Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis - PubMed (original) (raw)
. 2011 Mar 15;108 Suppl 1(Suppl 1):4615-22.
doi: 10.1073/pnas.1000082107. Epub 2010 Jul 26.
Affiliations
- PMID: 20660719
- PMCID: PMC3063590
- DOI: 10.1073/pnas.1000082107
Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis
Yun Kyung Lee et al. Proc Natl Acad Sci U S A. 2011.
Abstract
Although the effects of commensal bacteria on intestinal immune development seem to be profound, it remains speculative whether the gut microbiota influences extraintestinal biological functions. Multiple sclerosis (MS) is a devastating autoimmune disease leading to progressive deterioration of neurological function. Although the cause of MS is unknown, microorganisms seem to be important for the onset and/or progression of disease. However, it is unclear how microbial colonization, either symbiotic or infectious, affects autoimmunity. Herein, we investigate a role for the microbiota during the induction of experimental autoimmune encephalomyelitis (EAE), an animal model for MS. Mice maintained under germ-free conditions develop significantly attenuated EAE compared with conventionally colonized mice. Germ-free animals, induced for EAE, produce lower levels of the proinflammatory cytokines IFN-γ and IL-17A in both the intestine and spinal cord but display a reciprocal increase in CD4(+)CD25(+)Foxp3(+) regulatory T cells (Tregs). Mechanistically, we show that gut dendritic cells from germ-free animals are reduced in the ability to stimulate proinflammatory T cell responses. Intestinal colonization with segmented filamentous bacteria (SFB) is known to promote IL-17 production in the gut; here, we show that SFBs also induced IL-17A-producing CD4(+) T cells (Th17) in the CNS. Remarkably, germ-free animals harboring SFBs alone developed EAE, showing that gut bacteria can affect neurologic inflammation. These findings reveal that the intestinal microbiota profoundly impacts the balance between pro- and antiinflammatory immune responses during EAE and suggest that modulation of gut bacteria may provide therapeutic targets for extraintestinal inflammatory diseases such as MS.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Fig. 1.
GF mice develop less severe EAE than SPF mice. (A) Clinical EAE scores of SPF (dark symbols) and GF (light symbols) mice immunized with MOG peptide in CFA plus pertussis toxin. Data are representative of three independent experiments. Symbols represent the mean ± SD at each time point. (B) Proportions of SPF and GF mice that develop various degrees of EAE. Results are combined from four independent experiments, with a total number of animals per group of 20 for SPF and 22 for GF mice. (C) H&E (Upper) and myelin basic protein (Lower) staining of the spinal cord of SPF and GF mice at the onset of EAE on day 12. Each panel is representative of tissue analyzed from at least four mice. (D) EAE score of conventional Rag−/− recipient mice (n = 4) that were reconstituted with in vitro primed CD4+ cells from SPF (black symbols) or GF (gray symbols) mice immunized with MOG/CFA and pertussis toxin. Data are representative of three independent experiments. Symbols represent the mean ± SD at each time point.
Fig. 2.
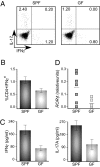
Inflammation is attenuated in GF mice on EAE induction. (A) Lymphocytes were harvested from draining LN of treated SPF and GF mice at 8 d after immunization with MOG/CFA. Intracellular staining of CD4+ T cell for IFNγ and IL-17A after 3 d in vitro culture with MOG peptide, restimulated with phorbol 12-myristate 13-acetate (PMA)/ionomycin for the last 5 h. Numbers in each quadrant indicate percentage of cytokine-positive CD4+ T cells. (B) Mean ± SD of the CD4+IFNγ+ T cell subsets from A. Data are representative of three independent experiments with at least four mice per group. (C) Real-time PCR of the transcription factor RORγt in cells isolated by Percoll gradient from spinal cords of SPF and GF mice at the onset of disease (day 8). Each symbol represents a single mouse. (D) IFNγ and IL-17A cytokine ELISA from cells harvested from draining LN of SPF and GF mice 8 d after immunization with MOG/CFA and cultured for 3 d in vitro with MOG peptide. Data are representative of three independent experiments, with mean ± SD of samples run in triplicate.
Fig. 3.
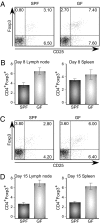
Increase in CD25+Foxp3+ Treg cells after induction of EAE in GF mice. Intracellular staining of CD4+CD25+Foxp3+ T cells cultured for 3 d with MOG peptide, restimulated with PMA/ionomycin for the last 5 h. Lymphocytes were harvested from draining lymph nodes (LN) and spleens of SPF and GF mice at 8 d p.i. (A and B) or 15 d p.i. (C and D) after i.v. immunization with MOG/CFA. In A and C, numbers in each quadrant indicate percentage of positive cells. In B and D, results are shown for mean ± SD of the CD4+ subsets of the cells from draining LN and spleen on day 8 and day 15 postimmunization. Data are representative of three independent experiments with at least four mice per group.
Fig. 4.
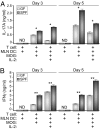
DCs from GF animals are defective in inducing Th17 and Th1 responses from CD4+ T cells. Purified CD4+ T cells from MOG-Tg mice were cultured with MLN DCs from SPF or GF mice in the absence or presence of MOG peptide and IL-2. At day 3 and day 5 of culture, IL-17A (A) and IFNγ (B) cytokines were measured by ELISA. Data are representative of two independent experiments, with mean ± SD of samples run in triplicate. *P < 0.05; **P < 0.005.
Fig. 5.
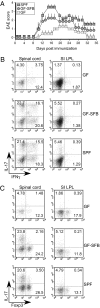
SFBs promote proinflammatory T cell responses outside the gut during EAE. (A) Clinical EAE scores of SPF, GF, and GF-SFB colonized mice immunized with MOG-peptide in CFA plus pertussis toxin. GF mice were colonized with SFB for 3 wk before MOG/CFA immunization (GF-SFB). Data are representative of two independent experiments with at least four mice per group. Symbols represent the mean ± SD at each time point. (B) Representative FC plots of IL-17A– and IFNγ-producing CD4+ T cells from LPLs of spinal cords and small intestines of SPF, GF, and GF-SFB colonized mice at day 15 after EAE induction. Numbers in each quadrant indicate percentage of cytokine-positive CD4+ T cells. Data are representative of two independent experiments with at least four mice per group. (C) Representative FC plots of IL-17A– and Foxp3-producing CD4+ T cells from LPLs of spinal cords and small intestines of SPF, GF, and GF-SFB colonized mice at day 15 after EAE induction. Numbers in each quadrant indicate percentage of positive CD4+ T cells. Data are representative of two independent experiments with at least four mice per group.
Similar articles
- Human Commensal Prevotella histicola Ameliorates Disease as Effectively as Interferon-Beta in the Experimental Autoimmune Encephalomyelitis.
Shahi SK, Jensen SN, Murra AC, Tang N, Guo H, Gibson-Corley KN, Zhang J, Karandikar NJ, Murray JA, Mangalam AK. Shahi SK, et al. Front Immunol. 2020 Dec 11;11:578648. doi: 10.3389/fimmu.2020.578648. eCollection 2020. Front Immunol. 2020. PMID: 33362764 Free PMC article. - 1,25-dihydroxyvitamin D3 -induced dendritic cells suppress experimental autoimmune encephalomyelitis by increasing proportions of the regulatory lymphocytes and reducing T helper type 1 and type 17 cells.
Xie Z, Chen J, Zheng C, Wu J, Cheng Y, Zhu S, Lin C, Cao Q, Zhu J, Jin T. Xie Z, et al. Immunology. 2017 Nov;152(3):414-424. doi: 10.1111/imm.12776. Epub 2017 Jul 10. Immunology. 2017. PMID: 28617989 Free PMC article. - Therapeutic Effect of Ginsenoside Rd on Experimental Autoimmune Encephalomyelitis Model Mice: Regulation of Inflammation and Treg/Th17 Cell Balance.
Jin B, Zhang C, Geng Y, Liu M. Jin B, et al. Mediators Inflamm. 2020 Dec 17;2020:8827527. doi: 10.1155/2020/8827527. eCollection 2020. Mediators Inflamm. 2020. PMID: 33380901 Free PMC article. - Immunoregulation by the gut microbiota.
Nishio J, Honda K. Nishio J, et al. Cell Mol Life Sci. 2012 Nov;69(21):3635-50. doi: 10.1007/s00018-012-0993-6. Epub 2012 Apr 22. Cell Mol Life Sci. 2012. PMID: 22527722 Free PMC article. Review. - The Gut-Brain Axis in Multiple Sclerosis. Is Its Dysfunction a Pathological Trigger or a Consequence of the Disease?
Parodi B, Kerlero de Rosbo N. Parodi B, et al. Front Immunol. 2021 Sep 21;12:718220. doi: 10.3389/fimmu.2021.718220. eCollection 2021. Front Immunol. 2021. PMID: 34621267 Free PMC article. Review.
Cited by
- The brain-gut axis: communication mechanisms and the role of the microbiome as a neuroprotective factor in the development of neurodegenerative diseases: A literature overview.
Białoń MN, Górka DHNOZD, Górka MM. Białoń MN, et al. AIMS Neurosci. 2024 Aug 28;11(3):289-311. doi: 10.3934/Neuroscience.2024019. eCollection 2024. AIMS Neurosci. 2024. PMID: 39431278 Free PMC article. Review. - Anti-Inflammatory and Immunomodulatory Effects of Probiotics in Gut Inflammation: A Door to the Body.
Cristofori F, Dargenio VN, Dargenio C, Miniello VL, Barone M, Francavilla R. Cristofori F, et al. Front Immunol. 2021 Feb 26;12:578386. doi: 10.3389/fimmu.2021.578386. eCollection 2021. Front Immunol. 2021. PMID: 33717063 Free PMC article. Review. - Type 1 diabetes: translating mechanistic observations into effective clinical outcomes.
Herold KC, Vignali DA, Cooke A, Bluestone JA. Herold KC, et al. Nat Rev Immunol. 2013 Apr;13(4):243-56. doi: 10.1038/nri3422. Nat Rev Immunol. 2013. PMID: 23524461 Free PMC article. Review. - Diet, Microbiota and Brain Health: Unraveling the Network Intersecting Metabolism and Neurodegeneration.
Gentile F, Doneddu PE, Riva N, Nobile-Orazio E, Quattrini A. Gentile F, et al. Int J Mol Sci. 2020 Oct 10;21(20):7471. doi: 10.3390/ijms21207471. Int J Mol Sci. 2020. PMID: 33050475 Free PMC article. Review. - Psychological Stress, Intestinal Barrier Dysfunctions, and Autoimmune Disorders: An Overview.
Ilchmann-Diounou H, Menard S. Ilchmann-Diounou H, et al. Front Immunol. 2020 Aug 25;11:1823. doi: 10.3389/fimmu.2020.01823. eCollection 2020. Front Immunol. 2020. PMID: 32983091 Free PMC article. Review.
References
- Bhat R, Steinman L. Innate and adaptive autoimmunity directed to the central nervous system. Neuron. 2009;64:123–132. - PubMed
- Stromnes IM, Goverman JM. Active induction of experimental allergic encephalomyelitis. Nat Protoc. 2006;1:1810–1819. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials