A prospective study of protein excretion using short-interval timed urine collections in patients with lupus nephritis - PubMed (original) (raw)
Comparative Study
. 2009 Dec;76(12):1284-8.
doi: 10.1038/ki.2009.344. Epub 2009 Sep 16.
Affiliations
- PMID: 19759526
- PMCID: PMC3093656
- DOI: 10.1038/ki.2009.344
Comparative Study
A prospective study of protein excretion using short-interval timed urine collections in patients with lupus nephritis
Derek M Fine et al. Kidney Int. 2009 Dec.
Abstract
The 24-h urine protein-to-creatinine ratio is the gold standard in evaluating proteinuria in lupus nephritis; however, the urine collection is inconvenient to the patient. Random spot urine protein-to-creatinine ratios, although convenient, have poor agreement with the 24-h ratios in these patients. Here, we sought to define a timed collection interval providing accurate and precise data and patient convenience. Urine from 41 patients, in 2 medical centers, with biopsy-proven lupus nephritis was collected at 6-h intervals for 24 h. The protein-to-creatinine ratio of each short collection was then compared with that of a 24-h collection made by combining the 6-h samples. A first morning void and spot urine samples were collected before and after the 24-h collection, respectively. There was significant diurnal variation with peak proteinuria at 6-12 h and nadir at 18-24 h. Each 6-h collection showed excellent correlation and concordance with the 24-h protein-to-creatinine ratio, but the 12-24-h interval had the best agreement. In contrast to the random spot urines, the first morning void also had excellent correlation and concordance, but underestimated the 24-h protein-to-creatinine ratio. Our study shows that a 12-h overnight urine collection is the best surrogate, with excellent agreement with the 24-h protein-to-creatinine ratio, and it is convenient for patients. There was little variability between centers, an important feature for clinical trials.
Conflict of interest statement
DISCLOSURE
All the authors declared no conflict of interests.
Figures
Figure 1. Collection timeline for interval collections
Spot 0 (SP0) was first morning urine and spot 2 (SP24) was obtained after the 24-h urine collection was completed. Note that for Hopkins lupus cohort (HLC), SP24 was obtained as the second morning void and for Ohio SLE Study (OSS), as a random spot collection. SLE, systemic lupus erythematosus.
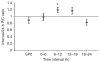
Figure 2. Diurnal variation of protein excretion
Interval protein/creatinine (P/C) ratios were divided by their corresponding 24-h P/C ratios for the indicated intervals. SP0 represents the first morning void. The middle bar represents the mean ratio, and the top and bottom bars represent ±2 standard errors of the mean. *P<0.001 versus 18–24-h interval.
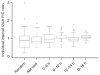
Figure 3. Variability of interval protein/creatinine (P/C) ratio declines as the duration of the interval approaches 24 h
Box-and-whisker plots of individual interval P/C ratios divided by their corresponding 24-h P/C ratio. Boxes represent the interquartile range. Whisker lengths represent up to 1.5 times the interquartile range. Outliers beyond the whiskers are shown with open circles.
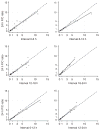
Figure 4. Correlation of 6- and 12-h protein/creatinine (P/C) ratio with 24-h P/C ratio
The dashed line represents the line of perfect concordance (45° line); the solid line is the best-fit regression.
Similar articles
- Urine protein-to-creatinine ratio is a reliable measure of proteinuria in lupus nephritis.
Christopher-Stine L, Petri M, Astor BC, Fine D. Christopher-Stine L, et al. J Rheumatol. 2004 Aug;31(8):1557-9. J Rheumatol. 2004. PMID: 15290735 - Random spot urine protein to creatinine ratio is a reliable measure of proteinuria in lupus nephritis in Koreans.
Choi IA, Park JK, Lee EY, Song YW, Lee EB. Choi IA, et al. Clin Exp Rheumatol. 2013 Jul-Aug;31(4):584-8. Epub 2013 May 27. Clin Exp Rheumatol. 2013. PMID: 23711154 - Spot urine protein/creatinine ratios are unreliable estimates of 24 h proteinuria in most systemic lupus erythematosus nephritis flares.
Birmingham DJ, Rovin BH, Shidham G, Nagaraja HN, Zou X, Bissell M, Yu CY, Hebert LA. Birmingham DJ, et al. Kidney Int. 2007 Oct;72(7):865-70. doi: 10.1038/sj.ki.5002421. Epub 2007 Jul 25. Kidney Int. 2007. PMID: 17653137 - Commentary on the Current Guidelines for the Diagnosis of Lupus Nephritis Flare.
Ayoub I, Birmingham D, Rovin B, Hebert L. Ayoub I, et al. Curr Rheumatol Rep. 2019 Feb 27;21(4):12. doi: 10.1007/s11926-019-0809-x. Curr Rheumatol Rep. 2019. PMID: 30810824 Review. - Diagnostic utility of protein to creatinine ratio (P/C ratio) in spot urine sample within routine clinical practice.
Kamińska J, Dymicka-Piekarska V, Tomaszewska J, Matowicka-Karna J, Koper-Lenkiewicz OM. Kamińska J, et al. Crit Rev Clin Lab Sci. 2020 Aug;57(5):345-364. doi: 10.1080/10408363.2020.1723487. Epub 2020 Feb 14. Crit Rev Clin Lab Sci. 2020. PMID: 32058809 Review.
Cited by
- Random spot urine protein/creatinine ratio is unreliable for estimating 24-hour proteinuria in individual systemic lupus erythematosus nephritis patients.
Hebert LA, Birmingham DJ, Shidham G, Rovin B, Nagaraja HN, Yu CY. Hebert LA, et al. Nephron Clin Pract. 2009;113(3):c177-82. doi: 10.1159/000232599. Epub 2009 Aug 12. Nephron Clin Pract. 2009. PMID: 19672116 Free PMC article. - Differential diagnosis of glomerular disease: a systematic and inclusive approach.
Hebert LA, Parikh S, Prosek J, Nadasdy T, Rovin BH. Hebert LA, et al. Am J Nephrol. 2013;38(3):253-66. doi: 10.1159/000354390. Epub 2013 Sep 13. Am J Nephrol. 2013. PMID: 24052039 Free PMC article. Review. - Chapter 2: General principles in the management of glomerular disease.
[No authors listed] [No authors listed] Kidney Int Suppl (2011). 2012 Jun;2(2):156-162. doi: 10.1038/kisup.2012.15. Kidney Int Suppl (2011). 2012. PMID: 25018928 Free PMC article. No abstract available. - Use of biomarkers in the management of children with lupus.
Binder E, Edelbauer M. Binder E, et al. Curr Rheumatol Rep. 2013 Mar;15(3):312. doi: 10.1007/s11926-012-0312-0. Curr Rheumatol Rep. 2013. PMID: 23355230 Review. - Intraindividual variations of urinary biomarkers in hospitalized children with glomerular diseases: a prospective observational study.
Zhou J, Zhong X, Xiao H, Xu K, Nair V, Larkina M, Ju W, Ding J. Zhou J, et al. Eur J Pediatr. 2023 Aug;182(8):3755-3764. doi: 10.1007/s00431-023-05042-9. Epub 2023 Jun 10. Eur J Pediatr. 2023. PMID: 37300718 Free PMC article.
References
- Austin HA. Clinical evaluation and monitoring of lupus kidney disease. Lupus. 1998;7:618–621. - PubMed
- Balow JE. Clinical presentation and monitoring of lupus nephritis. Lupus. 2005;14:25–30. - PubMed
- Contreras G, Pardo V, Cely C, et al. Factors associated with poor outcomes in patients with lupus nephritis. Lupus. 2005;14:890–895. - PubMed
- Renal Disease Subcommittee of the American College of Rheumatology Ad Hoc Committee on Systemic Lupus Erythematosus Response Criteria. The American College of Rheumatology response criteria for proliferative and membranous renal disease in systemic lupus erythematosus clinical trials. Arthritis Rheum. 2006;54:421–432. - PubMed
- Christopher-Stine L, Petri M, Astor BC, et al. Urine protein-to-creatinine ratio is a reliable measure of proteinuria in lupus nephritis. J Rheumatol. 2004;31:1557–1559. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P01 DK055546/DK/NIDDK NIH HHS/United States
- UL1RR025755-01/RR/NCRR NIH HHS/United States
- DK55546/DK/NIDDK NIH HHS/United States
- M01-RR00052/RR/NCRR NIH HHS/United States
- M01 RR000052/RR/NCRR NIH HHS/United States
- R01 DK074661/DK/NIDDK NIH HHS/United States
- AR43727/AR/NIAMS NIH HHS/United States
- R01 AR043727/AR/NIAMS NIH HHS/United States
- UL1 RR025755/RR/NCRR NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources