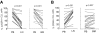The lymph node microenvironment promotes B-cell receptor signaling, NF-kappaB activation, and tumor proliferation in chronic lymphocytic leukemia - PubMed (original) (raw)
Clinical Trial
. 2011 Jan 13;117(2):563-74.
doi: 10.1182/blood-2010-05-284984. Epub 2010 Oct 12.
Patricia Pérez-Galán, Delong Liu, Angélique Biancotto, Stefania Pittaluga, Berengere Vire, Federica Gibellini, Ndegwa Njuguna, Elinor Lee, Lawrence Stennett, Nalini Raghavachari, Poching Liu, J Philip McCoy, Mark Raffeld, Maryalice Stetler-Stevenson, Constance Yuan, Richard Sherry, Diane C Arthur, Irina Maric, Therese White, Gerald E Marti, Peter Munson, Wyndham H Wilson, Adrian Wiestner
Affiliations
- PMID: 20940416
- PMCID: PMC3031480
- DOI: 10.1182/blood-2010-05-284984
Clinical Trial
The lymph node microenvironment promotes B-cell receptor signaling, NF-kappaB activation, and tumor proliferation in chronic lymphocytic leukemia
Yair Herishanu et al. Blood. 2011.
Abstract
Chronic lymphocytic leukemia (CLL), an incurable malignancy of mature B lymphocytes, involves blood, bone marrow, and secondary lymphoid organs such as the lymph nodes (LN). A role of the tissue microenvironment in the pathogenesis of CLL is hypothesized based on in vitro observations, but its contribution in vivo remains ill-defined. To elucidate the effects of tumor-host interactions in vivo, we purified tumor cells from 24 treatment-naive patients. Samples were obtained concurrently from blood, bone marrow, and/or LN and analyzed by gene expression profiling. We identified the LN as a key site in CLL pathogenesis. CLL cells in the LN showed up-regulation of gene signatures, indicating B-cell receptor (BCR) and nuclear factor-κB activation. Consistent with antigen-dependent BCR signaling and canonical nuclear factor-κB activation, we detected phosphorylation of SYK and IκBα, respectively. Expression of BCR target genes was stronger in clinically more aggressive CLL, indicating more effective BCR signaling in this subtype in vivo. Tumor proliferation, quantified by the expression of the E2F and c-MYC target genes and verified with Ki67 staining by flow cytometry, was highest in the LN and was correlated with clinical disease progression. These data identify the disruption of tumor microenvironment interactions and the inhibition of BCR signaling as promising therapeutic strategies in CLL. This study is registered at http://clinicaltrials.gov as NCT00019370.
Figures
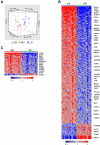
Figure 1
The tissue microenvironment affects the tumor biology of CLL cells in vivo. (A) Gene expression analysis of fresh CLL tumor cells derived from PB, BM, and LN in treatment-naive patients in whom all 3 sites were evaluable (n = 12). Principal component analysis of gene expression normalized for patient effect (see “Microarray hybridization, data processing, and analysis”) is shown. (B) Heat map of 151 genes differentially expressed in purified CLL cells obtained simultaneously from LN and PB in 17 patients (> 2-fold change, FDR < 20%). Patient samples are arranged in columns keeping the same order in the LN and PB groups. Gene expression is median-centered and scaled as indicated. Gene symbols highlight select genes. (C) Heat map of 26 differentially expressed genes in purified CLL cells isolated from BM aspirates compared with cells simultaneously obtained from the PB in 19 patients (> 2-fold change, FDR < 20%).
Figure 2
Dynamic changes in CXCR4 and CD69 expression on CLL cells in different anatomic compartments. (A) CXCR4 was quantified by flow cytometry on CD19+-gated cells and expressed as the percentage of cells expressing CXCR4 above isotype control. Samples obtained simultaneously from the same patient are connected by a line. Mononuclear cells were obtained by density gradient centrifugation, maintained on ice, and analyzed within 24 hours. Normal B cells constituted less than 1% of CD19+ cells. Comparison was by paired t test. (B) CD69 was quantified by flow cytometry on CD19+-gated cells and expressed as the percentage of cells expressing CD69 above isotype control. Sample collection and analysis were as in panel A.
Figure 3
Activation of the B-cell receptor on CLL cells induces a characteristic gene expression signature and phosphorylation of SYK. (A) Gene expression changes in UM-CLL (n = 4) and M-CLL (n = 4) samples 6 hours after in vitro IgM cross-linking. Sixty-one genes showing a > 2-fold change between unstimulated and stimulated cells (P < .001) were identified and constitute a “CLL-BCR gene signature.” Gene expression changes in response to BCR engagement is depicted in a heat map according to the scale shown. Genes are sorted top to bottom for the ratio between the average up-regulation in UM-CLL compared with M-CLL cells. (B) Phosphorylation of SYK (p-SYK) after in vitro IgM cross-linking was assessed by flow cytometry in cells gated on CD3−SYK+IgM+ at the indicated time points. (C) Mean and SD of the percentage of pSYK-positive cells in CLL subtypes (UM-CLL n = 14, M-CLL n = 13) and normal B cells (control n = 11) are shown. Differences between groups was assessed by Student t test (P = .013 for UM-CLL vs M-CLL at 45 minutes; all other comparisons were not significant).
Figure 4
BCR activation in LN-resident CLL cells. (A) Genes in the CLL-BCR signature that were most significantly enriched in the LN as identified by GSEA (leading-edge genes) are depicted in a heat map for the 12 patients having contributed cells from all 3 compartments. (B) The BCR score was computed as the average of the mRNA expression level of the leading edge genes for each sample. Shown is the ratio of the BCR score in BM (n = 19) or LN (n = 17) relative to the score of the matched PB sample. Comparison between BM and LN was by Student t test. The sample with exceptionally high score in BM was obtained from CLL_C10. (C, D) Relative gene expression changes of BCR-regulated genes between UM-CLL and M-CLL. Each gene is represented by a bar, select genes are highlighted. (C) Ratio of relative gene expression change after IgM cross-linking between UM-CLL (n = 4) and M-CLL (n = 4). Bars represent 61 genes, as described in Figure 3A. The fold up-regulation of each gene was averaged for each subtype and the ratio between UM-CLL and M-CLL is shown on a log2 scale. (D) Ratio of relative gene expression change in UM-CLL (n = 12) and M-CLL (n = 5) between the LN- and matched PB-derived cells. Bars represent all 61 genes as in panel C. The fold up-regulation of each gene in the LN-derived cells compared with the matched PB sample was averaged within each subtype, and the ratio between UM-CLL and M-CLL is shown on a log2 scale. (E) Phosphorylation of SYK was assessed by flow cytometry in CD3− cells from the indicated anatomical site. Shown are 2 representative patients of a total of 6 analyzed; in 5 patients, staining for pSYK was increased in LN- compared with PB-derived cells (median 7-fold increase).
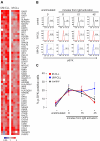
Figure 5
NF-κB is activated through the canonical pathway in LN-derived CLL cells. (A) Genes in the NF-κB signature that were most significantly enriched in the LN as identified by GSEA (leading edge genes) are depicted in a heat map for the 12 patients having contributed cells from all 3 compartments. (B) The NF-κB score was computed as the average of the mRNA expression level of the leading edge genes for each sample. Shown is the ratio of the BCR score in BM (n = 19) or LN (n = 17) relative to the score of the matched PB sample. Comparison between BM and LN was by Student t test. The sample with the exceptionally high score in the BM was obtained from CLL_C10. (C) Western blot of nuclear lysates from purified CLL cells; JUNB expression was normalized to TBP and the ratio between LN- and PB-derived cells is shown. Western blots of cytoplasmic protein fractions from purified CLL cells were probed for phosphorylated IκBα (p-IκBα, Ser 32-36; D) and for total IκBα (E). IκBα and p-IκBα expression quantified by densitometry was normalized to γ-tubulin and the ratio between LN- and PB-derived cells is shown. (F) Pearson correlation between IκBα expression and the NF-κB gene expression score.
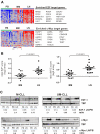
Figure 6
Tumor proliferation and c-MYC activation in the tissue microenvironment. (A) Genes of the E2F and c-MYC signatures that were most significantly enriched in the LN as identified by GSEA (leading-edge genes) are depicted in a heat map for the 12 patients having contributed cells from all 3 compartments. For E2F only genes that were at least 1.5-fold more highly expressed in LN than PB are shown. (B) The E2F and c-MYC scores were computed individually as the average of the mRNA expression level of the leading-edge genes for each sample. Shown is the ratio of the E2F and c-MYC scores in BM (n = 19) or LN (n = 17) relative to the score of the matched PB sample. Comparison between BM and LN was by Student t test. (C) Western blot of nuclear protein extracts from purified CLL cells were probed with anti-E2F1 or anti–c-MYC antibody, quantified by densitometry, and normalized to TBP. The ratios of LN- to PB-derived cells for E2F1 and c-MYC protein expression are indicated.
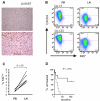
Figure 7
Tumor proliferation in the LN correlates with disease progression. (A) Magnification (×200) of LN biopsies of 2 representative CLL samples: top panel, preferential Ki67 staining in proliferation centers; bottom panel, diffuse Ki67 positivity. (B) Ki67 expression was assessed by flow cytometry in CD3− cells from the indicated anatomical sites. Shown are 2 representative patients of a total of 6 analyzed. Cutoffs in each sample were chosen so that PB cells were around 1% Ki67+ cells. (C) Ki67 expression in CD3− cells from the indicated anatomic sites. Statistical comparison was by paired Student t test. (D) Kaplan-Meier analysis of time to treatment in all patients with LN biopsies (n = 17) based on E2F scores: dashed line, scores above the median; solid line, scores below the median.
Comment in
- CLL microenvironment: macro important.
Friedberg JW. Friedberg JW. Blood. 2011 Jan 13;117(2):377-8. doi: 10.1182/blood-2010-11-314468. Blood. 2011. PMID: 21233321 No abstract available.
Similar articles
- Chronic lymphocytic leukemia cells in a lymph node microenvironment depict molecular signature associated with an aggressive disease.
Mittal AK, Chaturvedi NK, Rai KJ, Gilling-Cutucache CE, Nordgren TM, Moragues M, Lu R, Opavsky R, Bociek GR, Weisenburger DD, Iqbal J, Joshi SS. Mittal AK, et al. Mol Med. 2014 Jul 15;20(1):290-301. doi: 10.2119/molmed.2012.00303. Mol Med. 2014. PMID: 24800836 Free PMC article. - TLR Signaling Is Activated in Lymph Node-Resident CLL Cells and Is Only Partially Inhibited by Ibrutinib.
Dadashian EL, McAuley EM, Liu D, Shaffer AL 3rd, Young RM, Iyer JR, Kruhlak MJ, Staudt LM, Wiestner A, Herman SEM. Dadashian EL, et al. Cancer Res. 2019 Jan 15;79(2):360-371. doi: 10.1158/0008-5472.CAN-18-0781. Epub 2018 Nov 29. Cancer Res. 2019. PMID: 30498085 Free PMC article. - Modeling tumor-host interactions of chronic lymphocytic leukemia in xenografted mice to study tumor biology and evaluate targeted therapy.
Herman SE, Sun X, McAuley EM, Hsieh MM, Pittaluga S, Raffeld M, Liu D, Keyvanfar K, Chapman CM, Chen J, Buggy JJ, Aue G, Tisdale JF, Pérez-Galán P, Wiestner A. Herman SE, et al. Leukemia. 2013 Dec;27(12):2311-21. doi: 10.1038/leu.2013.131. Epub 2013 Apr 26. Leukemia. 2013. PMID: 23619564 Free PMC article. - Microenvironment interactions and B-cell receptor signaling in Chronic Lymphocytic Leukemia: Implications for disease pathogenesis and treatment.
Ten Hacken E, Burger JA. Ten Hacken E, et al. Biochim Biophys Acta. 2016 Mar;1863(3):401-413. doi: 10.1016/j.bbamcr.2015.07.009. Epub 2015 Jul 17. Biochim Biophys Acta. 2016. PMID: 26193078 Free PMC article. Review. - NF-κB activation in chronic lymphocytic leukemia: A point of convergence of external triggers and intrinsic lesions.
Mansouri L, Papakonstantinou N, Ntoufa S, Stamatopoulos K, Rosenquist R. Mansouri L, et al. Semin Cancer Biol. 2016 Aug;39:40-8. doi: 10.1016/j.semcancer.2016.07.005. Epub 2016 Aug 1. Semin Cancer Biol. 2016. PMID: 27491692 Review.
Cited by
- Old and New Facts and Speculations on the Role of the B Cell Receptor in the Origin of Chronic Lymphocytic Leukemia.
Bagnara D, Mazzarello AN, Ghiotto F, Colombo M, Cutrona G, Fais F, Ferrarini M. Bagnara D, et al. Int J Mol Sci. 2022 Nov 17;23(22):14249. doi: 10.3390/ijms232214249. Int J Mol Sci. 2022. PMID: 36430731 Free PMC article. Review. - Pathogenic role of B-cell receptor signaling and canonical NF-κB activation in mantle cell lymphoma.
Saba NS, Liu D, Herman SE, Underbayev C, Tian X, Behrend D, Weniger MA, Skarzynski M, Gyamfi J, Fontan L, Melnick A, Grant C, Roschewski M, Navarro A, Beà S, Pittaluga S, Dunleavy K, Wilson WH, Wiestner A. Saba NS, et al. Blood. 2016 Jul 7;128(1):82-92. doi: 10.1182/blood-2015-11-681460. Epub 2016 Apr 28. Blood. 2016. PMID: 27127301 Free PMC article. Clinical Trial. - The Role of CD44 in the Pathophysiology of Chronic Lymphocytic Leukemia.
Gutjahr JC, Greil R, Hartmann TN. Gutjahr JC, et al. Front Immunol. 2015 Apr 20;6:177. doi: 10.3389/fimmu.2015.00177. eCollection 2015. Front Immunol. 2015. PMID: 25941526 Free PMC article. Review. - Targeting Nuclear NOTCH2 by Gliotoxin Recovers a Tumor-Suppressor NOTCH3 Activity in CLL.
Hubmann R, Schnabl S, Araghi M, Schmidl C, Rendeiro AF, Hilgarth M, Demirtas D, Ali F, Staber PB, Valent P, Zielinski C, Jäger U, Shehata M. Hubmann R, et al. Cells. 2020 Jun 18;9(6):1484. doi: 10.3390/cells9061484. Cells. 2020. PMID: 32570839 Free PMC article. - Krüppel-like factor 4 (KLF4) inactivation in chronic lymphocytic leukemia correlates with promoter DNA-methylation and can be reversed by inhibition of NOTCH signaling.
Filarsky K, Garding A, Becker N, Wolf C, Zucknick M, Claus R, Weichenhan D, Plass C, Döhner H, Stilgenbauer S, Lichter P, Mertens D. Filarsky K, et al. Haematologica. 2016 Jun;101(6):e249-53. doi: 10.3324/haematol.2015.138172. Epub 2016 Apr 14. Haematologica. 2016. PMID: 27081174 Free PMC article. No abstract available.
References
- Chiorazzi N, Rai KR, Ferrarini M. Chronic lymphocytic leukemia. N Engl J Med. 2005;352(8):804–815. - PubMed
- Wiestner A, Rosenwald A, Barry TS, et al. ZAP-70 expression identifies a chronic lymphocytic leukemia subtype with unmutated immunoglobulin genes, inferior clinical outcome, and distinct gene expression profile. Blood. 2003;101(12):4944–4951. - PubMed
- Crespo M, Bosch F, Villamor N, et al. ZAP-70 expression as a surrogate for immunoglobulin-variable-region mutations in chronic lymphocytic leukemia. N Engl J Med. 2003;348(18):1764–1775. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous