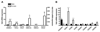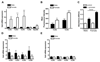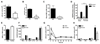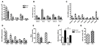Sex differences in resident immune cell phenotype underlie more efficient acute inflammatory responses in female mice - PubMed (original) (raw)
Sex differences in resident immune cell phenotype underlie more efficient acute inflammatory responses in female mice
Ramona S Scotland et al. Blood. 2011.
Abstract
Females are protected against mortality arising from severe sepsis; however, the precise mechanisms that confer this survival advantage in females over males are unclear. Resident leukocytes in resting tissues have a significant influence on circulating cytokine levels and recruitment of blood leukocytes during acute inflammatory responses. Whether the phenotype of resident leukocytes is distinct in females is unknown. In the present study, we show that the numbers of leukocytes occupying the naive peritoneal and pleural cavities is higher in female than in male mice and rats, comprising more T and B lymphocytes and macrophages. The altered immune cell composition of the female peritoneum is controlled by elevated tissue chemokine expression. Female resident macrophages also exhibit greater TLR expression and enhanced phagocytosis and NADPH oxidase-mediated bacterial killing. However, macrophage-derived cytokine production is diminished by proportionally more resident immunomodulatory CD4+ T lymphocytes. Ovarian hormones regulate macrophage phenotype, function, and numbers, but have no significant impact on T-lymphocyte populations in females. We have identified a fundamental sex difference in phenotype of resident leukocytes. We propose that the distinct resident leukocyte population in females allows aggressive recognition and elimination of diverse infectious stimuli without recruitment of circulating neutrophils or excessive cytokine production.
Conflict of interest statement
Disclosure of Conflict of Interest
The authors declare no conflicting financial interests.
Figures
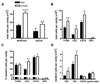
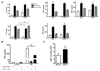
Figure 1. Distinct resident leukocyte population in the female peritoneal cavity.
(A) Increased total resident cell number in peritoneal (n=13 mice; 3 independent groups) and pleural (n=5 mice) cavities of female compared to male mice. (B) Total cell number and (C) percentage of F4/80+ macrophage, CD3+ T-lymphocytes, CD19+ B-lymphocytes and GR1+ granulocytes in peritoneal cavity of male and female mice were determined by flow cytometry (n=7 mice). (D) Increased total resident CD8+ and CD4+ T-lymphocytes but not CD4+/CD25+ T-regulatory or δγ T-lymphocytes in female peritoneal cavity (n=4 mice). All values are expressed as mean ± sem. All comparisons are relative to male. *P<0.05, **P<0.01 and ***P<0.001 by Student’s t-test.
Figure 2. Increased homeostatic leukocyte recruitment into female peritoneal cavity.
(A) Basal mRNA expression of chemokines CX3CL1, CCL2, CCL7, CXCL12, CXCL5 and CCL5 in mesenteric tissue (n=6 mice). (B) Chemokine receptor mRNA expression in resident peritoneal cells (n=4-6 mice). Levels of mRNA for each sample are normalized to corresponding mRNA levels of housekeeping gene for small 18S and calculated as fold expression relative to the mean value in males, except CX3CR1 (relative to female). All values are expressed as mean ± sem. *P<0.05 and **P<0.01 by Student’s t-test. ND denotes chemokine expression that was not detected within 35 PCR cycles.
Figure 3. Elevated pathogen-sensing and phagocytosis by female macrophage.
(A) Basal mRNA expression of Toll-like receptors and Myd88 in naive peritoneal cells (n=5-6 mice) and (B) Flow cytometry analysis of surface TLR2 and TLR4 protein expression on resident F4/80+ peritoneal macrophage (n=6-8; 2 independent experiments). (C) Phagocytosis of zymosan A (5x106 particles/105 cells, 30min) by equivalent numbers of resident peritoneal leukocytes (macrophage and lymphocytes), measured in vitro by a colorimetric assay (n=5 mice). Basal levels of TLR mRNA in (D) mesenteric tissue and (E) aortae of male and female mice (n=6 mice). Levels of mRNA for each sample are normalized to corresponding mRNA levels of housekeeping gene for small 18S and calculated as fold expression relative to the mean value in females. All results are shown as mean ± sem. *P<0.05, **P<0.01 and ***P<0.001 compared to male by Student’s t-test.
Figure 4. Reduced severity and neutrophil recruitment in peritonitis in females.
(A-C & F) Male and female mice were treated with Group B Streptococcus (GBS; 30x106 bacteria per mouse, ip; n=7 mice) for 3h. (A) Total cell number recovered from the peritoneal cavity, (B) sepsis severity score, and (C) whole blood bacterial count. (D) Phorbol ester PMA (1pg/ml)-induced NADPHoxidase activity in male and female resident peritoneal macrophage (105 cells/sample), measured in vitro by Amplex® Red over 7 mins (n=3 mice). (E) GBS levels following incubation in vitro (104 bacteria/sample) for 1h at 37°C with normal mouse plasma (n=3 samples from 6 mice in each group). (F) Concentration of GBS-induced cytokines in cell-free peritoneal lavage (n=7 mice). (G&H) Male and female mice were injected with zymosan A (1mg, ip). (G) Total peritoneal cell number (n=5-10; 2 independent experiments) and (H) number of F4/80+ macrophage, CD3+ T-lymphocytes, CD19+ B-lymphocytes and GR1+ granulocytes in peritoneal cavity of male and female mice 3h after injection of zymosan A (n=6 mice). All values (A-H) are expressed as mean ± sem. All comparisons are relative to male. *P<0.05, **P<0.01 and ***P<0.001 by Student’s t-test. § P<0.05 by two-way ANOVA followed by Bonferroni post-test; #P<0.001.
Figure 5. T-lymphocytes control the severity of innate inflammatory responses
(A) Cytokine production in vitro by male and female resident peritoneal cells (2x105 cells/sample, n=6 mice) following 3h stimulation by TLR4-specific LPS (0.1µg/ml) or TLR2 agonist Pam3CSK4 (Pam3, 0.1µg/ml). (B) TNFα production by isolated resident male peritoneal macrophage (1.5x105 cells/sample, n=3 mice) treated with LPS (0.1µg/ml, 18h) in the absence or presence of CD4+ve T lymphocytes (1.5x105 cells). (C) Zymosan-induced (1mg, ip, 3h) recruitment of GR1+ granulocytes into peritoneal cavity of C57BL/6 (wild type) and T-lymphocyte deficient Rag2 knockout (KO) mice (n=5 mice). All values are expressed as mean ± sem. #P<0.05 by one-way ANOVA compared to male and § P<0.05 by one-way ANOVA relative to macrophage alone. *P<0.05 by Students t-test relative to wild type.
Figure 6. Ovarian sex-hormones contribute to sex-differences in resident immune cell population.
Ovariectomy (OVX) or sham-operation was performed on female mice at 4 weeks of age and allowed to recover for 4-5 weeks. (A) Total number of resident F4/80+ macrophage, CD3+ or CD8+ T-lymphocytes, CD19+ B-lymphocytes in peritoneal cavity (n=5 mice). Basal mRNA expression of (B) mesenteric tissue chemokines (n=6-7 mice), (C) chemokine receptors on resident peritoneal leukocytes (n=6 mice), and (D) peritoneal leukocyte TLR expression (n=6 mice). (E) Surface expression of TLR2 or TLR4 on F4/80+ resident peritoneal macrophage (n=3-5 mice). (F) Phagocytosis of zymosan A (5x106 particles/105 cells, 30min, n=4 mice) by resident peritoneal cells in vitro. (G) Group B Streptococcus-induced (30x106 bacteria/mouse, ip, 3h, n=7 mice) accumulation of leukocytes in peritoneal cavity. All values (A-G) are expressed as mean ± sem. Expression of mRNA for each sample is normalized to corresponding levels of the housekeeping gene for small 18S and calculated as fold expression relative to ovariectomized (OVX) females. All comparisons are relative to sham-operated females. *P<0.05, **P<0.01 and ***P<0.001 by Student’s t-test. NS denotes P>0.05.
Comment in
- Go girls! Efficient female innate immunity.
Howie SE. Howie SE. Blood. 2011 Nov 24;118(22):5718-9. doi: 10.1182/blood-2011-09-381137. Blood. 2011. PMID: 22123906 No abstract available.
Similar articles
- LSP1 modulates leukocyte populations in resting and inflamed peritoneum.
Jongstra-Bilen J, Misener VL, Wang C, Ginzberg H, Auerbach A, Joyner AL, Downey GP, Jongstra J. Jongstra-Bilen J, et al. Blood. 2000 Sep 1;96(5):1827-35. Blood. 2000. PMID: 10961883 - Resident peritoneal leukocytes are important sources of MMP-9 during zymosan peritonitis: superior contribution of macrophages over mast cells.
Kolaczkowska E, Lelito M, Kozakiewicz E, van Rooijen N, Plytycz B, Arnold B. Kolaczkowska E, et al. Immunol Lett. 2007 Nov 15;113(2):99-106. doi: 10.1016/j.imlet.2007.07.017. Epub 2007 Aug 24. Immunol Lett. 2007. PMID: 17826846 - Resident peritoneal macrophages and mast cells are important cellular sites of COX-1 and COX-2 activity during acute peritoneal inflammation.
Kolaczkowska E, Goldys A, Kozakiewicz E, Lelito M, Plytycz B, van Rooijen N, Arnold B. Kolaczkowska E, et al. Arch Immunol Ther Exp (Warsz). 2009 Nov-Dec;57(6):459-66. doi: 10.1007/s00005-009-0053-6. Epub 2009 Nov 3. Arch Immunol Ther Exp (Warsz). 2009. PMID: 19885646 - Induction of triggering receptor expressed on myeloid cells 1 in murine resident peritoneal macrophages by monosodium urate monohydrate crystals.
Murakami Y, Akahoshi T, Hayashi I, Endo H, Kawai S, Inoue M, Kondo H, Kitasato H. Murakami Y, et al. Arthritis Rheum. 2006 Feb;54(2):455-62. doi: 10.1002/art.21633. Arthritis Rheum. 2006. PMID: 16447220 - Shades of white: new insights into tissue-resident leukocyte heterogeneity.
Psaila AM, Vohralik EJ, Quinlan KGR. Psaila AM, et al. FEBS J. 2022 Jan;289(2):308-318. doi: 10.1111/febs.15737. Epub 2021 Feb 16. FEBS J. 2022. PMID: 33513286 Review.
Cited by
- Cross-species comparison of airway epithelium transcriptomics.
Gui B, Wang Q, Wang J, Li X, Wu Q, Chen H. Gui B, et al. Heliyon. 2024 Sep 22;10(19):e38259. doi: 10.1016/j.heliyon.2024.e38259. eCollection 2024 Oct 15. Heliyon. 2024. PMID: 39391497 Free PMC article. - Endothelial Cell-Derived Extracellular Vesicles Promote Aberrant Neutrophil Trafficking and Subsequent Remote Lung Injury.
Zi SF, Wu XJ, Tang Y, Liang YP, Liu X, Wang L, Li SL, Wu CD, Xu JY, Liu T, Huang W, Xie JF, Liu L, Chao J, Qiu HB. Zi SF, et al. Adv Sci (Weinh). 2024 Oct;11(38):e2400647. doi: 10.1002/advs.202400647. Epub 2024 Aug 9. Adv Sci (Weinh). 2024. PMID: 39119837 Free PMC article. - Role of sex as a biological variable in neonatal alveolar macrophages.
Leek C, Cantu A, Sonti S, Gutierrez MC, Eldredge L, Sajti E, Xu HN, Lingappan K. Leek C, et al. Redox Biol. 2024 Sep;75:103296. doi: 10.1016/j.redox.2024.103296. Epub 2024 Aug 2. Redox Biol. 2024. PMID: 39098263 Free PMC article. - A New Application for Cenicriviroc, a Dual CCR2/CCR5 Antagonist, in the Treatment of Painful Diabetic Neuropathy in a Mouse Model.
Bober A, Piotrowska A, Pawlik K, Ciapała K, Maciuszek M, Makuch W, Mika J. Bober A, et al. Int J Mol Sci. 2024 Jul 5;25(13):7410. doi: 10.3390/ijms25137410. Int J Mol Sci. 2024. PMID: 39000516 Free PMC article. - Sex-related immunity: could Toll-like receptors be the answer in acute inflammatory response?
Popotas A, Casimir GJ, Corazza F, Lefèvre N. Popotas A, et al. Front Immunol. 2024 May 21;15:1379754. doi: 10.3389/fimmu.2024.1379754. eCollection 2024. Front Immunol. 2024. PMID: 38835761 Free PMC article. Review.
References
- Adrie C, Azoulay E, Francais A, et al. Influence of gender on the outcome of severe sepsis: a reappraisal. Chest. 2007;132:1786–1793. - PubMed
- Marriott I, Huet-Hudson YM. Sexual dimorphism in innate immune responses to infectious organisms. Immunol Res. 2006;34(3):177–92. - PubMed
- Ridker PM, Cook NR, Lee I-M, et al. A randomized trial of low-dose aspirin in the primary prevention of cardiovascular disease in women. N Engl J Med. 2005;352(13):1293–304. - PubMed
- Rosenberg H, Allard D. Evidence for caution: Women and statin use. Women and Health Protection Report. 2007 Jun
- Straub RH. The complex role of estrogens in inflammation. Endocr Rev. 2007;28:521–74. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials