Macrophage heterogeneity in atherosclerotic plaques - PubMed (original) (raw)
Review
Macrophage heterogeneity in atherosclerotic plaques
Jason L Johnson et al. Curr Opin Lipidol. 2009 Oct.
Abstract
Purpose of review: The varied behaviour of macrophages and foam cells during atherosclerosis and its clinical sequelae prompt the question whether all these activities can be the property of a single cell population.
Recent findings: Subsets of monocytes with distinct patterns of surface markers and behaviours during inflammation have recently been characterized and shown to have complementary roles during progression of atherosclerosis. A variety of macrophage phenotypes derived from these monocyte subsets in response to mediators of innate and acquired immunity have also been found in plaques. Based on functional properties and genomic signatures, they may have different impacts on facets of plaque development, including fibrous cap and lipid core formation.
Summary: Monocyte and macrophage phenotypic diversity is important in atherogenesis. More work is needed to define consistent marker sets for the different foam cell phenotypes in experimental animals and humans. Cell tracking studies are needed to establish their relationship with monocyte subtypes. In addition, genetic and pharmacological manipulation of phenotypes will be useful to define their functions and exploit the resulting therapeutic potential.
Figures
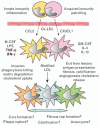
Figure 1. Origins of macrophage diversity
Recruitment of two different populations of monocytes and action of different mediators may lead to distinct macrophage phenotypes. Uptake of modified lipoproteins converts these to foam cells that may exert a variety of functions in plaque development. GM-CSF, granulocyte macrophage-colony stimulating factor; LPS, lipopolysaccharide; M-CSF, macrophage-colony stimulating factor.
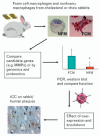
Figure 2. A platform for studying function of foamy and non-foamy macrophages generated in vivo
Generation of nonfoamy and foam cell macrophages in subcutaneous sponges in rabbits yields pur preparations that can be subjected to analysis or gene transfer to determine functions that can be verified by immunocytochemistry in tissue sections. FCM, foam cell macrophage; ICC, immunocytochemistry; MMP, metalloproteinase; NFM, nonfoamy macrophage.
Similar articles
- Role of colony-stimulating factors in atherosclerosis.
Di Gregoli K, Johnson JL. Di Gregoli K, et al. Curr Opin Lipidol. 2012 Oct;23(5):412-21. doi: 10.1097/MOL.0b013e328357ca6e. Curr Opin Lipidol. 2012. PMID: 22964991 Review. - Vascular Macrophages in Atherosclerosis.
Xu H, Jiang J, Chen W, Li W, Chen Z. Xu H, et al. J Immunol Res. 2019 Dec 1;2019:4354786. doi: 10.1155/2019/4354786. eCollection 2019. J Immunol Res. 2019. PMID: 31886303 Free PMC article. Review. - Macrophage heterogeneity in atherosclerosis: A matter of context.
Wieland EB, Kempen LJ, Donners MM, Biessen EA, Goossens P. Wieland EB, et al. Eur J Immunol. 2024 Jan;54(1):e2350464. doi: 10.1002/eji.202350464. Epub 2023 Nov 9. Eur J Immunol. 2024. PMID: 37943053 Review. - Curcumin as a potential modulator of M1 and M2 macrophages: new insights in atherosclerosis therapy.
Momtazi-Borojeni AA, Abdollahi E, Nikfar B, Chaichian S, Ekhlasi-Hundrieser M. Momtazi-Borojeni AA, et al. Heart Fail Rev. 2019 May;24(3):399-409. doi: 10.1007/s10741-018-09764-z. Heart Fail Rev. 2019. PMID: 30673930 Review. - Insulin-Like Growth Factor-1 Receptor Deficiency in Macrophages Accelerates Atherosclerosis and Induces an Unstable Plaque Phenotype in Apolipoprotein E-Deficient Mice.
Higashi Y, Sukhanov S, Shai SY, Danchuk S, Tang R, Snarski P, Li Z, Lobelle-Rich P, Wang M, Wang D, Yu H, Korthuis R, Delafontaine P. Higashi Y, et al. Circulation. 2016 Jun 7;133(23):2263-78. doi: 10.1161/CIRCULATIONAHA.116.021805. Epub 2016 May 6. Circulation. 2016. PMID: 27154724 Free PMC article.
Cited by
- Amaranth Oil Increases Total and LDL Cholesterol Levels without Influencing Early Markers of Atherosclerosis in an Overweight and Obese Population: A Randomized Double-Blind Cross-Over Study in Comparison with Rapeseed Oil Supplementation.
Dus-Zuchowska M, Walkowiak J, Morawska A, Krzyzanowska-Jankowska P, Miskiewicz-Chotnicka A, Przyslawski J, Lisowska A. Dus-Zuchowska M, et al. Nutrients. 2019 Dec 16;11(12):3069. doi: 10.3390/nu11123069. Nutrients. 2019. PMID: 31888234 Free PMC article. Clinical Trial. - Tissue-engineered collagenous fibrous cap models to systematically elucidate atherosclerotic plaque rupture.
Wissing TB, Van der Heiden K, Serra SM, Smits AIPM, Bouten CVC, Gijsen FJH. Wissing TB, et al. Sci Rep. 2022 Mar 31;12(1):5434. doi: 10.1038/s41598-022-08425-4. Sci Rep. 2022. PMID: 35361847 Free PMC article. - High-density lipoprotein heterogeneity and function in reverse cholesterol transport.
Rothblat GH, Phillips MC. Rothblat GH, et al. Curr Opin Lipidol. 2010 Jun;21(3):229-38. doi: 10.1097/mol.0b013e328338472d. Curr Opin Lipidol. 2010. PMID: 20480549 Free PMC article. Review. - The LDL-HDL profile determines the risk of atherosclerosis: a mathematical model.
Hao W, Friedman A. Hao W, et al. PLoS One. 2014 Mar 12;9(3):e90497. doi: 10.1371/journal.pone.0090497. eCollection 2014. PLoS One. 2014. PMID: 24621857 Free PMC article. - Induction of the nuclear receptor PPAR-γ by the cytokine GM-CSF is critical for the differentiation of fetal monocytes into alveolar macrophages.
Schneider C, Nobs SP, Kurrer M, Rehrauer H, Thiele C, Kopf M. Schneider C, et al. Nat Immunol. 2014 Nov;15(11):1026-37. doi: 10.1038/ni.3005. Epub 2014 Sep 28. Nat Immunol. 2014. PMID: 25263125
References
- Hansson GK. Mechanisms of disease – inflammation, atherosclerosis, and coronary artery disease. New Engl J Med. 2005;352:1685–1695. - PubMed
- Nakashima Y, Fujii H, Sumiyoshi S, et al. Early human atherosclerosis: accumulation of lipid and proteoglycans in intimal thickenings followed by macrophage infiltration. Arterioscler Thromb Vasc Biol. 2007;27:1159–1165. - PubMed
- Aikawa M, Rabkin E, Voglic SJ, et al. Lipid lowering promotes accumulation of mature smooth muscle cells expressing smooth muscle myosin heavy chain isoforms in rabbit atheroma. Circ Res. 1998;83:1015–1026. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous