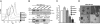Axl is an essential epithelial-to-mesenchymal transition-induced regulator of breast cancer metastasis and patient survival - PubMed (original) (raw)
. 2010 Jan 19;107(3):1124-9.
doi: 10.1073/pnas.0909333107. Epub 2009 Dec 28.
Crina Tiron, Torill Høiby, Ingunn Stefansson, Hallvard Haugen, Tone Sandal, Karin Collett, Shan Li, Emmet McCormack, Bjørn Tore Gjertsen, David R Micklem, Lars A Akslen, Carlotta Glackin, James B Lorens
Affiliations
- PMID: 20080645
- PMCID: PMC2824310
- DOI: 10.1073/pnas.0909333107
Axl is an essential epithelial-to-mesenchymal transition-induced regulator of breast cancer metastasis and patient survival
Christine Gjerdrum et al. Proc Natl Acad Sci U S A. 2010.
Abstract
Metastasis underlies the majority of cancer-related deaths. Thus, furthering our understanding of the molecular mechanisms that enable tumor cell dissemination is a vital health issue. Epithelial-to-mesenchymal transitions (EMTs) endow carcinoma cells with enhanced migratory and survival attributes that facilitate malignant progression. Characterization of EMT effectors is likely to yield new insights into metastasis and novel avenues for treatment. We show that the presence of the receptor tyrosine kinase Axl in primary breast cancers independently predicts strongly reduced overall patient survival, and that matched patient metastatic lesions show enhanced Axl expression. We demonstrate that Axl is strongly induced by EMT in immortalized mammary epithelial cells that establishes an autocrine signaling loop with its ligand, Gas6. Epiallelic RNA interference analysis in metastatic breast cancer cells delineated a distinct threshold of Axl expression for mesenchymal-like in vitro cell invasiveness and formation of tumors in foreign and tissue-engineered microenvironments in vivo. Importantly, in two different optical imaging-based experimental breast cancer models, Axl knockdown completely prevented the spread of highly metastatic breast carcinoma cells from the mammary gland to lymph nodes and several major organs and increased overall survival. These findings suggest that Axl represents a downstream effector of the tumor cell EMT that is required for breast cancer metastasis. Thus, the detection and targeted treatment of Axl-expressing tumors represents an important new therapeutic strategy for breast cancer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
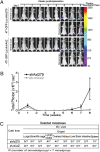
Fig. 1.
Axl expression is a strong negative prognostic factor for human breast cancer survival. (A) Breast tumors are stratified by weak (Upper, 60% of patients) and strong (Lower, 40% of patients) Axl expression. (B) Univariate survival analysis (Kaplan-Meier method) of breast cancer patients shows a strong negative prognostic value of Axl expression (P = .035, log-rank test). (C) Multivariate survival analysis (proportional hazards method) indicates a negative, independent prognostic importance of Axl expression (P = .021, likelihood ratio test), in addition to the independent prognostic impact of histological grade and axillary lymph node status. HR, hazard ratio. (D) Axl expression was significantly stronger in metastases (P < 0.0005, Wilcoxon rank-sum test) from matched pairs (n = 37) of primary (Left, Upper, Axl-negative; Lower, Axl-positive) and metastatic (Right, Upper, liver; Lower, bone) breast carcinoma.
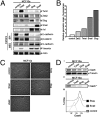
Fig. 2.
Axl is required for breast cancer cell invasiveness (A and B) Epiallelic MDA-MB-231 cells comprising Axl-targeting shRNA (shAxl2, shAxl280, shAxl278, and shAxl279) and control shRNA (shLuc) show graded Axl surface expression by flow cytometry (A) and total and phosporylated Axl (pAxl) levels by immunoblotting (B). (C) Invasiveness of epiallelic MDA-MB-231 cells is Axl-dependent. *P < .05, (paired t test). (D) Axl is required for invasive, stellate growth of MDA-MB-231 cells in 3D Matrigel (Lower, shAxl2; Upper, shLuc).
Fig. 3.
Axl is up-regulated by EMT inducers in breast epithelial cells. (A) MCF10a cells transduced with retroviral vectors encoding Twist, Zeb2, Slug, and Snail up-regulate Axl, lose epithelial markers (EPI), and increase mesenchymal (Mes) markers relative to vector control (GFP). (B) Axl surface expression is up-regulated in MCF10a cells transduced with Twist, Zeb2, Slug, or Snail retroviral vectors measured by flow cytometry. (C) These cells acquire mesenchymal morphology at 72 h postseeding. (D) MCF10a cells constitutively express (Top, Western blot, total lysate) and secrete (Middle, Western blot, conditioned medium) Gas6 that becomes cell-associated (Bottom, anti-Gas6 flow cytometry analysis) on Slug- and Snail-induced Axl expression.
Fig. 4.
Tissue-engineered breast tumors require Axl expression. (A) MDA-MB-231/GFP-Luc tumor growth and colonization within polylactic acid tissue engineering scaffolds in NOD-SCID mice (n = 6 per group) is Axl dependent. (B) Axl is required for the growth of tricellular tissue implants comprising primary human microvascular cells (ECs), vascular smooth muscle cells (SMCs), and MDA-MB-231/GFP-Luc in NOD-SCID mice (n = 7 per group). Temporal in vivo bioluminescence image analysis was used to measure tumor cell number (Upper, total photon) and the extent of radial infiltration (Lower, signal diameter) in controls (solid line) and shAxl2 implants (dashed line). (C) Intrascaffold human vessel diameter (Upper) is unaffected, whereas microvascular density (Lower) is slightly enhanced in tissue-engineered (tricellular) tumors inhibited by shAxl2 expression. *P < .05; **P < .005; ***P < .0005 (paired t test) versus control.
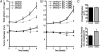
Fig. 5.
In vivo epiallelic analysis reveals a distinct Axl expression threshold required for breast tumor formation. (A) Temporal in vivo bioluminescence image analysis of tumor growth (total photon) and radial infiltration (signal diameter) by s.c. Axl epiallelic MDA-MB-231/GFP-Luc xenografts in NOD/SCID mice (n = 6 per group) comprising shAxl278, shAx280, and shAxl2 normalized to shAxl279 (ineffective shRNA). (B) Tumor growth (normalized 28-day total photon measurements) plotted versus Axl knockdown (geometric mean fluorescence) reveal a threshold of 80% reduced surface expression to attain an antitumor effect (therapeutic benefit). *P < .05; **P < .005 (paired t test).
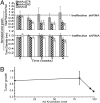
Fig. 6.
Axl is required for human breast carcinoma cells to metastasize from the mammary gland of NOD-SCID mice. (A) Temporal in vivo bioluminescence imaging of mammary fat pad growth of orthotropically injected control and shAxl2-expressing MDA-MB-231-D3H2LN /GFP-Luc cells in NOD/SCID mice (Upper) and Axl-dependent formation of spontaneous thoracic sentinel lymph node metastasis (Lower). (B) Biophotonic quantitation of primary mammary tumor cell number (total photon) and radial infiltration (signal diameter; Upper) and thoracic lymph node metastatic cell number (total photon; Lower) in MDA-MB-231-D3H2LN/GFP-Luc control (solid line) and shAxl2 (dashed line) cell–injected NOD/SCID mice (n = 6 per group). *P < .05; **P < .005; ***P < .0005 compared with control (paired t test). (C) Survey of spontaneous metastasis (at 9 weeks postorthotopic implantation) site monitored by ex vivo bioluminescence detection of MDA-MB-231-D3H2LN/GFP-Luc cells in excised organs shows that tumor cell dissemination is Axl-dependent. (D) Univariate survival analysis (Kaplan-Meier method) between control (vector) and shAxl2 orthographically MDA-MB-231-D3H2LN/GFP-Luc–injected mice demonstrates that overall survival is Axl-dependent (P = .001, log-rank test).
Fig. 7.
Axl is essential for postimmune response recurrence and metastasis of syngeneic breast carcinoma cells in BALB/c mice. (A) Temporal in vivo bioluminescence imaging of orthotropic (mammary fat pad)–injected syngenic 4T1-GFP-Luc cells expressing mouse Axl shRNA (4T1-GFP-Luc-shmAxl2) or negative control human Axl shRNA (4T1-GFP-Luc-shAxl279) into BALB/c mice. (B) Quantitation of whole-body bioluminescence (total photon) in control (4T1-GFP-Luc-shAxl279; solid line) and Axl-knockdown (4T1-GFP-Luc-shmAxl2; gray line)-injected BALB/c mice over an 8-week period reveals that metastasis is Axl-dependent. *P < .05 (t test); n = 7 mice per group. (C) Survey of metastasis sites (at 8 weeks postorthotopic implantation) to monitored by ex vivo bioluminescence detection of 4T1-GFP-Luc cells in excised organs from control or Axl-knockdown tumor-bearing BALB/c mice.
Similar articles
- Warfarin Blocks Gas6-Mediated Axl Activation Required for Pancreatic Cancer Epithelial Plasticity and Metastasis.
Kirane A, Ludwig KF, Sorrelle N, Haaland G, Sandal T, Ranaweera R, Toombs JE, Wang M, Dineen SP, Micklem D, Dellinger MT, Lorens JB, Brekken RA. Kirane A, et al. Cancer Res. 2015 Sep 15;75(18):3699-705. doi: 10.1158/0008-5472.CAN-14-2887-T. Epub 2015 Jul 23. Cancer Res. 2015. PMID: 26206560 Free PMC article. - The Receptor Tyrosine Kinase AXL Is Required at Multiple Steps of the Metastatic Cascade during HER2-Positive Breast Cancer Progression.
Goyette MA, Duhamel S, Aubert L, Pelletier A, Savage P, Thibault MP, Johnson RM, Carmeliet P, Basik M, Gaboury L, Muller WJ, Park M, Roux PP, Gratton JP, Côté JF. Goyette MA, et al. Cell Rep. 2018 May 1;23(5):1476-1490. doi: 10.1016/j.celrep.2018.04.019. Cell Rep. 2018. PMID: 29719259 - R428, a selective small molecule inhibitor of Axl kinase, blocks tumor spread and prolongs survival in models of metastatic breast cancer.
Holland SJ, Pan A, Franci C, Hu Y, Chang B, Li W, Duan M, Torneros A, Yu J, Heckrodt TJ, Zhang J, Ding P, Apatira A, Chua J, Brandt R, Pine P, Goff D, Singh R, Payan DG, Hitoshi Y. Holland SJ, et al. Cancer Res. 2010 Feb 15;70(4):1544-54. doi: 10.1158/0008-5472.CAN-09-2997. Epub 2010 Feb 9. Cancer Res. 2010. PMID: 20145120 - Epithelial-to-mesenchymal transition: lessons from development, insights into cancer and the potential of EMT-subtype based therapeutic intervention.
Antony J, Thiery JP, Huang RY. Antony J, et al. Phys Biol. 2019 May 7;16(4):041004. doi: 10.1088/1478-3975/ab157a. Phys Biol. 2019. PMID: 30939460 Review. - AXL Receptor in Breast Cancer: Molecular Involvement and Therapeutic Limitations.
Falcone I, Conciatori F, Bazzichetto C, Bria E, Carbognin L, Malaguti P, Ferretti G, Cognetti F, Milella M, Ciuffreda L. Falcone I, et al. Int J Mol Sci. 2020 Nov 10;21(22):8419. doi: 10.3390/ijms21228419. Int J Mol Sci. 2020. PMID: 33182542 Free PMC article. Review.
Cited by
- AXL, an Important Host Factor for DENV and ZIKV Replication.
Xie S, Zhang H, Liang Z, Yang X, Cao R. Xie S, et al. Front Cell Infect Microbiol. 2021 Mar 22;11:575346. doi: 10.3389/fcimb.2021.575346. eCollection 2021. Front Cell Infect Microbiol. 2021. PMID: 33954117 Free PMC article. Review. - Mer receptor tyrosine kinase is frequently overexpressed in human non-small cell lung cancer, confirming resistance to erlotinib.
Xie S, Li Y, Li X, Wang L, Yang N, Wang Y, Wei H. Xie S, et al. Oncotarget. 2015 Apr 20;6(11):9206-19. doi: 10.18632/oncotarget.3280. Oncotarget. 2015. PMID: 25826078 Free PMC article. - Warfarin Blocks Gas6-Mediated Axl Activation Required for Pancreatic Cancer Epithelial Plasticity and Metastasis.
Kirane A, Ludwig KF, Sorrelle N, Haaland G, Sandal T, Ranaweera R, Toombs JE, Wang M, Dineen SP, Micklem D, Dellinger MT, Lorens JB, Brekken RA. Kirane A, et al. Cancer Res. 2015 Sep 15;75(18):3699-705. doi: 10.1158/0008-5472.CAN-14-2887-T. Epub 2015 Jul 23. Cancer Res. 2015. PMID: 26206560 Free PMC article. - Boosting immunotherapy efficacy: Empowering the Potency of Dendritic cells loaded with breast cancer lysates through CTLA-4 suppression.
Bakhshivand M, Masoumi J, Ghorbaninezhad F, Aghebati-Maleki L, Shanebandi D, Sandoghchian Shotorbani S, Jadidi-Niaragh F, Baghbanzadeh A, Hemmat N, Baghbani E, Ghaffari A, Baradaran B. Bakhshivand M, et al. Heliyon. 2024 Sep 10;10(18):e37699. doi: 10.1016/j.heliyon.2024.e37699. eCollection 2024 Sep 30. Heliyon. 2024. PMID: 39309891 Free PMC article. - Engineered Adoptive T-Cell Therapies for Breast Cancer: Current Progress, Challenges, and Potential.
Chamorro DF, Somes LK, Hoyos V. Chamorro DF, et al. Cancers (Basel). 2023 Dec 26;16(1):124. doi: 10.3390/cancers16010124. Cancers (Basel). 2023. PMID: 38201551 Free PMC article. Review.
References
- Gupta PB, Mani S, Yang J, Hartwell K, Weinberg RA. The evolving portrait of cancer metastasis. Cold Spring Harb Symp Quant Biol. 2005;70:291–297. - PubMed
- Kaplan RN, Psaila B, Lyden D. Bone marrow cells in the “pre-metastatic niche”: Within bone and beyond. Cancer Metastasis Rev. 2006;25:521–529. - PubMed
- Thiery JP. Epithelial–mesenchymal transitions in tumour progression. Nat Rev Cancer. 2002;2:442–454. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous