Naturally occurring human urinary peptides for use in diagnosis of chronic kidney disease - PubMed (original) (raw)
. 2010 Nov;9(11):2424-37.
doi: 10.1074/mcp.M110.001917. Epub 2010 Jul 8.
Petra Zürbig, Angel Argilés, Hartwig W Bauer, Georg Behrens, Joshua J Coon, Mohammed Dakna, Stéphane Decramer, Christian Delles, Anna F Dominiczak, Jochen H H Ehrich, Frank Eitner, Danilo Fliser, Moritz Frommberger, Arnold Ganser, Mark A Girolami, Igor Golovko, Wilfried Gwinner, Marion Haubitz, Stefan Herget-Rosenthal, Joachim Jankowski, Holger Jahn, George Jerums, Bruce A Julian, Markus Kellmann, Volker Kliem, Walter Kolch, Andrzej S Krolewski, Mario Luppi, Ziad Massy, Michael Melter, Christian Neusüss, Jan Novak, Karlheinz Peter, Kasper Rossing, Harald Rupprecht, Joost P Schanstra, Eric Schiffer, Jens-Uwe Stolzenburg, Lise Tarnow, Dan Theodorescu, Visith Thongboonkerd, Raymond Vanholder, Eva M Weissinger, Harald Mischak, Philippe Schmitt-Kopplin
Affiliations
- PMID: 20616184
- PMCID: PMC2984241
- DOI: 10.1074/mcp.M110.001917
Naturally occurring human urinary peptides for use in diagnosis of chronic kidney disease
David M Good et al. Mol Cell Proteomics. 2010 Nov.
Abstract
Because of its availability, ease of collection, and correlation with physiology and pathology, urine is an attractive source for clinical proteomics/peptidomics. However, the lack of comparable data sets from large cohorts has greatly hindered the development of clinical proteomics. Here, we report the establishment of a reproducible, high resolution method for peptidome analysis of naturally occurring human urinary peptides and proteins, ranging from 800 to 17,000 Da, using samples from 3,600 individuals analyzed by capillary electrophoresis coupled to MS. All processed data were deposited in an Structured Query Language (SQL) database. This database currently contains 5,010 relevant unique urinary peptides that serve as a pool of potential classifiers for diagnosis and monitoring of various diseases. As an example, by using this source of information, we were able to define urinary peptide biomarkers for chronic kidney diseases, allowing diagnosis of these diseases with high accuracy. Application of the chronic kidney disease-specific biomarker set to an independent test cohort in the subsequent replication phase resulted in 85.5% sensitivity and 100% specificity. These results indicate the potential usefulness of capillary electrophoresis coupled to MS for clinical applications in the analysis of naturally occurring urinary peptides.
Figures
Fig. 1.
Peptide patterns distinguishing patients with CKD from HC. The compiled data sets of urine samples from 230 patients with CKD (left panel) and 379 healthy control subjects (right panel) included in the training set are shown. Normalized molecular mass (y axis) is plotted against normalized CE migration time (x axis). The mean signal intensity is represented in three-dimensional depiction.
Fig. 2.
ROC curves for classification of patient cohorts with “CKD pattern.” ROC analysis for CKD diagnosis of the training set and the test set after unblinding is shown. AUC, area under the curve.
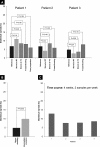
Fig. 3.
Analytical variability of CE-MS-based CKD platform. A, statistical spread of temperature stability, freeze/thaw stability, and postpreparation stability are shown. Reproducibility and intermediate precision (B) and time course (C) are plotted against their statistical spread. p values are depicted in boxes.
References
- Alebiosu C. O., Ayodele O. E. (2005) The global burden of chronic kidney disease and the way forward. Ethn. Dis. 15, 418–423 - PubMed
- Good D. M., Thongboonkerd V., Novak J., Bascands J. L., Schanstra J. P., Coon J. J., Dominiczak A., Mischak H. (2007) Body fluid proteomics for biomarker discovery: lessons from the past hold the key to success in the future. J. Proteome Res. 6, 4549–4555 - PubMed
- Kolch W., Neusüss C., Pelzing M., Mischak H. (2005) Capillary electrophoresis-mass spectrometry as a powerful tool in clinical diagnosis and biomarker discovery. Mass Spectrom. Rev. 24, 959–977 - PubMed
- González-Buitrago J. M., Ferreira L., Lorenzo I. (2007) Urinary proteomics. Clin. Chim. Acta 375, 49–56 - PubMed
- Fliser D., Novak J., Thongboonkerd V., Argilés A., Jankowski V., Girolami M. A., Jankowski J., Mischak H. (2007) Advances in urinary proteome analysis and biomarker discovery. J. Am. Soc. Nephrol. 18, 1057–1071 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- DK082753/DK/NIDDK NIH HHS/United States
- R21 DK075868/DK/NIDDK NIH HHS/United States
- DK080301/DK/NIDDK NIH HHS/United States
- R21 DK080301/DK/NIDDK NIH HHS/United States
- R01 DK078244/DK/NIDDK NIH HHS/United States
- RG/02/012/BHF_/British Heart Foundation/United Kingdom
- T32 GM008349/GM/NIGMS NIH HHS/United States
- 29240/WT_/Wellcome Trust/United Kingdom
- R56 DK078244/DK/NIDDK NIH HHS/United States
- DK075868/DK/NIDDK NIH HHS/United States
- R01 DK067638/DK/NIDDK NIH HHS/United States
- G0401466/MRC_/Medical Research Council/United Kingdom
- DK061525/DK/NIDDK NIH HHS/United States
- R01 DK082753/DK/NIDDK NIH HHS/United States
- DK078244/DK/NIDDK NIH HHS/United States
- P01 DK061525/DK/NIDDK NIH HHS/United States
- DK067638/DK/NIDDK NIH HHS/United States
- 5T32GM08349/GM/NIGMS NIH HHS/United States
- R01 DK041526/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials