The development of female sexual behavior requires prepubertal estradiol - PubMed (original) (raw)
The development of female sexual behavior requires prepubertal estradiol
Olivier Brock et al. J Neurosci. 2011.
Abstract
The classic view of brain and behavioral sexual differentiation holds that the neural mechanisms controlling sexual behavior in female rodents develop in the absence of ovarian sex hormone actions. However, in a previous study, female aromatase knock-out (ArKO) mice, which cannot convert testosterone to estradiol, showed deficient male-oriented partner preference and lordosis behaviors in response to adult ovarian hormones, raising the possibility that estradiol may contribute to the development of these female sexual behaviors. In the present experiments, administering estradiol prepubertally [between postnatal day 15 (P15) and P25] significantly enhanced the ability of ArKO female mice to display lordosis behavior in response to ovarian hormones administered later in adulthood, whereas treatment with estradiol over an earlier postnatal period (P5-P15) had no such effect. Treatment of ArKO females with estradiol between P15 and P25 also rescued their later preference to approach distal cues from an intact male over an estrous female. ArKO females also displayed significantly less female-directed (male-typical) mounting behavior than wild-type control females when treated with testosterone in adulthood. Prepubertal estradiol treatment failed to reverse this deficit in ArKO females, whereas earlier postnatal estradiol augmented later mounting in both genotypes. Our results provide new evidence for an organizing role of prepubertal estradiol in the development of neural mechanisms that control female-typical sexual behavior.
Figures
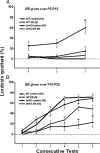
Figure 1.
Effect of early postnatal (A; P5–P15) or prepubertal (B; P15–P25) EB treatment of WT and ArKO female mice on the expression of lordosis behavior in adulthood following ovariectomy and treatment with ovarian hormones. Mean ± SEM lordosis quotients over consecutive tests in WT control females, WT females treated with EB, ArKO control females, and ArKO females treated with EB are shown. A, Values for WT control females on test 3 were significantly higher than values for the other 3 groups of female mice (*p < 0.05) by post hoc tests. B, Values for the ArKO control females on tests 5 and 6 were significantly lower than values for the other 3 groups of female mice (*p < 0.05) by post hoc tests. The number of female mice in each group is given in parentheses.
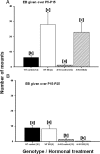
Figure 2.
Effect of early postnatal (A; P5–P15) or prepubertal (B; P15–P25) EB treatment of WT and ArKO female mice on the later display of female-directed mounting behavior in adulthood following ovariectomy and treatment with testosterone. Mean ± SEM numbers of mounts displayed by WT control females, WT females treated with EB, ArKO control females, and ArKO females treated with EB are shown. Means with different superscript letters are significantly different from each other by post hoc comparisons (p < 0.05). The number of female mice in each group is given in parentheses.
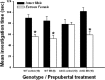
Figure 3.
Effect of prepubertal (P15–P25) EB treatment of WT and ArKO females on mate preference in adulthood following ovariectomy and treatment with ovarian hormones. Shown are mean ± SEM times spent by WT control females, WT females treated prepubertally with EB, ArKO control females, and ArKO females treated prepubertally with EB investigating the compartment containing an intact male versus an estrous female. *p < 0.05, post hoc comparisons of time spent investigating the two types of stimulus animals. The number of female mice in each group is given in parentheses.
Similar articles
- The aromatase knock-out mouse provides new evidence that estradiol is required during development in the female for the expression of sociosexual behaviors in adulthood.
Bakker J, Honda S, Harada N, Balthazart J. Bakker J, et al. J Neurosci. 2002 Oct 15;22(20):9104-12. doi: 10.1523/JNEUROSCI.22-20-09104.2002. J Neurosci. 2002. PMID: 12388618 Free PMC article. - Sexual partner preference requires a functional aromatase (cyp19) gene in male mice.
Bakker J, Honda S, Harada N, Balthazart J. Bakker J, et al. Horm Behav. 2002 Sep;42(2):158-71. doi: 10.1006/hbeh.2002.1805. Horm Behav. 2002. PMID: 12367569 - Attraction thresholds and sex discrimination of urinary odorants in male and female aromatase knockout (ArKO) mice.
Pierman S, Douhard Q, Balthazart J, Baum MJ, Bakker J. Pierman S, et al. Horm Behav. 2006 Jan;49(1):96-104. doi: 10.1016/j.yhbeh.2005.05.007. Epub 2005 Jun 15. Horm Behav. 2006. PMID: 15961088 - The aromatase knockout (ArKO) mouse provides new evidence that estrogens are required for the development of the female brain.
Bakker J, Honda S, Harada N, Balthazart J. Bakker J, et al. Ann N Y Acad Sci. 2003 Dec;1007:251-62. doi: 10.1196/annals.1286.024. Ann N Y Acad Sci. 2003. PMID: 14993058 Review. - Role for estradiol in female-typical brain and behavioral sexual differentiation.
Bakker J, Baum MJ. Bakker J, et al. Front Neuroendocrinol. 2008 Jan;29(1):1-16. doi: 10.1016/j.yfrne.2007.06.001. Epub 2007 Jul 26. Front Neuroendocrinol. 2008. PMID: 17720235 Free PMC article. Review.
Cited by
- Maternal androgen excess significantly impairs sexual behavior in male and female mouse offspring: Perspective for a biological origin of sexual dysfunction in PCOS.
Donaldson NM, Prescott M, Ruddenklau A, Campbell RE, Desroziers E. Donaldson NM, et al. Front Endocrinol (Lausanne). 2023 Feb 15;14:1116482. doi: 10.3389/fendo.2023.1116482. eCollection 2023. Front Endocrinol (Lausanne). 2023. PMID: 36875467 Free PMC article. - Neuroanatomy and sex differences of the lordosis-inhibiting system in the lateral septum.
Tsukahara S, Kanaya M, Yamanouchi K. Tsukahara S, et al. Front Neurosci. 2014 Sep 17;8:299. doi: 10.3389/fnins.2014.00299. eCollection 2014. Front Neurosci. 2014. PMID: 25278832 Free PMC article. Review. - Contribution of pheromones processed by the main olfactory system to mate recognition in female mammals.
Baum MJ. Baum MJ. Front Neuroanat. 2012 Jun 5;6:20. doi: 10.3389/fnana.2012.00020. eCollection 2012. Front Neuroanat. 2012. PMID: 22679420 Free PMC article. - Touch neurons underlying dopaminergic pleasurable touch and sexual receptivity.
Elias LJ, Succi IK, Schaffler MD, Foster W, Gradwell MA, Bohic M, Fushiki A, Upadhyay A, Ejoh LL, Schwark R, Frazer R, Bistis B, Burke JE, Saltz V, Boyce JE, Jhumka A, Costa RM, Abraira VE, Abdus-Saboor I. Elias LJ, et al. Cell. 2023 Feb 2;186(3):577-590.e16. doi: 10.1016/j.cell.2022.12.034. Epub 2023 Jan 23. Cell. 2023. PMID: 36693373 Free PMC article.
References
- Baum MJ. Differentiation of coital behavior in mammals: a comparative analysis. Neurosci Biobehav Rev. 1979;3:265–284. - PubMed
- Baum MJ, Tobet SA. Effect of prenatal exposure to aromatase inhibitor, testosterone, or antiandrogen on the development of feminine sexual behavior in ferrets of both sexes. Physiol Behav. 1986;37:111–118. - PubMed
- Baum MJ, Carroll RS, Cherrv JA, Tobet SA. Steroidal control of behavioural, neuroendocrine and brain sexual differentiation: studies in a carnivore, the ferret. J Neuroendocrinol. 1990;2:401–418. - PubMed
- Baum MJ, Woutersen PJ, Slob AK. Sex difference in whole-body androgen content in rats on fetal days 18 and 19 without evidence that androgen passes from males to females. Biol Reprod. 1991;44:747–751. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous