Dimerization of oskar 3' UTRs promotes hitchhiking for RNA localization in the Drosophila oocyte - PubMed (original) (raw)
Dimerization of oskar 3' UTRs promotes hitchhiking for RNA localization in the Drosophila oocyte
Helena Jambor et al. RNA. 2011 Dec.
Abstract
mRNA localization coupled with translational control is a highly conserved and widespread mechanism for restricting protein expression to specific sites within eukaryotic cells. In Drosophila, patterning of the embryo requires oskar mRNA transport to the posterior pole of the oocyte and translational repression prior to localization. oskar RNA splicing and the 3' untranslated region (UTR) are required for posterior enrichment of the mRNA. However, reporter RNAs harboring the oskar 3' UTR can localize by hitchhiking with endogenous oskar transcripts. Here we show that the oskar 3' UTR contains a stem-loop structure that promotes RNA dimerization in vitro and hitchhiking in vivo. Mutations in the loop that abolish in vitro dimerization interfere with reporter RNA localization, and restoring loop complementarity restores hitchhiking. Our analysis provides insight into the molecular basis of RNA hitchhiking, whereby localization-incompetent RNA molecules can become locally enriched in the cytoplasm, by virtue of their association with transport-competent RNAs.
Figures
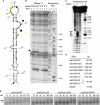
FIGURE 1.
osk RNAs associate in vitro via a conserved region in the 3′ UTR. (A) Schematic representation of osk 3′-UTR variants analyzed in this study. (B) Full-length and 3′ half osk 3′-UTR RNAs incubated in low- (M, monomer) or high-salt (D, dimer) buffers were separated by nondenaturing gel electrophoresis. A mobility shift occurs for both osk 3′-UTR and 3′ half RNAs in buffer D (lanes 2,6). A mobility shift of intermediate size (arrowhead) is observed when full-length and 3′ half RNAs were mixed in buffer D (lanes 3,4). Radioactive-labeled RNAs are indicated by a star (*) for each lane. (C) 5′ half and 3′ half of osk 3′-UTR RNAs incubated in water, low- (M) or high-salt (D) buffers were separated by nondenaturing gel electrophoresis and stained with ethidium bromide. In water the RNAs run as a single band of low mobility (lanes 3,6). The mobility of the 5′ half of osk 3′ UTR is identical in buffers D and M (lanes 4,5). In contrast, the 3′ half RNA migrates as a band of slower mobility at the higher salt concentration (buffer D, lane 1) than in low salt (buffer M, lane 2). (D) Alignment of oskar 3′-UTR cDNA sequences from Drosophila melanogaster, Drosophila yakuba, Drosophila pseudoobscura, Drosophila virilis, Drosophila immigrans, Drosophila mercantorum, and Zaprionus sepsoides, showing a central region of high conservation among Drosophilidae species (nucleotides 714–827 of the D. melanogaster osk 3′ UTR). Highlighted in gray are regions of >75% similarity. The predicted secondary structure of the D. melanogaster osk is depicted below: (a parenthesis) a base-paired nucleotide; (a dot) a single-stranded nucleotide; (blue rectangle) the palindromic sequence within the terminal loop.
FIGURE 2.
A secondary structure in the 3′ half of the osk 3′ UTR promotes dimerization. (A) Proposed secondary structure of nucleotides 714–827, the conserved region within the dimerization-competent portion of the osk 3′ UTR. Data supporting this prediction were collected by chemical (dimethyl-sulfate, DMS) and enzymatic (RNase T1 and T2) probing and are summarized in the figure. DMS (blue squares, weak; yellow circles, medium; red circles, strong modification) modifies unpaired adenines and cytosines, RNase T1 (solid circle, strong; open circle, medium; gray open circle, weak cleavage) cleaves unpaired guanines, and RNase T2 (solid square, strong; open square, medium; gray open square, weak cleavage) efficiently targets single-stranded nucleotides with a preference for adenines. (B) Enzymatic probing of the 3′ half of the osk 3′ UTR using RNase T1. Hydrolysis was conducted in buffer M or D, and the reaction was incubated for 2, 4, and 8 min. An incubation control (C) was included that did not contain the enzyme. Sequencing lanes (U,A,C,G) were run in parallel. Nucleotide 766 was specifically protected in buffer D but not in buffer M (arrow). (C) Phosphate ethylation interference by ENU. Autoradiography of the fractionation gel after modification conducted on the dimerization domain of osk RNA. (M and D) RNA extracted from the monomer and the dimer bands, respectively; (T) total population of modified RNA; (C) incubation control. Lane R corresponds to the RNase T1 ladder. Negative interference is indicated by circles: A complete disappearance of a band revealed a “strong” interference (solid circle), a slight disappearance of a band indicated a “weak” interference (open circle). (D) Summary of experiments probing the dimerization capacity (_K_D) of _osk_3′half wild-type, osk 3′half RNAs with mutations in the conserved stem–loop region and oskDD. Association of RNAs was strongly reduced when RNAs with partial or complete deletion of the stem–loop (osk3′half-ΔDD, -ΔDDd) or with substitutions in the terminal loop were analyzed. Dimerization was restored when RNAs with compensatory mutations (osk3′half-AA and -UU) were coincubated. _K_D's are indicated on the right. (E) Kinetics of dimerization of osk3′half-WT, osk3′half-ΔDD, osk3′half-ΔDDd, and _osk3′half_-GNRA RNAs as a function of RNA concentration. Indicated is the amount of nonradioactive RNA (nanomolar, nM); in addition, trace amounts of radioactive RNAs were added for visualization. Representative autoradiographs of the agarose gels show monomeric (gray arrow) and dimeric (black arrow) RNA species. Fifty percent of osk3′half-WT RNA formed dimers at 72–100 nM. RNAs bearing a mutation in the dimerization domain dimerized at >400 nM.
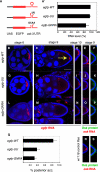
FIGURE 3.
The dimerization domain promotes osk RNA interaction during hitchhiking in vivo. (A) Schematic representation of the reporter constructs used in this panel. (A′–Q). Egg chambers from w1118 flies expressing egfp-WT, -UU, or -GNRA under the control of a _mat-α4-tub_-Gal4 driver. (A′) qRT-PCR experiment probing the relative levels of gene expression of egfp-WT, egfp-UU, and egfp-GNRA RNAs. RNA expression is shown as the percentage of the egfp-WT RNA level. All RNA levels were normalized to rp49 RNA. (B–P,R,S) Reporter RNAs are detected using an _egfp_-antisense probe, endogenous osk is visualized using a probe antisense to the osk coding sequence, and Osk protein is stained with anti-Osk antibodies. All egg chambers shown were counterstained with DAPI (blue). Bar, 50 μm. The panels highlighting the posterior pole of different oocytes (D–F,I–K,N–P) show images at the same magnification as C, H, and M. (B,G,L) egfp-WT, -UU, and GNRA reporter RNAs accumulate in young oocytes (stage 6). (C,H,M) Posterior accumulation of mutant egfp-UU and -GNRA reporter RNAs is strongly reduced in comparison with egfp-WT RNA at stage 9. The arrow in C indicates the position of the posterior pole. (D,I,N) egfp-WT, and to a lesser degree also egfp-UU and -GNRA, RNAs are present at the posterior pole of stage 10 oocytes. (E,J,O) Endogenous osk mRNA at stage 9 is localized in the presence of reporter RNAs. (F,K,P) Osk protein is expressed in stage 9 oocytes in the presence of reporter RNAs. (Q) Percentage of oocytes showing a posterior accumulation of egfp-WT, -UU, or -GNRA reporter RNAs in stage 9 egg chambers. The accumulation of egfp RNAs was scored in at least two independent experiments (egfp-WT: n = 127; egfp-UU: n = 61; egfp-GNRA: n = 71). (R,S) w1118 control flies, which do not express transgenic reporter3′UTR, were also stained for osk mRNA and Osk protein for comparison.
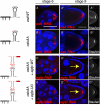
FIGURE 4.
osk RNAs engage in direct interactions in the oocyte. Egg chambers from osk RNA null flies [;;_osk_A87/Df(3R)pXT103] expressing full-length transgenic osk mRNA under the control of pCog and _nos_-Gal4 drivers. (_A_–_A_″,_B_–_B_″) RNA expressed from oskWT (_A_–_A_″) and oskAA (_B_–_B_″) transgenes is transported into the oocyte (stage 6) and localizes at the posterior pole of stage 9 egg chambers (oskWT, 93%; oskAA, 91%). In a subset of the egg chambers (oskWT, 30%; oskAA, 39%), ectopic osk mRNA is additionally detected. Staufen protein is equally enriched at the posterior pole of egg chambers. As established by qRT-PCR, oskWT and oskAA were overexpressed to similar levels in comparison to osk levels of w1118 flies. osk RNAs were detected using an osk (coding region)–antisense probe (red) immuno-stained with anti-Staufen antibodies (white). (_C_–_C_″,_D_–_D_″) Egg chambers coexpressing oskAA with either egfp-WT or egfp-UU. Both reporter RNAs enrich in young oocytes (C,D), but posterior pole accumulation of the egfp-WT is strongly reduced (C′) (16% ± 13%) compared with the egfp-UU (D′) (91% ± 5%) at stage 9 of oogenesis. Staufen enriches equally in the presence of either RNA (C″,D″). A summary of reporter3′UTR RNA hitchhiking at stage 9 of oogenesis is shown below. All egg chambers were simultaneously stained with _egfp_-antisense probe (red) to specifically detect the reporter RNAs and anti-Staufen antibodies (white) to also detect full-length osk mRNA. The accumulation of _egfp_-reporter RNA at the posterior pole was scored in at least two independent experiments, analyzing at least 15 egg chambers per experiment. All egg chambers were stained for RNA (red) by whole-mount in situ hybridization and counterstained with DAPI (blue). Bar, 50 μm.
Similar articles
- Multiple _cis_-acting signals, some weak by necessity, collectively direct robust transport of oskar mRNA to the oocyte.
Ryu YH, Kenny A, Gim Y, Snee M, Macdonald PM. Ryu YH, et al. J Cell Sci. 2017 Sep 15;130(18):3060-3071. doi: 10.1242/jcs.202069. Epub 2017 Jul 31. J Cell Sci. 2017. PMID: 28760927 Free PMC article. - mRNA localization and translational control in Drosophila oogenesis.
Lasko P. Lasko P. Cold Spring Harb Perspect Biol. 2012 Oct 1;4(10):a012294. doi: 10.1101/cshperspect.a012294. Cold Spring Harb Perspect Biol. 2012. PMID: 22865893 Free PMC article. Review. - Phylogenetic comparison of oskar mRNA localization signals.
Kim J, Lee J, Lee S, Lee B, Kim-Ha J. Kim J, et al. Biochem Biophys Res Commun. 2014 Jan 31;444(1):98-103. doi: 10.1016/j.bbrc.2014.01.021. Epub 2014 Jan 16. Biochem Biophys Res Commun. 2014. PMID: 24440702 - Control of RNP motility and localization by a splicing-dependent structure in oskar mRNA.
Ghosh S, Marchand V, Gáspár I, Ephrussi A. Ghosh S, et al. Nat Struct Mol Biol. 2012 Mar 18;19(4):441-9. doi: 10.1038/nsmb.2257. Nat Struct Mol Biol. 2012. PMID: 22426546 - Posttranscriptional regulation in Drosophila oocytes and early embryos.
Lasko P. Lasko P. Wiley Interdiscip Rev RNA. 2011 May-Jun;2(3):408-16. doi: 10.1002/wrna.70. Epub 2011 Jan 28. Wiley Interdiscip Rev RNA. 2011. PMID: 21957026 Review.
Cited by
- Two Arabidopsis late pollen transcripts are detected in cytoplasmic granules.
Scarpin MR, Sigaut L, Temprana SG, Boccaccio GL, Pietrasanta LI, Muschietti JP. Scarpin MR, et al. Plant Direct. 2017 Oct 16;1(4):e00012. doi: 10.1002/pld3.12. eCollection 2017 Oct. Plant Direct. 2017. PMID: 31245661 Free PMC article. - Multiple _cis_-acting signals, some weak by necessity, collectively direct robust transport of oskar mRNA to the oocyte.
Ryu YH, Kenny A, Gim Y, Snee M, Macdonald PM. Ryu YH, et al. J Cell Sci. 2017 Sep 15;130(18):3060-3071. doi: 10.1242/jcs.202069. Epub 2017 Jul 31. J Cell Sci. 2017. PMID: 28760927 Free PMC article. - Region-specific activation of oskar mRNA translation by inhibition of Bruno-mediated repression.
Kim G, Pai CI, Sato K, Person MD, Nakamura A, Macdonald PM. Kim G, et al. PLoS Genet. 2015 Feb 27;11(2):e1004992. doi: 10.1371/journal.pgen.1004992. eCollection 2015. PLoS Genet. 2015. PMID: 25723530 Free PMC article. - Formation, function, and pathology of RNP granules.
Ripin N, Parker R. Ripin N, et al. Cell. 2023 Oct 26;186(22):4737-4756. doi: 10.1016/j.cell.2023.09.006. Cell. 2023. PMID: 37890457 Free PMC article. Review. - mRNA localization and translational control in Drosophila oogenesis.
Lasko P. Lasko P. Cold Spring Harb Perspect Biol. 2012 Oct 1;4(10):a012294. doi: 10.1101/cshperspect.a012294. Cold Spring Harb Perspect Biol. 2012. PMID: 22865893 Free PMC article. Review.
References
- Brunel C, Marquet R, Romby P, Ehresmann C 2002. RNA loop–loop interactions as dynamic functional motifs. Biochimie 84: 925–944 - PubMed
- Chekulaeva M, Hentze MW, Ephrussi A 2006. Bruno acts as a dual repressor of oskar translation, promoting mRNA oligomerization and formation of silencing particles. Cell 124: 521–533 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases