Inflammatory bowel disease and mutations affecting the interleukin-10 receptor - PubMed (original) (raw)
Case Reports
. 2009 Nov 19;361(21):2033-45.
doi: 10.1056/NEJMoa0907206. Epub 2009 Nov 4.
Daniel Kotlarz, Kaan Boztug, E Michael Gertz, Alejandro A Schäffer, Fatih Noyan, Mario Perro, Jana Diestelhorst, Anna Allroth, Dhaarini Murugan, Nadine Hätscher, Dietmar Pfeifer, Karl-Walter Sykora, Martin Sauer, Hans Kreipe, Martin Lacher, Rainer Nustede, Cristina Woellner, Ulrich Baumann, Ulrich Salzer, Sibylle Koletzko, Neil Shah, Anthony W Segal, Axel Sauerbrey, Stephan Buderus, Scott B Snapper, Bodo Grimbacher, Christoph Klein
Affiliations
- PMID: 19890111
- PMCID: PMC2787406
- DOI: 10.1056/NEJMoa0907206
Case Reports
Inflammatory bowel disease and mutations affecting the interleukin-10 receptor
Erik-Oliver Glocker et al. N Engl J Med. 2009.
Abstract
Background: The molecular cause of inflammatory bowel disease is largely unknown.
Methods: We performed genetic-linkage analysis and candidate-gene sequencing on samples from two unrelated consanguineous families with children who were affected by early-onset inflammatory bowel disease. We screened six additional patients with early-onset colitis for mutations in two candidate genes and carried out functional assays in patients' peripheral-blood mononuclear cells. We performed an allogeneic hematopoietic stem-cell transplantation in one patient.
Results: In four of nine patients with early-onset colitis, we identified three distinct homozygous mutations in genes IL10RA and IL10RB, encoding the IL10R1 and IL10R2 proteins, respectively, which form a heterotetramer to make up the interleukin-10 receptor. The mutations abrogate interleukin-10-induced signaling, as shown by deficient STAT3 (signal transducer and activator of transcription 3) phosphorylation on stimulation with interleukin-10. Consistent with this observation was the increased secretion of tumor necrosis factor alpha and other proinflammatory cytokines from peripheral-blood mononuclear cells from patients who were deficient in IL10R subunit proteins, suggesting that interleukin-10-dependent "negative feedback" regulation is disrupted in these cells. The allogeneic stem-cell transplantation performed in one patient was successful.
Conclusions: Mutations in genes encoding the IL10R subunit proteins were found in patients with early-onset enterocolitis, involving hyperinflammatory immune responses in the intestine. Allogeneic stem-cell transplantation resulted in disease remission in one patient.
2009 Massachusetts Medical Society
Figures
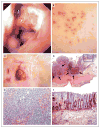
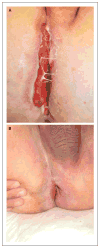
Figure 1. Clinical Phenotype in a Patient with a Deficiency in the Interleukin-10 Receptor
The index patient in Family B, Patient II-5, had evidence of erosive lesions on colonoscopy, shown in Panel A, and of skin folliculitis, shown in Panel B. Panel C shows the patient’s status after ileostomy. Panel D shows the histopathological findings in a specimen from the colon obtained during resection in the patient, revealing circumscribed ulcerations of the mucosa from which pear-shaped intramural abscesses (arrows) extend into the submucosa and muscularis propria. Panel E shows a higher magnification of inset 1 in Panel D, revealing intramural microabscesses. Panel F shows a higher magnification of inset 2 in Panel D, revealing neighboring mucosa with scarce, superficial lymphoplasmocytic infiltrates without glandular distortion. There is minimal intramural lymphocytic hyperplasia (as seen in the dark area just to the left of inset 1 in Panel D) and no thickening of the intestinal wall as a result of fibrosis or granulomas.
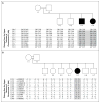
Figure 2. Haplotypes in Families A and B
Panel A shows the pedigree of consanguineous Family A, which had two children with early-onset inflammatory bowel disease (Patients II-3 and II-4) and allelic segregation on chromosome 21q. Panel B shows the pedigree of a second consanguineous family, Family B, which had one affected child (Patient II-5) and allelic segregation on chromosome 11q. Squares indicate male family members, and circles female family members. Shading indicates the series of homozygous markers in the affected patients.
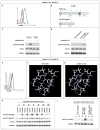
Figure 3. Structure and Functional Analysis of Mutations in the Interleukin-10 Receptor in Two Patients
Panel A shows a representative analysis by fluorescence-activated cell sorting (FACS) that illustrates the lack of expression of IL10R2 protein in the index patient in Family A (Patient II-3), who carried an IL10RB W159X mutation. An isotype control sample is indicated by gray shading, a sample from a control subject by a blue line, and the sample from Patient II-3 by a red line. Panel B shows a schematic drawing of the IL10R2 protein, indicating known protein domains and localization of the nonsense W159X mutation, as compared with a wild-type sample. ECD denotes extracellular domain, ICD intracellular domain, SP signaling peptide, and TMD transmembrane domain. Panel C shows defective STAT3 (signal transducer and activator of transcription 3) phosphorylation (p-STAT3) at the residue tyrosine 705 on stimulation with interleukin-10 in peripheral-blood mononuclear cells (PBMCs) from Patient II-3, as compared with intact phosphorylation in a control sample. Panel D shows the reconstitution of STAT3 phosphorylation on transduction of cell lines from Patient II-3, with a lentiviral vector encoding wild-type (WT) IL10R2. Panel E shows normal expression levels of IL10R1 on FACS analysis in the index patient in Family B (Patient II-5), who carried an IL10RA G141R mutation. An isotype control sample is indicated by gray shading, a sample from a control subject by a blue line, and the sample from Patient II-5 by a red line. Panel F shows a structural model of wild-type IL10R1. Residues Leu132, Glu133, Ile139, Gly141, Lys142, and Phe190 are labeled. Also shown are nearby residues that participate with Gly141 or its neighbors in a beta-sheet conformation. The residues that are illustrated are those that are reported to have contact with Gly141 in wild-type IL10R1 (according to RankViaContact31) or with Arg141 in the G141R mutation and that lie within 6 Å. Panel G shows a structural model of the G141R mutation in IL10R1 (from Protein Data Bank structure 1Y6K, chain R). Putative hydrogen bonds are shown as green dashed lines in Panels F and G, and steric clashes are shown as purple dashed lines in Panel G. Panel H shows a Western blot analysis of STAT3 in PBMCs from Patient II-5 after stimulation for various periods with interleukin-10, as compared with a control sample. Panel I shows a Western blot analysis of phosphorylation of STAT3 at tyrosine 705 in interleukin-10–stimulated HeLa cells that were retrovirally transduced with wild-type IL10R2 along with either wild-type IL10R1 or IL10R1 with mutant G141R. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as a control for the experiments in Panels C, D, H, and I.
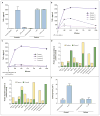
Figure 4. Defective Down-Regulation of Proinflammatory Cytokines Mediated by Interleukin-10 in Mononuclear Cells with a Mutation in the Interleukin-10 Receptor
Panel A shows defective inhibition of the release of tumor necrosis factor α (TNF- α) by costimulation with interleukin-10 in lipopolysaccharide (LPS)–stimulated macrophages from the index patient in Family A (Patient II-3), as compared with the mean value from samples obtained from five control subjects, as measured by enzyme-linked immunosorbent assay (ELISA). Panel B shows a similar ELISA measuring TNF- α secretion in LPS-stimulated peripheral-blood mononuclear cells (PBMCs) from the index patient in Family B (Patient II-5) and three control subjects. Panel C shows defective interleukin-10–mediated suppression of TNF- α secretion in LPS-stimulated cells from Patient II-5 in Family B. Panel D shows increased secretion of proinflammatory cytokines upon stimulation of cells with LPS in Patient II-5, as compared with a healthy control subject. These cytokines include MIP-1_α_ and MIP-1_β_ (macrophage inflammatory proteins 1_α_ and 1_β_), MCP1 (monocyte chemoattractant protein), and RANTES (regulated on activation, normal T expressed and secreted protein). Panel E shows the abrogated effect of interleukin-10 on the release of proinflammatory cytokines in cells from the patient that were costimulated with LPS and interleukin-10, as compared with a healthy control subject. Cells from Patient II-5 show increased secretion of inflammatory cytokines, which could not be counteracted by costimulation with exogenous interleukin-10. Panel F shows defective interleukin-10–mediated induction of messenger RNA expression in the suppressor of cytokine signaling 3 gene (SOCS3) in Patient II-3 in Family A; induction was more than four times as great in a control sample. Expression levels were measured relative to _β_-actin as a housekeeping gene. The I bars indicate standard errors.
Figure 5. Success of Allogeneic Hematopoietic Stem-Cell Transplantation
In Patient II-3 from Family A, who had severe anocutaneous fistulas that were resistant to therapy (Panel A), allogeneic stem-cell transplantation resulted in clinical amelioration of all effects of disease (Panel B).
Comment in
- Interleukin-10 in inflammatory bowel disease.
Kelsall B. Kelsall B. N Engl J Med. 2009 Nov 19;361(21):2091-3. doi: 10.1056/NEJMe0909225. Epub 2009 Nov 4. N Engl J Med. 2009. PMID: 19890110 No abstract available. - Defective interleukin-10 signaling in human inflammatory bowel disease.
Watanabe T, Chiba T. Watanabe T, et al. Gastroenterology. 2010 May;138(5):2016-8. doi: 10.1053/j.gastro.2010.03.018. Epub 2010 Mar 19. Gastroenterology. 2010. PMID: 20303964 No abstract available. - IL-10RA truncation mutations and Semite populations.
Gasche C, Reinisch W, Huber WD, Leshinsky-Silver E, Levine A, Abdul-Baki H, Sharara AI, Ajlouni Y, Sefiani A. Gasche C, et al. Inflamm Bowel Dis. 2011 Jun;17(6):1438. doi: 10.1002/ibd.21495. Epub 2010 Sep 14. Inflamm Bowel Dis. 2011. PMID: 20842643 No abstract available.
References
- Podolsky DK. Inflammatory bowel disease. N Engl J Med. 2002;347:417–29. - PubMed
- Xavier RJ, Podolsky DK. Unravelling the pathogenesis of inflammatory bowel disease. Nature. 2007;448:427–34. - PubMed
- Fried K, Vure E. A lethal autosomal recessive enterocolitis of early infancy. Clin Genet. 1974;6:195–6. - PubMed
- Mégarbané A, Sayad R. Early lethal autosomal recessive enterocolitis: report of a second family. Clin Genet. 2007;71:89–90. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous