Bam and Bgcn antagonize Nanos-dependent germ-line stem cell maintenance - PubMed (original) (raw)
Bam and Bgcn antagonize Nanos-dependent germ-line stem cell maintenance
Yun Li et al. Proc Natl Acad Sci U S A. 2009.
Abstract
The balance between germ-line stem cell (GSC) self-renewal and differentiation in Drosophila ovaries is mediated by the antagonistic relationship between the Nanos (Nos)-Pumilio translational repressor complex, which promotes GSC self-renewal, and expression of Bam, a key differentiation factor. Here, we find that Bam and Nos proteins are expressed in reciprocal patterns in young germ cells. Repression of Nos in Bam-expressing cells depends on sequences in the nos 3'-UTR, suggesting that Nos is regulated by translational repression. Ectopic Bam causes differentiation of GSCs, and this activity depends on the endogenous nos 3'-UTR sequence. Previous evidence showed that Bgcn is an obligate factor for the ability of Bam to drive differentiation, and we now report that Bam forms a complex with Bgcn, a protein related to the RNA-interacting DExH-box polypeptides. Together, these observations suggest that Bam-Bgcn act together to antagonize Nos expression; thus, derepressing cystoblast-promoting factors. These findings emphasize the importance of translational repression in balancing stem cell self-renewal and differentiation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
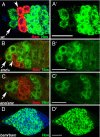
Fig. 1.
Bam and Nos are expressed in reciprocal patterns. (A) Bam protein (red, detected by anti-GFP against a Bam:GFP rescuing transgene) and Nos protein (green, detected by anti-myc against a Nos:Myc rescuing transgene) are expressed in nonoverlapping domains in early germ cells. Stem stems (arrows), CBs (arrowheads). (B and C) Germ cells from encore mutant flies (C) display an expansion of Bam expression compared with enc/+ and (B) a corresponding delay in late Nos accumulation (red, anti-Bam; green, Nos:GFP, detected by anti-GFP against a rescuing transgene). Stem cells (arrows), CBs (arrowheads). Cystoblasts are out of plane of focus in C. (D) Nos (green) remains high in germ cells lacking Bam. (A′–D′) Nos channel alone. (Magnification bars, 25 μm.)
Fig. 2.
Genetic interactions reveal an antagonistic relationship between bam and nos. (A) Ovaries from _nos_[RC] mutants are rudimentary (arrow) with few maturing egg chambers (arrowhead). (B) Introduction of a single loss of function bam allele suppressed the _nos_[RC] phenotype and ovaries contained many more maturing egg chambers (arrows). (C) Suppression was quantified by counting the number of egg chambers per terminal filament in ovaries from _nos_[RC] and _nos_[RD] ± bam or bgcn. The difference between _nos_[RC]/Df and _nos_[RD]/Df may reflect suppressing background mutation(s) on the _nos_[RD] chromosome, because the _bam_[BG] _nos_[RD] recombinant chromosome produced a more severe phenotype over Df than the original _nos_[RD] chromosome.
Fig. 3.
Nos accumulation depends on the 3′-UTR. (A) Nos promoter driving GFP expression accumulates GFP throughout region 1 of the germarium. Stem cells (arrows), CBs (arrowheads). (B) Nos protein (green, anti-GFP against a rescuing _nos_-GFP transgene) accumulates in stem cells (arrow) and precystoblasts, is lost in Bam+ cells (red, anti-Bam), and reaccumulates in 16-cell cysts where Bam expression is no longer detected. (C) Nos expressed from the nos promoter but with the heterologous tubulin 3′-UTR accumulates uniformly in region 1 of the germarium, including in Bam+ cells (arrowhead). (A′–C′) Nos channel alone.
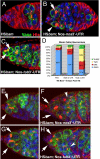
Fig. 4.
Bam-dependent GSC loss acts via the nos 3′-UTR. (A–C) Green, Vasa; Red, Hts; and Blue, DNA. (A) The p[HS-_bam_] ovaries maintained at 37 °C for 1 h were examined 3 days post heat shock; 86% of these germaria were devoid of stem cells. (B) Germaria from animals carrying a wild-type nos transgene were similarly depleted of stem cells by HS-bam treatment (71% of germaria had no stem cells). Arrow points to a 4-cell cyst and arrowhead indicates a CB or 2-cell cyst in the GSC position. (C) Germaria from animals carrying a nos transgene with a tubulin 3′-UTR were protected from HS-bam induced stem cell depletion. Arrows indicate 2 GSCs (81% of germaria contained 1 or more stem cells). (D) Quantification of GSC retention or loss after ectopic Bam expression. (E–H) Green, Nos:GFP; Red, Hts; and Blue, DNA. Arrows point to GSCs. Detectible Nos declines in stem cells after HS-bam, except when Nos is expressed with the tubulin 3′-UTR. (E) Nos:GFP expression levels in germarium of p[_nos_P-Nos:GFP-_nos_3′-UTR] animal without heat shock. (F) Nos:GFP levels in p[_nos_P-Nos:GFP-_nos_3′-UTR] animal 9.5 h after heat shock. Arrows indicate GSCs; arrowheads indicate 16-cell cysts. (G) Nos:GFP expression levels in germarium of p[_nos_P-Nos:GFP-_tub_3′-UTR] animal without heat shock. (H) Nos:GFP expression levels in germarium of p[_nos_P-Nos:GFP-_tub_3′-UTR] animal 9.5 h after heat shock. Arrows indicate GSCs; arrowheads indicate 16-cell cysts.
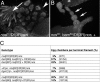
Fig. 5.
Bam and Bgcn form a complex. (A) Immunoblot probed with anti-V5 antibody after “pulldown” experiments with epitope-tagged Bam:GST and Bgcn:V5 proteins. (lane 1) Yeast extract expressing Bgcn:V5. (lanes 2–4) Samples from pellets recovered by incubation with glutathione-coupled agarose beads (GST pulldowns). Bgcn:V5 was recovered in the pellet only when the extracts from Bgcn:V5 yeast were incubated with Bam:GST bacterial extracts. (B) Bam and Bgcn coimmunoprecipitate from ovaries. Ovarian extracts from flies expressing Bgcn:GFP and Bam:HA (lane 2) or _w_1118 flies (lane 3) were incubated with anti-GFP or anti- HA antibodies for immunoprecipitation and the immunoblots were probed with anti-GFP and anti-HA antibodies separately. Lane 1, 10% of extract; lane 2, flies _w_−;[w+,_bam_P-Bam:HA];[w+,_bgcn_P-Bgcn:GFP] anti-GFP IP; lane 3, flies _w_1118 anti-GFP IP; lane 4, flies _w_−;[w+,_bam_P-Bam:HA];[w+,_bgcn_P-Bgcn:GFP] anti-HA IP; lane 5, flies _w_1118 anti-HA IP. (C) Bam and Bgcn coimmunoprecipitate from S2 cells that expressed tagged proteins. Tagged proteins (Bam:HA; Bgcn:Myc) were over expressed in S2 cells and the cell lysate were used for anti-HA or anti-Myc immunoprecipitation. Lane 1, 10% of S2 cell lysate; lane 2, S2 cell lysate expressing Bam:HA and Bgcn:Myc immunoprecipitated with anti-HA antibody; lane 3, S2 cell lysate expressing Bgcn:Myc immunoprecipitated with anti-HA antibody; lane 4, S2 cell lysate expressing Bam:HA and Bgcn:Myc immunoprecipitated with anti-Myc antibody; lane 5, S2 cell lysate expressing Bam:HA immunoprecipitated with anti-Myc antibody.
Similar articles
- Direct inhibition of Pumilo activity by Bam and Bgcn in Drosophila germ line stem cell differentiation.
Kim JY, Lee YC, Kim C. Kim JY, et al. J Biol Chem. 2010 Feb 12;285(7):4741-6. doi: 10.1074/jbc.M109.002014. Epub 2009 Dec 14. J Biol Chem. 2010. PMID: 20018853 Free PMC article. - Bam and Bgcn in Drosophila germline stem cell differentiation.
Perinthottathil S, Kim C. Perinthottathil S, et al. Vitam Horm. 2011;87:399-416. doi: 10.1016/B978-0-12-386015-6.00038-X. Vitam Horm. 2011. PMID: 22127253 Review. - Histone H3K9 trimethylase Eggless controls germline stem cell maintenance and differentiation.
Wang X, Pan L, Wang S, Zhou J, McDowell W, Park J, Haug J, Staehling K, Tang H, Xie T. Wang X, et al. PLoS Genet. 2011 Dec;7(12):e1002426. doi: 10.1371/journal.pgen.1002426. Epub 2011 Dec 22. PLoS Genet. 2011. PMID: 22216012 Free PMC article. - Mechanisms ensuring robust repression of the Drosophila female germline stem cell maintenance factor Nanos via posttranscriptional regulation.
Malik S, Jang W, Kim JY, Kim C. Malik S, et al. FASEB J. 2020 Sep;34(9):11421-11430. doi: 10.1096/fj.202000656R. Epub 2020 Jul 12. FASEB J. 2020. PMID: 32654316 - Germline stem cells.
Spradling A, Fuller MT, Braun RE, Yoshida S. Spradling A, et al. Cold Spring Harb Perspect Biol. 2011 Nov 1;3(11):a002642. doi: 10.1101/cshperspect.a002642. Cold Spring Harb Perspect Biol. 2011. PMID: 21791699 Free PMC article. Review.
Cited by
- Diffusible fraction of niche BMP ligand safeguards stem-cell differentiation.
Ridwan SM, Twillie A, Poursaeid S, Beard EK, Bener MB, Antel M, Cowan AE, Matsuda S, Inaba M. Ridwan SM, et al. Nat Commun. 2024 Feb 7;15(1):1166. doi: 10.1038/s41467-024-45408-7. Nat Commun. 2024. PMID: 38326318 Free PMC article. - A nuclear architecture screen in Drosophila identifies Stonewall as a link between chromatin position at the nuclear periphery and germline stem cell fate.
Chavan A, Isenhart R, Nguyen SC, Kotb N, Harke J, Sintsova A, Ulukaya G, Uliana F, Ashiono C, Kutay U, Pegoraro G, Rangan P, Joyce EF, Jagannathan M. Chavan A, et al. bioRxiv [Preprint]. 2023 Nov 17:2023.11.17.567611. doi: 10.1101/2023.11.17.567611. bioRxiv. 2023. PMID: 38014085 Free PMC article. Updated. Preprint. - Direct inhibition of Pumilo activity by Bam and Bgcn in Drosophila germ line stem cell differentiation.
Kim JY, Lee YC, Kim C. Kim JY, et al. J Biol Chem. 2010 Feb 12;285(7):4741-6. doi: 10.1074/jbc.M109.002014. Epub 2009 Dec 14. J Biol Chem. 2010. PMID: 20018853 Free PMC article. - Drosophila ataxin 2-binding protein 1 marks an intermediate step in the molecular differentiation of female germline cysts.
Tastan OY, Maines JZ, Li Y, McKearin DM, Buszczak M. Tastan OY, et al. Development. 2010 Oct;137(19):3167-76. doi: 10.1242/dev.050575. Epub 2010 Aug 19. Development. 2010. PMID: 20724451 Free PMC article. - Germ line differentiation factor Bag of Marbles is a regulator of hematopoietic progenitor maintenance during Drosophila hematopoiesis.
Tokusumi T, Tokusumi Y, Hopkins DW, Shoue DA, Corona L, Schulz RA. Tokusumi T, et al. Development. 2011 Sep;138(18):3879-84. doi: 10.1242/dev.069336. Epub 2011 Aug 3. Development. 2011. PMID: 21813570 Free PMC article.
References
- Xie T, Spradling AC. decapentaplegic is essential for the maintenance and division of germline stem cells in the Drosophila ovary. Cell. 1998;94:251–260. - PubMed
- Chen D, McKearin D. Dpp signaling silences bam transcription directly to establish asymmetric divisions of germline stem cells. Curr Biol. 2003;13:1786–1791. - PubMed
- Casanueva MO, Ferguson EL. Germline stem cell number in the Drosophila ovary is regulated by redundant mechanisms that control Dpp signaling. Development. 2004;131:1881–1890. - PubMed
- Song X, et al. Bmp signals from niche cells directly repress transcription of a differentiation-promoting gene, bag of marbles, in germline stem cells in the Drosophila ovary. Development. 2004;131:1353–1364. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous