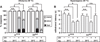Hedgehog signaling regulates nociceptive sensitization - PubMed (original) (raw)
Hedgehog signaling regulates nociceptive sensitization
Daniel T Babcock et al. Curr Biol. 2011.
Abstract
Background: Nociceptive sensitization is a tissue damage response whereby sensory neurons near damaged tissue enhance their responsiveness to external stimuli. This sensitization manifests as allodynia (aversive withdrawal to previously nonnoxious stimuli) and/or hyperalgesia (exaggerated responsiveness to noxious stimuli). Although some factors mediating nociceptive sensitization are known, inadequacies of current analgesic drugs have prompted a search for additional targets.
Results: Here we use a Drosophila model of thermal nociceptive sensitization to show that Hedgehog (Hh) signaling is required for both thermal allodynia and hyperalgesia following ultraviolet irradiation (UV)-induced tissue damage. Sensitization does not appear to result from developmental changes in the differentiation or arborization of nociceptive sensory neurons. Genetic analysis shows that Hh signaling acts in parallel to tumor necrosis factor (TNF) signaling to mediate allodynia and that distinct transient receptor potential (TRP) channels mediate allodynia and hyperalgesia downstream of these pathways. We also demonstrate a role for Hh in analgesic signaling in mammals. Intrathecal or peripheral administration of cyclopamine (CP), a specific inhibitor of Sonic Hedgehog signaling, blocked the development of analgesic tolerance to morphine (MS) or morphine antinociception in standard assays of inflammatory pain in rats and synergistically augmented and sustained morphine analgesia in assays of neuropathic pain.
Conclusions: We demonstrate a novel physiological role for Hh signaling, which has not previously been implicated in nociception. Our results also identify new potential therapeutic targets for pain treatment.
Copyright © 2011 Elsevier Ltd. All rights reserved.
Figures
Figure 1. hedgehog Mutants Fail to Develop Thermal Allodynia and Hyperalgesia
(A) Behavioral responses of control (w1118) and hedgehog (hhts2) mutant larvae to a normally nonnoxious stimulus of 38°C. Behaviors were classified as no response (white, >20 s), slow withdrawal (gray, between 5 and 20 s), or fast withdrawal (black, <5 s). Responses were measured with and without ultraviolet irradiation (UV)-induced tissue damage, in the presence or absence of a 24 hr 29°C heat shock administered after UV treatment. n = triplicate sets of 30 larvae per condition. Brackets with asterisks represent statistically significant comparisons by Fisher’s exact test. (B) Behavioral responses to a noxious suprathreshold stimulus of 45°C. Withdrawal latency was measured with and without UV-induced tissue damage in the presence or absence of an 8 hr 29°C heat shock administered after UV treatment. n = 50 larvae per condition. Error bars represent standard error of the mean (SEM). Brackets with asterisks represent statistically significant comparisons by two-way repeated-measures analysis of variance (ANOVA). n.s. indicates not significant.
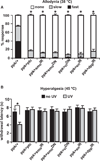
Figure 2. Hedgehog Signaling Components Are Required in Nociceptive Sensory Neurons
In both panels, ppk1.9-Gal4 drives (>) expression of the indicated upstream activating sequence (UAS) transgenes or no transgene (ppk1.9-Gal4 crossed to w1118 = ppk/+) in nociceptive sensory neurons. (A) Behavioral responses of larvae of indicated genotypes to a stimulus of 38°C 24 hr after UV treatment. n = triplicate sets of 30 larvae per condition. Asterisk represents statistical significance (p < 0.05) versus ppk/+ control by Fisher’s exact test. (B) Response of larvae of indicated genotypes to a 45°C stimulus without UV and 8 hr after UV treatment. n = 50 larvae. Bracket with asterisk represents statistically significant comparison by Fisher’s exact test. Error bars represent SEM. See also Figure S1.
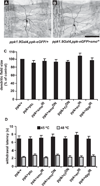
Figure 3. Blocking Hedgehog Signaling Components Does Not Affect Nociceptive Sensory Neuron Morphology or Baseline Nociception
(A and B) Whole mounts of dissected L3 larvae of the indicated genotypes. Sensory neuron morphology (ppk1.9Gal4, ppk-eGFP) is shown. (A) Control. (B) UAS-smoIR. Scale bar represents 100 µm. (C) Quantification of total dendritic arbor size (see Experimental Procedures) when ppk1.9-Gal4 drives expression of the indicated UAS transgenes in nociceptive sensory neurons. n = 10 larvae per genotype. (D) Baseline nociception in response to a 45°C or 48°C stimulus in the absence of UV damage when ppk1.9-Gal4 drives expression of the indicated UAS transgenes in nociceptive sensory neurons. n = 50 larvae. Error bars represent SEM. See also Figures S2 and S3.
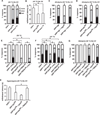
Figure 4. Epistasis Analysis of Tumor Necrosis Factor versus Hedgehog and Tumor Necrosis Factor or Hedgehog versus Transient Receptor Potential Channels
ppk1.9-Gal4 drives expression of the indicated UAS transgenes in nociceptive sensory neurons. (A and B) Constitutive activation of Hedgehog (Hh) signaling in the absence of UV irradiation produced thermal allodynia to a 38°C stimulus (A) and thermal hyperalgesia to a 45°C stimulus (B). (C) Constitutive activation of tumor necrosis factor (TNF) signaling causes allodynia that is reduced by knockdown of Wengen but not Smoothened. (D) Constitutive activation of Hh signaling causes allodynia that is reduced by knockdown of Smoothened but not Wengen. (E) RNAi directed against both Painless and TRPA1 reduce UV-induced thermal allodynia. (F) RNAi directed against TRPA1 prevents UV-induced thermal hyperalgesia whereas Painless RNAi-expressing larvae develop thermal hyperalgesia. Both comparisons are to the altered thresholds for baseline 45C in RNAi-expressing larvae. (G) Hh- and TNF-induced allodynia depend on Painless but not TRPA1. (H) Both Painless and TRPA1 reduce responsiveness to a 45°C stimulus in unirradiated larvae. Abbreviations are as follows: IR, inverted repeat; DN, dominant negative. n = triplicate sets of 30 larvae per condition. Error bars represent SEM.
Figure 5. Cyclopamine Modulates Morphine Analgesia in Rat Models of Inflammatory and Neuropathic Pain
(A) Inflammatory pain paradigm. Hindpaws were injected with saline (sham, n = 6), or complete Freund’s adjuvant (CFA). Thermal analgesia was assessed using paw withdrawal latency (PWL) following intrathecal administration of 10% Captisol (vehicle, n = 5), cyclopamine (CP, n = 6), morphine (MS, n = 6), or cyclopamine and morphine (MS + CP, n = 6). Abbreviations are as follows: BS, baseline before paw injection; 0, baseline after paw injection, prior to intrathecal drug administration. *p < 0.01 versus vehicle. (B) Neuropathic pain paradigm. Rats underwent sciatic nerve ligation or sham (n = 7) operation. Animals received daily intrathecal injections of vehicle (n = 3) or drugs (CP, n = 6; MS, n = 6; or MS + CP, n = 5). Mechanical allodynia was assessed using Von Frey filaments. Abbreviations are as follows: BS, baseline measurement prior to operation; 0, baseline 2 weeks after operation, prior to intrathecal drug administration. *p < 0.001 versus vehicle. All data are ±SEM. (C) Inflammatory pain paradigm with peripheral drug administration. Hindpaws were injected with CFA and cyclopamine, morphine, or a combination of both drugs. Animals received daily intraplantar injections of drugs. n = 9 animals per treatment group. Thermal analgesia was assessed using PWL. *p < 0.0001 versus vehicle (two-way repeated-measures ANOVA). All data are 6SEM.
Figure 6. Model of Hedgehog-Induced Thermal Allodynia and Hyperalgesia
Hh (from an as yet undetermined source tissue) acts through Patched (whose overexpression blocks sensitization), which subsequently inhibits Smoothened (required for sensitization) to activate signal transduction. The subsequent signaling steps likely involve the transcriptional activity of Cubitus interruptus (required for sensitization) and other components of the canonical Hh signaling pathway. For thermal allodynia, this pathway acts through the transient receptor potential (TRP) channel Painless, and for thermal hyperalgesia it requires the TRP channel dTRPA1.
Comment in
- Pain: A prickly solution?
Flight MH. Flight MH. Nat Rev Neurosci. 2011 Oct 5;12(11):618. doi: 10.1038/nrn3124. Nat Rev Neurosci. 2011. PMID: 21971069 No abstract available.
Similar articles
- The brinker repressor system regulates injury-induced nociceptive sensitization in Drosophila melanogaster.
McParland A, Moulton J, Brann C, Hale C, Otis Y, Ganter G. McParland A, et al. Mol Pain. 2021 Jan-Dec;17:17448069211037401. doi: 10.1177/17448069211037401. Mol Pain. 2021. PMID: 34399634 Free PMC article. - Tachykinin acts upstream of autocrine Hedgehog signaling during nociceptive sensitization in Drosophila.
Im SH, Takle K, Jo J, Babcock DT, Ma Z, Xiang Y, Galko MJ. Im SH, et al. Elife. 2015 Nov 17;4:e10735. doi: 10.7554/eLife.10735. Elife. 2015. PMID: 26575288 Free PMC article. - Cytokine signaling mediates UV-induced nociceptive sensitization in Drosophila larvae.
Babcock DT, Landry C, Galko MJ. Babcock DT, et al. Curr Biol. 2009 May 26;19(10):799-806. doi: 10.1016/j.cub.2009.03.062. Epub 2009 Apr 16. Curr Biol. 2009. PMID: 19375319 Free PMC article. - Sensory and signaling mechanisms of bradykinin, eicosanoids, platelet-activating factor, and nitric oxide in peripheral nociceptors.
Petho G, Reeh PW. Petho G, et al. Physiol Rev. 2012 Oct;92(4):1699-775. doi: 10.1152/physrev.00048.2010. Physiol Rev. 2012. PMID: 23073630 Review. - Pokes, sunburn, and hot sauce: Drosophila as an emerging model for the biology of nociception.
Im SH, Galko MJ. Im SH, et al. Dev Dyn. 2012 Jan;241(1):16-26. doi: 10.1002/dvdy.22737. Epub 2011 Sep 19. Dev Dyn. 2012. PMID: 21932321 Free PMC article. Review.
Cited by
- Nociception in fruit fly larvae.
Boivin JC, Zhu J, Ohyama T. Boivin JC, et al. Front Pain Res (Lausanne). 2023 Mar 17;4:1076017. doi: 10.3389/fpain.2023.1076017. eCollection 2023. Front Pain Res (Lausanne). 2023. PMID: 37006412 Free PMC article. Review. - Sonic Hedgehog Signaling Contributes to Chronic Post-Thoracotomy Pain via Activating BDNF/TrkB Pathway in Rats.
Yang Y, Wang X, Zhang X, You S, Feng L, Zhang Y, Shi Y, Xu Y, Zhang H. Yang Y, et al. J Pain Res. 2020 Jul 10;13:1737-1746. doi: 10.2147/JPR.S245515. eCollection 2020. J Pain Res. 2020. PMID: 32765048 Free PMC article. - Wengen, the sole tumour necrosis factor receptor in Drosophila, collaborates with moesin to control photoreceptor axon targeting during development.
Ruan W, Unsain N, Desbarats J, Fon EA, Barker PA. Ruan W, et al. PLoS One. 2013;8(3):e60091. doi: 10.1371/journal.pone.0060091. Epub 2013 Mar 27. PLoS One. 2013. PMID: 23544124 Free PMC article. - Growth Factor Signaling Regulates Mechanical Nociception in Flies and Vertebrates.
Lopez-Bellido R, Puig S, Huang PJ, Tsai CR, Turner HN, Galko MJ, Gutstein HB. Lopez-Bellido R, et al. J Neurosci. 2019 Jul 24;39(30):6012-6030. doi: 10.1523/JNEUROSCI.2950-18.2019. Epub 2019 May 28. J Neurosci. 2019. PMID: 31138657 Free PMC article. - Epigenetic modification of DRG neuronal gene expression subsequent to nerve injury: etiological contribution to complex regional pain syndromes (Part II).
Wang F, Stefano GB, Kream RM. Wang F, et al. Med Sci Monit. 2014 Jul 12;20:1188-200. doi: 10.12659/MSM.890707. Med Sci Monit. 2014. PMID: 25027291 Free PMC article. Review.
References
- Woolf CJ. Overcoming obstacles to developing new analgesics. Nat. Med. 2010;16:1241–1247. - PubMed
- Julius D, Basbaum AI. Molecular mechanisms of nociception. Nature. 2001;413:203–210. - PubMed
- Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D. The capsaicin receptor: A heat-activated ion channel in the pain pathway. Nature. 1997;389:816–824. - PubMed
- Davis JB, Gray J, Gunthorpe MJ, Hatcher JP, Davey PT, Overend P, Harries MH, Latcham J, Clapham C, Atkinson K, et al. Vanilloid receptor-1 is essential for inflammatory thermal hyperalgesia. Nature. 2000;405:183–187. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 NS069828-03/NS/NINDS NIH HHS/United States
- R01 AA016157/AA/NIAAA NIH HHS/United States
- R01 NS069828/NS/NINDS NIH HHS/United States
- R01 DA015146/DA/NIDA NIH HHS/United States
- R01 DA05146/DA/NIDA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous