Minireview: kisspeptin/neurokinin B/dynorphin (KNDy) cells of the arcuate nucleus: a central node in the control of gonadotropin-releasing hormone secretion - PubMed (original) (raw)
Review
. 2010 Aug;151(8):3479-89.
doi: 10.1210/en.2010-0022. Epub 2010 May 25.
Affiliations
- PMID: 20501670
- PMCID: PMC2940527
- DOI: 10.1210/en.2010-0022
Review
Minireview: kisspeptin/neurokinin B/dynorphin (KNDy) cells of the arcuate nucleus: a central node in the control of gonadotropin-releasing hormone secretion
Michael N Lehman et al. Endocrinology. 2010 Aug.
Abstract
Recently, a subset of neurons was identified in the arcuate nucleus of the hypothalamus that colocalize three neuropeptides, kisspeptin, neurokinin B, and dynorphin, each of which has been shown to play a critical role in the central control of reproduction. Growing evidence suggests that these neurons, abbreviated as the KNDy subpopulation, are strongly conserved across a range of species from rodents to humans and play a key role in the physiological regulation of GnRH neurons. KNDy cells are a major target for steroid hormones, form a reciprocally interconnected network, and have direct projections to GnRH cell bodies and terminals, features that position them well to convey steroid feedback control to GnRH neurons and potentially serve as a component of the GnRH pulse generator. In addition, recent work suggests that alterations in KNDy cell peptides may underlie neuroendocrine defects seen in clinical reproductive disorders such as polycystic ovarian syndrome. Taken together, this evidence suggests a key role for the KNDy subpopulation as a focal point in the control of reproductive function in health and disease.
Figures
Figure 1
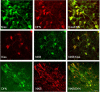
Fluorescent images showing immunocytochemical colocalization of kisspeptin (Kiss) and DYN, kisspeptin and NKB, and DYN and NKB in KNDy cells of the ovine ARC. Bar, 20 μm. (Modified from Refs. and .)
Figure 2
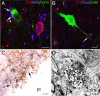
Evidence that KNDy cells directly contact GnRH cell bodies (A and B) and terminals in the median eminence (C and D) in the sheep. A and B, Confocal images (1-μm optical sections) of triple-labeled sections through the ovine MBH showing DYN/NKB-positive (A) and DYN/Kiss-positive (B) axon terminals in close contact with a GnRH soma (A) and dendrite (B), respectively. Note, in A, the presence of a DYN/NKB-positive KNDy cell body nearby. Bars in A and B, 10 μm. C and D, Light microscopic (C) and electron microscopic (D) evidence of direct contacts between DYN and GnRH-positive terminals in the median eminence of a luteal-phase ewe. The section shown in C was processed for dual-label immunoperoxidase detection of DYN (_blue_-black) and GnRH (brown) and shows close associations (arrows) between DYN and GnRH fibers. D is an electron micrograph of dual-labeled DYN/GnRH section through the median eminence, generated using a preembedding dual-immunoperoxidase technique (110). A terminal containing DYN-positive dense-core vesicles (arrowheads) is seen in direct contact to a GnRH terminal that contains large neurosecretory granules. pt, Pars tuberalis; ze, zona externa of the median eminence. Bars in C and D, 10 μm (C) and 2 μm (D).
Figure 3
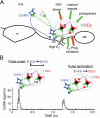
A, Schematic diagram showing potential regulatory signals conveyed by KNDy cells to GnRH neurons and the KNDy peptides (kisspeptin, green; NKB, magenta; DYN, red) currently implicated in each action. Note that in some cases (e.g. positive feedback actions of E2 and stress), two colors indicate that more than one KNDy peptide may be involved. In addition to direct contacts onto GnRH neurons in the POA and MBH, the potential for indirect influence via interneurons (gray cell) is also shown. MB, Mammillary bodies; me, median eminence; oc, optic chiasm; Prog, progesterone. B. Hypothetical model by which synchronous activity among KNDy cells may regulate GnRH pulse frequency (see text for details). GnRH pulse data was redrawn from Moenter et al. (85). Prog, Progesterone.
Similar articles
- Kisspeptin, neurokinin B, and dynorphin act in the arcuate nucleus to control activity of the GnRH pulse generator in ewes.
Goodman RL, Hileman SM, Nestor CC, Porter KL, Connors JM, Hardy SL, Millar RP, Cernea M, Coolen LM, Lehman MN. Goodman RL, et al. Endocrinology. 2013 Nov;154(11):4259-69. doi: 10.1210/en.2013-1331. Epub 2013 Aug 19. Endocrinology. 2013. PMID: 23959940 Free PMC article. - Prenatal testosterone excess decreases neurokinin 3 receptor immunoreactivity within the arcuate nucleus KNDy cell population.
Ahn T, Fergani C, Coolen LM, Padmanabhan V, Lehman MN. Ahn T, et al. J Neuroendocrinol. 2015 Feb;27(2):100-10. doi: 10.1111/jne.12244. J Neuroendocrinol. 2015. PMID: 25496429 Free PMC article. - The kisspeptin/neurokinin B/dynorphin (KNDy) cell population of the arcuate nucleus: sex differences and effects of prenatal testosterone in sheep.
Cheng G, Coolen LM, Padmanabhan V, Goodman RL, Lehman MN. Cheng G, et al. Endocrinology. 2010 Jan;151(1):301-11. doi: 10.1210/en.2009-0541. Epub 2009 Oct 30. Endocrinology. 2010. PMID: 19880810 Free PMC article. - Does the KNDy Model for the Control of Gonadotropin-Releasing Hormone Pulses Apply to Monkeys and Humans?
Lehman MN, He W, Coolen LM, Levine JE, Goodman RL. Lehman MN, et al. Semin Reprod Med. 2019 Mar;37(2):71-83. doi: 10.1055/s-0039-3400254. Epub 2019 Dec 17. Semin Reprod Med. 2019. PMID: 31847027 Free PMC article. Review. - Neuroendocrine control of gonadotropin-releasing hormone: Pulsatile and surge modes of secretion.
Goodman RL, Herbison AE, Lehman MN, Navarro VM. Goodman RL, et al. J Neuroendocrinol. 2022 May;34(5):e13094. doi: 10.1111/jne.13094. Epub 2022 Feb 2. J Neuroendocrinol. 2022. PMID: 35107859 Free PMC article. Review.
Cited by
- Neural mechanisms controlling seasonal reproduction: principles derived from the sheep model and its comparison with hamsters.
Weems PW, Goodman RL, Lehman MN. Weems PW, et al. Front Neuroendocrinol. 2015 Apr;37:43-51. doi: 10.1016/j.yfrne.2014.12.002. Epub 2015 Jan 9. Front Neuroendocrinol. 2015. PMID: 25582913 Free PMC article. Review. - Current and future applications of GnRH, kisspeptin and neurokinin B analogues.
Millar RP, Newton CL. Millar RP, et al. Nat Rev Endocrinol. 2013 Aug;9(8):451-66. doi: 10.1038/nrendo.2013.120. Epub 2013 Jul 2. Nat Rev Endocrinol. 2013. PMID: 23817290 Review. - Ovarian Suppression: Early Menopause, Late Effects.
Goldberg C, Greenberg MR, Noveihed A, Agrawal L, Omene C, Toppmeyer D, George MA. Goldberg C, et al. Curr Oncol Rep. 2024 May;26(5):427-438. doi: 10.1007/s11912-023-01491-5. Epub 2024 Jan 2. Curr Oncol Rep. 2024. PMID: 38305992 Review. - NKB signaling in the posterodorsal medial amygdala stimulates gonadotropin release in a kisspeptin-independent manner in female mice.
Fergani C, Leon S, Padilla SL, Verstegen AM, Palmiter RD, Navarro VM. Fergani C, et al. Elife. 2018 Dec 19;7:e40476. doi: 10.7554/eLife.40476. Elife. 2018. PMID: 30565563 Free PMC article. - Hypothalamic Kisspeptin Neurons: Integral Elements of the GnRH System.
Prashar V, Arora T, Singh R, Sharma A, Parkash J. Prashar V, et al. Reprod Sci. 2023 Mar;30(3):802-822. doi: 10.1007/s43032-022-01027-5. Epub 2022 Jul 7. Reprod Sci. 2023. PMID: 35799018 Review.
References
- Schally AV, Arimura A, Kastin AJ, Matsuo H, Baba Y, Redding TW, Nair RM, Debeljuk L, White WF 1971 Gonadotropin-releasing hormone: one polypeptide regulates secretion of luteinizing and follicle-stimulating hormones. Science 173:1036–1038 - PubMed
- Seminara SB, Messager S, Chatzidaki EE, Thresher RR, Acierno Jr JS, Shagoury JK, Bo-Abbas Y, Kuohung W, Schwinof KM, Hendrick AG, Zahn D, Dixon J, Kaiser UB, Slaugenhaupt SA, Gusella JF, O'Rahilly S, Carlton MB, Crowley Jr WF, Aparicio SA, Colledge WH 2003 The GPR54 gene as a regulator of puberty. N Engl J Med 349:1614–1627 - PubMed
- Plant TM 2006 The role of KiSS-1 in the regulation of puberty in higher primates. Eur J Endocrinol 155(Suppl 1):S11–S16 - PubMed
- Navarro VM, Castellano JM, García-Galiano D, Tena-Sempere M 2007 Neuroendocrine factors in the initiation of puberty: the emergent role of kisspeptin. Rev Endocr Metab Disord 8:11–20 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 86744/CAPMC/ CIHR/Canada
- P01 HD44232/HD/NICHD NIH HHS/United States
- R01 HD39916/HD/NICHD NIH HHS/United States
- P01 HD044232/HD/NICHD NIH HHS/United States
- R01 HD039916/HD/NICHD NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources