Murine features of neurogenesis in the human hippocampus across the lifespan from 0 to 100 years - PubMed (original) (raw)
Murine features of neurogenesis in the human hippocampus across the lifespan from 0 to 100 years
Rolf Knoth et al. PLoS One. 2010.
Abstract
Background: Essentially all knowledge about adult hippocampal neurogenesis in humans still comes from one seminal study by Eriksson et al. in 1998, although several others have provided suggestive findings. But only little information has been available in how far the situation in animal models would reflect the conditions in the adult and aging human brain. We therefore here mapped numerous features associated with adult neurogenesis in rodents in samples from human hippocampus across the entire lifespan. Such data would not offer proof of adult neurogenesis in humans, because it is based on the assumption that humans and rodents share marker expression patterns in adult neurogenesis. Nevertheless, together the data provide valuable information at least about the presence of markers, for which a link to adult neurogenesis might more reasonably be assumed than for others, in the adult human brain and their change with increasing age.
Methods and findings: In rodents, doublecortin (DCX) is transiently expressed during adult neurogenesis and within the neurogenic niche of the dentate gyrus can serve as a valuable marker. We validated DCX as marker of granule cell development in fetal human tissue and used DCX expression as seed to examine the dentate gyrus for additional neurogenesis-associated features across the lifespan. We studied 54 individuals and detected DCX expression between birth and 100 years of age. Caveats for post-mortem analyses of human tissues apply but all samples were free of signs of ischemia and activated caspase-3. Fourteen markers related to adult hippocampal neurogenesis in rodents were assessed in DCX-positive cells. Total numbers of DCX expressing cells declined exponentially with increasing age, and co-expression of DCX with the other markers decreased. This argued against a non-specific re-appearance of immature markers in specimen from old brains. Early postnatally all 14 markers were co-expressed in DCX-positive cells. Until 30 to 40 years of age, for example, an overlap of DCX with Ki67, Mcm2, Sox2, Nestin, Prox1, PSA-NCAM, Calretinin, NeuN, and others was detected, and some key markers (Nestin, Sox2, Prox1) remained co-expressed into oldest age.
Conclusions: Our data suggest that in the adult human hippocampus neurogenesis-associated features that have been identified in rodents show patterns, as well as qualitative and quantitative age-related changes, that are similar to the course of adult hippocampal neurogenesis in rodents. Consequently, although further validation as well as the application of independent methodology (e.g. electron microscopy and cell culture work) is desirable, our data will help to devise the framework for specific research on cellular plasticity in the aging human hippocampus.
Conflict of interest statement
Competing Interests: The authors have declared that no competing interests exist.
Figures
Figure 1. Electrophoretic separation of human brain proteins (25 µg in lane 1, and 50 µg in lanes 2 and 3) and subsequent immunoblotting against DCX and GAPDH.
Lane 1: Fetus of GW 20, elective abortion, cortical tissue; lane 2: child of age 3 years, healthy hippocampus; lane 3: adult of age 35 years, suffering from temporal lobe epilepsy (TLE), resected hippocampal tissue. The doublecortin antibody (sc-8066) reacted with proteins in the 45 kDa range, which corresponds to the MGrel. of DCX. The signal is strong in the fetal brain and much weaker in the juvenile and adult samples. The GAPDH signal at the 36 kDa position serves as MGrel and loading control.
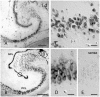
Figure 2. Combined in situ hybridization for DCX mRNA (blue-gray) and immunostaining for DCX protein (red) in human fetal tissue (GW 11).
A, B, The overlapping staining indicates the co-expression of DCX mRNA and protein. In cortical tissue (A), as well as in the ganglionic eminence (B), the mRNA is not restricted perinuclearly, but is also found in the transverse oriented processes of the round-shaped cells. C, Negative control of the in situ hybridization. DCX immunohistochemistry was performed following the in situ hybridization protocol with the DCX sense probe. Scale bars, 25 µm.
Figure 3. Staining of DCX mRNA by hybridization with an alkaline phosphatase labeled anti-sense cRNA probe in the hippocampus at different ages (non-radioactive in situ hybridization).
A, B, From birth to adulthood (A: 1 day, B: 28 years of age), hippocampal DCX labeling reveals a distinct staining pattern in the granule cell layer and hippocampus proper. C, In most granule cells (neonatal DG), the mRNA signal occurs outside the nucleus in a small cytoplasmic rim. D, Higher magnification of the DG area marked in (B). E, Application of a labelled sense cRNA probe to a serial section of (B). The absence of sense-probe labelling confirmed the specificity of the detection system for DCX-mRNA/cRNA antisense probe hybrids. Differential Interference Contrast. Scale bars A, B, 1 mm; C-E, 25 µm.
Figure 4. Morphologic variability of DCX+ cells in the granular cell layer (GCL) across the lifespan of humans (A-F) and mice (G-K).
Postnatal, numerous DCX+ neurite-bearing cells are scattered throughout the GCL (A). In the adulthood, DCX+ cells are gradually less differentiated in relation to growing age. Initially, some cells show a neuron-like morphology (B,D), whereas the majority of them appears increasingly more undifferentiated and strongly DCX-stained (B-F). In the juvenile mouse hippocampus a multitude of DCX+ cells delineate the GCL formation (G,I). Very old mice, however, exhibit only a few DCX expressing granule cells (H,K). DAB immunohistochemistry of DCX in paraffin embedded brain samples. Length of scale bars as indicated.
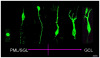
Figure 5. Survey of morphologic variation of DCX-expressing progenitor cells and immature neurons in the DG of a young human subject (5 years-old).
Increasing cellular differentiation (dendrites and axons) can be observed starting from the hilar polymorphic layer/subgranular layer (PML/SGL) throughout the GCL up to the inner molecular layer. However, undifferentiated round-shaped cells were always found throughout the entire GCL. This picture is a montage of DCX labeled granule cell and sorted increasingly maturated from left to right. Dendrites are directed towards the granule cell layer (top) and the axons towards the hilus (bottom). Scale bar, 10 µm.
Figure 6. Temporal expression profile and intracellular colabelling in human brain tissue of DCX and additional markers associated with adult hippocampal neurogenesis in rodents.
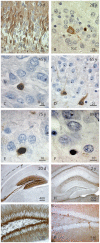
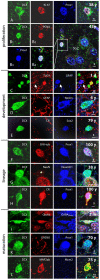
Figure 7. A, B Proliferation markers Ki67 or PCNA, co-expressed with the developmental and lineage markers DCX and Prox1, identified SGZ cells as cycling neuronal precursor cells.
C–E, The SGZ of juvenile and adult subjects exhibits immature DCX+ cells co-expressing development markers characteristic also for developing adult-generated granule cells in rodents. C, In neonates, some immature granule cells co-express DCX and early postmitotic neuronal marker, TUC4. Glia (arrow) did not react against the neuron-specific antibodies applied. D, Early neuro-ectodermal marker nestin labels a bipolar DCX+ cell and neuropil structures not matching GFAP+ astrocytic processes in the PML of a 6 years-old subject. E, Transcription factor Sox2, characteristic for precursor cells, remains expressed in DCX+ cells lacking the transitional maturation marker calretinin. F–H, Lineage markers were co-stained with DCX up to oldest age. F, β-III-tubulin, the early neuronal cytoskeleton marker, is expressed also in a DCX+/Prox1+ cell of a 100 year-old individual. G, At age 38 years, NeuroD1, one of the earliest known lineage markers in precursor cells, was detected in a round-shaped DCX+ precursor weakly reactive for maturation marker NeuN (asterisk). H, Even at age 100 years, granule cell-specific transcription factor Prox1 and transient postmitotic neuronal maturation marker calretinin were found together with DCX. I–L, Immunoreactivity for neuronal maturation markers in DCX+ cells between 2 month and 75 years. I, Bipolar migratory DCX+ cell expressed markers of neuronal transmission, like glutamatergic and GABAergic receptors (GluR4 and GABAARec.) K, DCX+/Prox1+ cells may show GABAergic synapses, here demonstrated by GAD65 in axonal terminals around the cell soma. L, MAP2ab immunostaining could be found among matured DCX+ cells in the adult SGZ. Length of scale bars as indicated.
Figure 8. Continuation of Fig. 7. M, DCX and proliferation marker Mcm2, further indicating a proliferative “neuroblast”-like cell.
N, Immature neurons express DCX together with postmitotic neuronal marker NeuN; here at age of 75 years. O, Expression of PCNA in GFAP+ cells might be indicative of ongoing stem cell activity in the SGZ but might also reflect classical astrocytic proliferation or DNA repair (see discussion for details). The Z/X- as well as Z/Y-view of the double labeled section clarifies the co-expression of both proteins in the round-shaped cell.
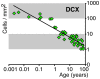
Figure 9. Estimation of DCX+ cell density in the GCL including SGZ across the entire human lifespan.
Comprising data from 45 subjects, between 1 day and 94 years, the number of labeled cells in the DG were plotted against the age of the individual. The regression curve shows a log-log-linear decrease (Pearson’s Correlation Coefficient (PPC): -0.95102; p = 0.0001).
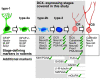
Figure 10. Summary of marker expression in the human hippocampus related to the milestones of adult hippocampal neurogenesis in rodents .
The frame indicates the stages of adult neurogenesis covered in this study: all additional markers were tested together with DCX. Marker combinations that we could detect in our samples are marked with a green check. Not all markers were found at all ages, see Figs. 6 for details.
Similar articles
- Characterization of dsRed2-positive cells in the doublecortin-dsRed2 transgenic adult rat retina.
Trost A, Schroedl F, Marschallinger J, Rivera FJ, Bogner B, Runge C, Couillard-Despres S, Aigner L, Reitsamer HA. Trost A, et al. Histochem Cell Biol. 2014 Dec;142(6):601-17. doi: 10.1007/s00418-014-1259-1. Epub 2014 Aug 20. Histochem Cell Biol. 2014. PMID: 25138677 - Properties of doublecortin-(DCX)-expressing cells in the piriform cortex compared to the neurogenic dentate gyrus of adult mice.
Klempin F, Kronenberg G, Cheung G, Kettenmann H, Kempermann G. Klempin F, et al. PLoS One. 2011;6(10):e25760. doi: 10.1371/journal.pone.0025760. Epub 2011 Oct 13. PLoS One. 2011. PMID: 22022443 Free PMC article. - Doublecortin-expressing cell types in temporal lobe epilepsy.
Liu JYW, Matarin M, Reeves C, McEvoy AW, Miserocchi A, Thompson P, Sisodiya SM, Thom M. Liu JYW, et al. Acta Neuropathol Commun. 2018 Jul 13;6(1):60. doi: 10.1186/s40478-018-0566-5. Acta Neuropathol Commun. 2018. PMID: 30005693 Free PMC article. - Positive Controls in Adults and Children Support That Very Few, If Any, New Neurons Are Born in the Adult Human Hippocampus.
Sorrells SF, Paredes MF, Zhang Z, Kang G, Pastor-Alonso O, Biagiotti S, Page CE, Sandoval K, Knox A, Connolly A, Huang EJ, Garcia-Verdugo JM, Oldham MC, Yang Z, Alvarez-Buylla A. Sorrells SF, et al. J Neurosci. 2021 Mar 24;41(12):2554-2565. doi: 10.1523/JNEUROSCI.0676-20.2020. J Neurosci. 2021. PMID: 33762407 Free PMC article. Review. - Immunohistological markers for proliferative events, gliogenesis, and neurogenesis within the adult hippocampus.
von Bohlen und Halbach O. von Bohlen und Halbach O. Cell Tissue Res. 2011 Jul;345(1):1-19. doi: 10.1007/s00441-011-1196-4. Epub 2011 Jun 7. Cell Tissue Res. 2011. PMID: 21647561 Review.
Cited by
- Crystal Structures of the Human Doublecortin C- and N-terminal Domains in Complex with Specific Antibodies.
Burger D, Stihle M, Sharma A, Di Lello P, Benz J, D'Arcy B, Debulpaep M, Fry D, Huber W, Kremer T, Laeremans T, Matile H, Ross A, Rufer AC, Schoch G, Steinmetz MO, Steyaert J, Rudolph MG, Thoma R, Ruf A. Burger D, et al. J Biol Chem. 2016 Jul 29;291(31):16292-306. doi: 10.1074/jbc.M116.726547. Epub 2016 May 10. J Biol Chem. 2016. PMID: 27226599 Free PMC article. - Mammalian target of rapamycin signaling is a key regulator of the transit-amplifying progenitor pool in the adult and aging forebrain.
Paliouras GN, Hamilton LK, Aumont A, Joppé SE, Barnabé-Heider F, Fernandes KJ. Paliouras GN, et al. J Neurosci. 2012 Oct 24;32(43):15012-26. doi: 10.1523/JNEUROSCI.2248-12.2012. J Neurosci. 2012. PMID: 23100423 Free PMC article. - Steps towards standardized quantification of adult neurogenesis.
Zhao X, van Praag H. Zhao X, et al. Nat Commun. 2020 Aug 26;11(1):4275. doi: 10.1038/s41467-020-18046-y. Nat Commun. 2020. PMID: 32848155 Free PMC article. Review. - Adult hippocampal neurogenesis in natural populations of mammals.
Amrein I. Amrein I. Cold Spring Harb Perspect Biol. 2015 May 1;7(5):a021295. doi: 10.1101/cshperspect.a021295. Cold Spring Harb Perspect Biol. 2015. PMID: 25934014 Free PMC article. Review. - The impact of age on the physical and cellular properties of the human limbal stem cell niche.
Notara M, Shortt AJ, O'Callaghan AR, Daniels JT. Notara M, et al. Age (Dordr). 2013 Apr;35(2):289-300. doi: 10.1007/s11357-011-9359-5. Epub 2012 Jan 15. Age (Dordr). 2013. PMID: 22252434 Free PMC article.
References
- Altman J, Das GD. Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats. J Comp Neurol. 1965;124:319–335. - PubMed
- Kempermann G, Kuhn HG, Gage FH. More hippocampal neurons in adult mice living in an enriched environment. Nature. 1997;386:493–495. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous