Pre-symptomatic detection of chronic motor deficits and genotype prediction in congenic B6.SOD1(G93A) ALS mouse model - PubMed (original) (raw)
Pre-symptomatic detection of chronic motor deficits and genotype prediction in congenic B6.SOD1(G93A) ALS mouse model
C R Hayworth et al. Neuroscience. 2009.
Abstract
Amyotrophic lateral sclerosis (ALS) is an incurable progressive paralytic motor neuron disease with limited therapeutic options. Since their creation by Gurney et al. (1994) [Science 264:1772-1775], transgenic superoxide dismutase-1 with glycine to alanine switch at codon 93 (SOD1(G93A)) mice have become the benchmark pre-clinical model for screening ALS therapies. Surprisingly, despite physiological, anatomical, ultrastructural and biochemical evidence of early motor system dysfunction, it has proven difficult to detect motor performance deficits in pre-symptomatic SOD1(G93A) mice. As an alternative to conventional forced motor tests, we investigated the progression of motor performance deficits in freely behaving pre-symptomatic congenic B6.SOD1(G93A) mice. We found that motor performance deficits began several weeks prior to the onset of overt clinical symptoms (postnatal day 45). More importantly, once motor performance deficits manifested, they persisted in parallel with disease progression. In addition, two physical measures of muscle girth revealed progressive hindlimb muscle atrophy that predicted genotype in individual pre-symptomatic mice with 80% accuracy. Together, these data suggest that muscle girth is a reliable and indirect measure of hindlimb muscle denervation and an early, objective marker for disease onset in congenic B6.SOD1(G93A) ALS mice. Moreover, we present regression equations based on hindlimb muscle girth for predicting genotype in future studies using B6.SOD1(G93A) mice. These findings support new objective criteria for clinical disease onset and provide objective measures that require little expertise. These studies demonstrate a cost-effective approach for more thorough evaluation of neuroprotective strategies that seek to disrupt disease mechanisms early in the disease process. To our knowledge, these findings are the first to report early chronic motor performance and physical deficits that are coincident with the earliest known motor dysfunction in any ALS mouse model.
Figures
Figure 1. Delayed clinical symptom onset in female B6.SOD1G93A mice
Kaplan-Meier curves to compare tremor onset and survival time (moribund animals unable to right themselves within 20 s) between male and female B6.SOD1G93A mice. Clinical symptom onset (tremor in at least 1 hindlimb upon tail suspension) was delayed ~10 days in females (n=39) compared to male mice (n=49). No significant difference in survival time between male and female B6.SOD1G93A mice was observed.
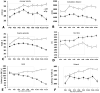
Figure 2. Progression of motor deficits in B6.SOD1G93A mice
Line graphs showing time course of disease progression from motor tests. Animals were monitored every 10 d from P45-P125 except for rotarod (P55-P125). For simplicity, all motor performance graphs for wildtype (WT, open circles) and B6.SOD1G93A (Tg, black squares) were collapsed across sex. (A-D) Open field measures. (E) Paw grip endurance (PaGE) test and (F) Rotarod. The dashed box indicates the time points for post hoc statistical analysis of male and female mice (see Table 2).
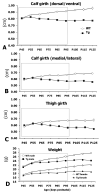
Figure 3. Progression of muscle size and body weight deficits in B6.SOD1G93A mice
Line graphs showing time course of disease progression from physical measures. Animals were monitored every 10 d from P45-P125. For clarity, all muscle girth graphs for each group (wildtype and B6.SOD1G93A) were collapsed across sex (A-C). The dashed box indicates the time points for post hoc statistical analysis of male and female mice (shaded region in Table 2).
Figure 4. Muscle girth predicts genotype in pre-symptomatic SOD1 G93A mice
Line graph showing at each time point the percentage of animals correctly classified as wildtype or transgenic based on calf (d/v) and thigh muscle girth. Notice that 27out of 35 females can be correctly g by P55 whereas 43 out of 54 males can be correctly classified at P65.
Figure 5. Phenotypic diagnostic accuracy in muscle girth but not motor performance measures
Receiver-operated-characteristics (ROC) curves to determine test accuracy were generated for each time point from P45 to P125 for all physical measures (A and B) and motor performance tests (C and D). Notice how rapidly the phenotypic diagnostic accuracy for calf (d/v) muscle girth measures reaches 80% in pre-symptomatic males and females (A and B) and is quickly matched by calf (m/l) and thigh at P65. While motor tasks are not as diagnostically accurate as muscle girth measures (C and D), in general, they are more accurate for males versus female mice. Shaded region indicates high diagnostic accuracy.
Similar articles
- Comparative Study of Structural and Functional Rearrangements in Skeletal Muscle Mitochondria of SOD1-G93A Transgenic Mice at Pre-, Early-, and Late-Symptomatic Stages of ALS Progression.
Belosludtseva NV, Ilzorkina AI, Dubinin MV, Mikheeva IB, Belosludtsev KN. Belosludtseva NV, et al. Front Biosci (Landmark Ed). 2025 Mar 18;30(3):28260. doi: 10.31083/FBL28260. Front Biosci (Landmark Ed). 2025. PMID: 40152389 - Progressive impairment of CaV1.1 function in the skeletal muscle of mice expressing a mutant type 1 Cu/Zn superoxide dismutase (G93A) linked to amyotrophic lateral sclerosis.
Beqollari D, Romberg CF, Dobrowolny G, Martini M, Voss AA, Musarò A, Bannister RA. Beqollari D, et al. Skelet Muscle. 2016 Jun 23;6:24. doi: 10.1186/s13395-016-0094-6. eCollection 2016. Skelet Muscle. 2016. PMID: 27340545 Free PMC article. - The effect of peripheral nerve injury on disease progression in the SOD1(G93A) mouse model of amyotrophic lateral sclerosis.
Sharp PS, Dick JR, Greensmith L. Sharp PS, et al. Neuroscience. 2005;130(4):897-910. doi: 10.1016/j.neuroscience.2004.09.069. Neuroscience. 2005. PMID: 15652988 - Unraveling the complexity of amyotrophic lateral sclerosis: recent advances from the transgenic mutant SOD1 mice.
Peviani M, Caron I, Pizzasegola C, Gensano F, Tortarolo M, Bendotti C. Peviani M, et al. CNS Neurol Disord Drug Targets. 2010 Aug;9(4):491-503. doi: 10.2174/187152710791556096. CNS Neurol Disord Drug Targets. 2010. PMID: 20522008 Review. - ALS genetic modifiers that increase survival of SOD1 mice and are suitable for therapeutic development.
Riboldi G, Nizzardo M, Simone C, Falcone M, Bresolin N, Comi GP, Corti S. Riboldi G, et al. Prog Neurobiol. 2011 Oct;95(2):133-48. doi: 10.1016/j.pneurobio.2011.07.009. Epub 2011 Jul 26. Prog Neurobiol. 2011. PMID: 21816207 Review.
Cited by
- Administration of Recombinant Heat Shock Protein 70 Delays Peripheral Muscle Denervation in the SOD1(G93A) Mouse Model of Amyotrophic Lateral Sclerosis.
Gifondorwa DJ, Jimenz-Moreno R, Hayes CD, Rouhani H, Robinson MB, Strupe JL, Caress J, Milligan C. Gifondorwa DJ, et al. Neurol Res Int. 2012;2012:170426. doi: 10.1155/2012/170426. Epub 2012 Aug 1. Neurol Res Int. 2012. PMID: 22900172 Free PMC article. - MicroNeurotrophins Improve Survival in Motor Neuron-Astrocyte Co-Cultures but Do Not Improve Disease Phenotypes in a Mutant SOD1 Mouse Model of Amyotrophic Lateral Sclerosis.
Glajch KE, Ferraiuolo L, Mueller KA, Stopford MJ, Prabhkar V, Gravanis A, Shaw PJ, Sadri-Vakili G. Glajch KE, et al. PLoS One. 2016 Oct 7;11(10):e0164103. doi: 10.1371/journal.pone.0164103. eCollection 2016. PLoS One. 2016. PMID: 27716798 Free PMC article. - Transcriptomic analysis of human ALS skeletal muscle reveals a disease-specific pattern of dysregulated circRNAs.
Tsitsipatis D, Mazan-Mamczarz K, Si Y, Herman AB, Yang JH, Guha A, Piao Y, Fan J, Martindale JL, Munk R, Yang X, De S, Singh BK, Ho R, Gorospe M, King PH. Tsitsipatis D, et al. Aging (Albany NY). 2022 Dec 30;14(24):9832-9859. doi: 10.18632/aging.204450. Epub 2022 Dec 30. Aging (Albany NY). 2022. PMID: 36585921 Free PMC article. - Carboxyl-terminal modulator protein regulates Akt signaling during skeletal muscle atrophy in vitro and a mouse model of amyotrophic lateral sclerosis.
Wang J, Fry CME, Walker CL. Wang J, et al. Sci Rep. 2019 Mar 8;9(1):3920. doi: 10.1038/s41598-019-40553-2. Sci Rep. 2019. PMID: 30850672 Free PMC article. - Microendoscopy detects altered muscular contractile dynamics in a mouse model of amyotrophic lateral sclerosis.
Chen X, Sanchez GN, Schnitzer MJ, Delp SL. Chen X, et al. Sci Rep. 2020 Jan 16;10(1):457. doi: 10.1038/s41598-019-56555-z. Sci Rep. 2020. PMID: 31949214 Free PMC article.
References
- Amendola J, Verrier B, Roubertoux P, Durand J. Altered sensorimotor development in a transgenic mouse model of amyotrophic lateral sclerosis. Eur J Neurosci. 2004;20:2822–2826. - PubMed
- Azari MF, Lopes EC, Stubna C, Turner BJ, Zang D, Nicola NA, Kurek JB, Cheema SS. Behavioural and anatomical effects of systemically administered leukemia inhibitory factor in the SOD1(G93A G1H) mouse model of familial amyotrophic lateral sclerosis. Brain Res. 2003;982:92–97. - PubMed
- Azzouz M, Krezel W, Dolle P, Vodouhe C, Warter JM, Poindron P, Borg J. Compensatory mechanism of motor defect in SOD1 transgenic mice by overactivation of striatal cholinergic neurons. Neuroreport. 1999;10:1013–1018. - PubMed
- Azzouz M, Ralph GS, Storkebaum E, Walmsley LE, Mitrophanous KA, Kingsman SM, Carmeliet P, Mazarakis ND. VEGF delivery with retrogradely transported lentivector prolongs survival in a mouse ALS model. Nature. 2004;429:413–417. - PubMed
- Boillee S, Yamanaka K, Lobsiger CS, Copeland NG, Jenkins NA, Kassiotis G, Kollias G, Cleveland DW. Onset and progression in inherited ALS determined by motor neurons and microglia. Science. 2006;312:1389–1392. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous