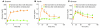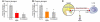Glucagonocentric restructuring of diabetes: a pathophysiologic and therapeutic makeover - PubMed (original) (raw)
Review
Glucagonocentric restructuring of diabetes: a pathophysiologic and therapeutic makeover
Roger H Unger et al. J Clin Invest. 2012 Jan.
Abstract
The hormone glucagon has long been dismissed as a minor contributor to metabolic disease. Here we propose that glucagon excess, rather than insulin deficiency, is the sine qua non of diabetes. We base this on the following evidence: (a) glucagon increases hepatic glucose and ketone production, catabolic features present in insulin deficiency; (b) hyperglucagonemia is present in every form of poorly controlled diabetes; (c) the glucagon suppressors leptin and somatostatin suppress all catabolic manifestations of diabetes during total insulin deficiency; (d) total β cell destruction in glucagon receptor-null mice does not cause diabetes; and (e) perfusion of normal pancreas with anti-insulin serum causes marked hyperglucagonemia. From this and other evidence, we conclude that glucose-responsive β cells normally regulate juxtaposed α cells and that without intraislet insulin, unregulated α cells hypersecrete glucagon, which directly causes the symptoms of diabetes. This indicates that glucagon suppression or inactivation may provide therapeutic advantages over insulin monotherapy.
Figures
Figure 1. Relationship between hepatic sinusoidal glucagon and glucose production in vivo.
A pancreatic clamp was used to keep plasma insulin basal and constant. The glucose production rate reflects the maximal effect of glucagon and was observed approximately 15 minutes after the change in the hormone level. In this way, the accompanying hyperglycemia was limited such that its inhibitory effect on glucose production was minimal. When glucagon was made deficient (i.e., 0 pg/ml), euglycemia was maintained by glucose infusion. The region shaded blue denotes the physiologic range of plasma glucagon. Figure adapted with permission from Handbook of Physiology (96).
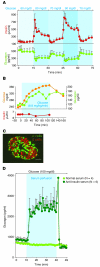
Figure 2. Relationship between insulin and glucagon secretion.
(A) Responses of insulin and glucagon to minor changes in glucose perfused into isolated pancreata of normal dogs. The perfusate glucose concentration varied from 60 to 90 mg/dl. Modest changes in the perfusing glucose concentration led to major reciprocal responses of both insulin and glucagon. Figure adapted with permission from Diabetologia (38). (B) Demonstration that a rise in glucose “paradoxically” stimulates glucagon secretion when it is not accompanied by the rise in insulin that normally accompanies elevations in glucose concentration. Figure adapted from Journal of Clinical Investigation (43). (C) Topographic scheme of a normal human islet showing the extensive juxtaposition of β cells (red) to α cells (green) that facilitates instantaneous insulin control of glucagon secretion via the interstitial space separating the two cells. Scale bar: 50 μm. Figure reproduced with permission from Diabetes (48). (D) Direct physiologic evidence of the paracrine role of insulin on α cell function in rodents. The isolated pancreata of normal rats are perfused with either nonimmune serum, as control, or a potent anti-insulin serum. The sudden rise in glucagon upon infusion of the anti-insulin serum indicates an ongoing paracrine inhibition of glucagon secretion by the insulin in the islets. Figure adapted from Journal of Clinical Investigation (53).
Figure 3. Glucagon is essential in diabetic hyperglycemia.
(A) Perfusion of a severely diabetic, insulin-deprived dog with somatostatin. The hyperglycemia and hyperglucagonemia are promptly suppressed by the somastotatin infusion, and both reappear when it is stopped. Figure adapted with permission from Science (58). (B) A similar experiment in type 1 diabetic humans receiving a suboptimal insulin dose administered by intravenous infusion (60). Their hyperglucagonemia, hyperglycemia, and glycosuria are suppressed soon after beginning an infusion of somastotatin, confirming earlier work by Gerich et al. (59). When hyperglucagonemia was restored by infusion of recombinant glucagon, hyperglycemia and glycosuria reappeared. Figure adapted with permission from New England Journal of Medicine (60).
Figure 4. Glucagon is the sine qua non of diabetes in mice.
(A) Glucose levels in normal wild-type mice and in Gcgr–/– mice after destruction of β cells by double-dose streptozotocin treatment. Gcgr–/– mice remain normoglycemic and exhibit no detectable metabolic consequence of total insulin deficiency. (B) Insulin response to oral glucose in Gcgr–/– mice before and after β cell destruction. (C) Oral glucose tolerance curve of Gcgr–/– mice before and after β cell destruction. Remarkably, although streptozotocin-treated Gcgr–/– mice were incapable of secreting insulin in response to an oral glucose tolerance test, their glucose tolerance curves did not differ significantly from Gcgr–/– mice with intact β cells and a robust insulin response. In other words, in this model of congenital absence of glucagon activity, insulin has become irrelevant. (A–C) Figure adapted with permission from Diabetes (71).
Figure 5. Why insulin monotherapy in T1DM cannot restore normal glycemic stability.
(A) Concentration disparity of secreted insulin normally delivered to target organs. Normal α cells receive 100 times more insulin than do peripheral tissues. (B) In T1DM, all targets receive the same concentration of injected insulin. Levels high enough to suppress α cells are too high for the liver and the peripheral tissues. (C) By lowering the insulin dose and suppressing hyperglucagonemia with a noninsulin glucagon suppressor, glycemic stability is achieved. (D) Suppression of glycemic volatility in T1DM. NOD mice were treated with optimal insulin dose (0.2 U twice daily); other mice were treated with a suboptimal insulin dose (0.02 U twice daily) and a subcutaneous infusion of leptin. Mean glucose values were determined at 10 a.m. and 5 p.m. Leptin suppressed glucose volatility in these mice by preventing hyperglucagonemia, and hypoglycemia was prevented by reducing the insulin. Figure adapted with permission from Proceedings of the National Academy of Sciences of the United States of America (51).
Figure 6. Pathways for the glucagon-suppressing action of leptin.
(A) Plasma glucagon in NOD mice treated with placebo (No Rx) or with leptin infused subcutaneously. Figure adapted with permission from Proceedings of the National Academy of Sciences of the United States of America (92). (B) Plasma glucagon in streptozotocin-diabetic mice treated with of placebo or leptin infused intracerebroventricularly. Figure adapted with permission from Proceedings of the National Academy of Sciences of the United States of America (91). (C) Proposed dual control model of α cell secretion. LepR, leptin receptor.
Similar articles
- Glucagon is the key factor in the development of diabetes.
Lee YH, Wang MY, Yu XX, Unger RH. Lee YH, et al. Diabetologia. 2016 Jul;59(7):1372-1375. doi: 10.1007/s00125-016-3965-9. Epub 2016 Apr 26. Diabetologia. 2016. PMID: 27115412 Review. - Role of hyperglucagonemia in catabolism associated with type 1 diabetes: effects on leucine metabolism and the resting metabolic rate.
Charlton MR, Nair KS. Charlton MR, et al. Diabetes. 1998 Nov;47(11):1748-56. doi: 10.2337/diabetes.47.11.1748. Diabetes. 1998. PMID: 9792544 - Alterations in somatostatin and other islet cell functions in the spontaneously diabetic BB Wistar rat: biochemical and morphological characterization.
Patel YC, Ruggere D, Malaisse-Lagae F, Orci L. Patel YC, et al. Metabolism. 1983 Jul;32(7 Suppl 1):18-25. doi: 10.1016/s0026-0495(83)80006-7. Metabolism. 1983. PMID: 6135136 - Insulin, glucagon, and somatostatin in normal physiology and diabetes mellitus.
Felig P, Wahren J, Sherwin R, Hendler R. Felig P, et al. Diabetes. 1976 Dec;25(12):1091-9. doi: 10.2337/diab.25.12.1091. Diabetes. 1976. PMID: 992227 - Glucagon and diabetes.
Raskin P, Unger RH. Raskin P, et al. Med Clin North Am. 1978 Jul;62(4):713-22. doi: 10.1016/s0025-7125(16)31767-9. Med Clin North Am. 1978. PMID: 355737 Review.
Cited by
- Ramadan and Diabetes: A Narrative Review and Practice Update.
Ahmed SH, Chowdhury TA, Hussain S, Syed A, Karamat A, Helmy A, Waqar S, Ali S, Dabhad A, Seal ST, Hodgkinson A, Azmi S, Ghouri N. Ahmed SH, et al. Diabetes Ther. 2020 Nov;11(11):2477-2520. doi: 10.1007/s13300-020-00886-y. Epub 2020 Sep 9. Diabetes Ther. 2020. PMID: 32909192 Free PMC article. Review. - Direct Hepatocyte Insulin Signaling Is Required for Lipogenesis but Is Dispensable for the Suppression of Glucose Production.
Titchenell PM, Quinn WJ, Lu M, Chu Q, Lu W, Li C, Chen H, Monks BR, Chen J, Rabinowitz JD, Birnbaum MJ. Titchenell PM, et al. Cell Metab. 2016 Jun 14;23(6):1154-1166. doi: 10.1016/j.cmet.2016.04.022. Epub 2016 May 26. Cell Metab. 2016. PMID: 27238637 Free PMC article. - Diabetes-related alterations in the enteric nervous system and its microenvironment.
Bagyánszki M, Bódi N. Bagyánszki M, et al. World J Diabetes. 2012 May 15;3(5):80-93. doi: 10.4239/wjd.v3.i5.80. World J Diabetes. 2012. PMID: 22645637 Free PMC article. - New strategy for the treatment of type 2 diabetes mellitus with incretin-based therapy.
Namba M, Katsuno T, Kusunoki Y, Matsuo T, Miuchi M, Miyagawa J. Namba M, et al. Clin Exp Nephrol. 2013 Feb;17(1):10-5. doi: 10.1007/s10157-012-0709-0. Epub 2012 Nov 8. Clin Exp Nephrol. 2013. PMID: 23135865 Review. - Hypothalamic glucagon signaling inhibits hepatic glucose production.
Mighiu PI, Yue JT, Filippi BM, Abraham MA, Chari M, Lam CK, Yang CS, Christian NR, Charron MJ, Lam TK. Mighiu PI, et al. Nat Med. 2013 Jun;19(6):766-72. doi: 10.1038/nm.3115. Epub 2013 May 19. Nat Med. 2013. PMID: 23685839
References
- Kimball CP, Murlin JR. Aqueous extracts of pancreas. III. Some precipitation reactions of insulin. J Biol Chem. 1923;58(1):337–346.
- Keller U, Chiasson JL, Liljenquist JE, Cherrington AD, Jennings AS, Crofford OS. The roles of insulin, glucagon, and free fatty acids in the regulation of ketogenesis in dogs. Diabetes. 1977;26(11):1040–1051. - PubMed
- Cherrington AD, Chiasson JL, Liljenquist JE, Lacy WW, Park CR. Control of hepatic glucose output by glucagon and insulin in the intact dog. Biochem Soc Symp. 1978;43(43):31–45. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical