dextran tracing from Cm or Tri muscle nerves and summary of Cm, Ld, Tri motor pool position. (b) PlexinD1 expression in TrkCon proprioceptive afferents (top) and Sema3e expression in motor neurons (bottom) projecting to Cm, Tri or Uln muscles. (c) PlexinD1 expression in e16.5 c7 DRG, matched with TrkC and TrkA. (d) PlexinD1 (left) and TrkC (right) expression in e16.5 DRG neurons after retrograde f-dextran tracing from Cm or Tri muscle nerves.
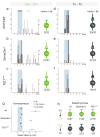
Figure 2. Loss of Sema3e or PlexinD1 function perturbs monosynaptic specificity
(a-f) Cm and Tri onset latency histograms upon homonymous muscle nerve stimulation (0-8ms in 0.15ms bins). Blue box: monosynaptic latency. Right of two lines: # neurons with onset latencies >8ms. Right of each histogram: Summary diagram # neurons with monosynaptic input/ #cells assayed. Sema3e or PlexinD1 mutation lead to monosynaptic Cm-Cm connections (light green), absent in wild-type (no presynaptic terminal). (g) Mean onset latencies (ms ± SEM) of homonymous pairs. Cm: data within (green) and outside (white) monosynaptic window. Significant differences: Cm; Tri in wild-type, Cm(no mono); Tri and Cm(no mono); Cm(mono) in Sema3e-/- and PlexinD1cond (p≤0.001; Student's t-test). (h) Summary of heteronymous recordings (Tri–Cm and Cm–Tri).
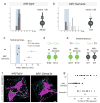
Figure 3. Increase in vGlut1 sensory contacts with Cm motor neurons in Sema3e mutant mice
(a) Digitalized vGlut1on proprioceptive terminals overlaid with ChATon motor neuron cell bodies in wild-type (c5 and c8), Sema3e-/- (c8) and PlexinD1cond (c8) mice. (b-d) vGlut1 apposition with f-dex labeled Tri (b) or Cm (c) motor neuron in wild-type (b, c) and Sema3e-/- (c) mice. Single confocal planes (b) or 3-dimensional view (c). White indicates contact site. (d) Frequency histograms of vGlut1 input to Cm motor neurons. (Bottom) average vGlut1 number in contact with Cm motor neurons (± SEM; p≤0.001; Student's t-test). Scale bar = (a): 30μm; (c):10μm.
Figure 4. Ectopic expression of Sema3e in motor neurons prevents monosynaptic connectivity
(a, b) Tri onset latency histograms upon Tri muscle nerve stimulation (0-8ms in 0.15ms bins). Blue box: monosynaptic latency. Right of each histogram: Summary diagram #neurons with monosynaptic input/ #cells assayed. Light grey color of presynaptic terminal (b): reduction in Tri motor neurons with monosynaptic Tri input. (c) Mean onset latencies (ms ± SEM) measured from homonymous pairs. Tri: data within (dark grey) and outside (light grey) monosynaptic window. Significant differences: Cm;Tri in wild-type mice (p≤0.001; Student's t-test) and Tri(mono);Tri(no mono) (p≤0.01; Student's t-test) and Tri(mono);Cm (p≤0.001; Student's t-test) in MN∷Sema3e mice. (d, e) Summary of Cm-Cm, Tri-Cm and Cm-Tri recordings. (f, g) Analysis of vGlut1 apposition with f-dex labeled Tri motor neurons (contact sites: white). (g) (top rows) values from individual motor neurons; (bottom) mean of analyzed cells (± SEM). Significant differences: wild-type; MN∷Sema3e and Sema3e-/-; MN∷Sema3e (p≤0.001; Student's t-test). Scale bar=10μm.
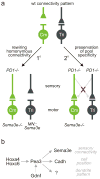
Figure 5. Regulation of synaptic specificity by repellant Sema3e-PlexinD1 signaling
(a) Connectivity patterns in Cm and Tri reflex arcs in wild-type mice (top) and with altered Sema3e-PlexinD1 signaling (bottom). Top: In wild-type, Tri but not Cm motor neurons receive direct homonymous proprioceptive inputs. Bottom left: changing the profile of Sema3e-PlexinD1 expression rewires homonymous connectivity. Loss of Sema3e-PlexinD1 signaling results in monosynaptic connections between Cm afferents and Cm motor neurons; Ectopic Sema3e expression reduces monosynaptic connections between Tri afferents and motor neurons. Bottom right: changing Sema3e-PlexinD1 signaling does not erode pool-specificity: no aberrant heteronymous sensory-motor connections are observed (red cross). (b) Genetic program for Sema3e expression in Cm motor neurons. Hox transcription factors program Cm motor pool identity, conferring Cm motor neuron sensitivity to the retrograde inductive influence of Gdnf. Pea3 expression by Cm motor neurons controls cell position, dendrite pattern and the selectivity of proprioceptive inputs through distinct downstream pathways, , .
Similar articles
- Specificity of monosynaptic sensory-motor connections imposed by repellent Sema3E-PlexinD1 signaling.
Fukuhara K, Imai F, Ladle DR, Katayama K, Leslie JR, Arber S, Jessell TM, Yoshida Y. Fukuhara K, et al. Cell Rep. 2013 Nov 14;5(3):748-58. doi: 10.1016/j.celrep.2013.10.005. Epub 2013 Nov 7. Cell Rep. 2013. PMID: 24210822 Free PMC article. - Semaphorin 3E-Plexin-D1 signaling controls pathway-specific synapse formation in the striatum.
Ding JB, Oh WJ, Sabatini BL, Gu C. Ding JB, et al. Nat Neurosci. 2011 Dec 18;15(2):215-23. doi: 10.1038/nn.3003. Nat Neurosci. 2011. PMID: 22179111 Free PMC article. - Semaphorin 3d and semaphorin 3e direct endothelial motility through distinct molecular signaling pathways.
Aghajanian H, Choi C, Ho VC, Gupta M, Singh MK, Epstein JA. Aghajanian H, et al. J Biol Chem. 2014 Jun 27;289(26):17971-9. doi: 10.1074/jbc.M113.544833. Epub 2014 May 13. J Biol Chem. 2014. PMID: 24825896 Free PMC article. - Molecular mechanisms underlying monosynaptic sensory-motor circuit development in the spinal cord.
Imai F, Yoshida Y. Imai F, et al. Dev Dyn. 2018 Apr;247(4):581-587. doi: 10.1002/dvdy.24611. Epub 2018 Jan 17. Dev Dyn. 2018. PMID: 29226492 Free PMC article. Review. - Transcriptional networks in the early development of sensory-motor circuits.
Dasen JS. Dasen JS. Curr Top Dev Biol. 2009;87:119-48. doi: 10.1016/S0070-2153(09)01204-6. Curr Top Dev Biol. 2009. PMID: 19427518 Review.
Cited by
- Activity Regulates the Incidence of Heteronymous Sensory-Motor Connections.
Mendelsohn AI, Simon CM, Abbott LF, Mentis GZ, Jessell TM. Mendelsohn AI, et al. Neuron. 2015 Jul 1;87(1):111-23. doi: 10.1016/j.neuron.2015.05.045. Epub 2015 Jun 18. Neuron. 2015. PMID: 26094608 Free PMC article. - Statistical connectivity provides a sufficient foundation for specific functional connectivity in neocortical neural microcircuits.
Hill SL, Wang Y, Riachi I, Schürmann F, Markram H. Hill SL, et al. Proc Natl Acad Sci U S A. 2012 Oct 16;109(42):E2885-94. doi: 10.1073/pnas.1202128109. Epub 2012 Sep 18. Proc Natl Acad Sci U S A. 2012. PMID: 22991468 Free PMC article. - Positive regulation of neocortical synapse formation by the Plexin-D1 receptor.
Wang F, Eagleson KL, Levitt P. Wang F, et al. Brain Res. 2015 Aug 7;1616:157-165. doi: 10.1016/j.brainres.2015.05.005. Epub 2015 May 11. Brain Res. 2015. PMID: 25976775 Free PMC article. - Sensory and descending motor circuitry during development and injury.
Plant GW, Weinrich JA, Kaltschmidt JA. Plant GW, et al. Curr Opin Neurobiol. 2018 Dec;53:156-161. doi: 10.1016/j.conb.2018.08.008. Epub 2018 Sep 8. Curr Opin Neurobiol. 2018. PMID: 30205323 Free PMC article. Review. - Midline signalling systems direct the formation of a neural map by dendritic targeting in the Drosophila motor system.
Mauss A, Tripodi M, Evers JF, Landgraf M. Mauss A, et al. PLoS Biol. 2009 Sep;7(9):e1000200. doi: 10.1371/journal.pbio.1000200. Epub 2009 Sep 22. PLoS Biol. 2009. PMID: 19771146 Free PMC article.
References
- Rossignol S, Dubuc R, Gossard JP. Dynamic sensorimotor interactions in locomotion. Physiological reviews. 2006;86:89–154. - PubMed
- Windhorst U. Muscle proprioceptive feedback and spinal networks. Brain research bulletin. 2007;73:155–202. - PubMed
- Brown A. Organization in the spinal cord. Springer; New York: 1981.
- Burke RE, Glenn LL. Horseradish peroxidase study of the spatial and electrotonic distribution of group Ia synapses on type-identified ankle extensor motoneurons in the cat. The Journal of comparative neurology. 1996;372:465–485. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases