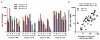Novel adenovirus-based vaccines induce broad and sustained T cell responses to HCV in man - PubMed (original) (raw)
. 2012 Jan 4;4(115):115ra1.
doi: 10.1126/scitranslmed.3003155.
Antonella Folgori, Stefania Capone, Leo Swadling, Stephen Aston, Ayako Kurioka, Joel Meyer, Rachel Huddart, Kira Smith, Rachel Townsend, Anthony Brown, Richard Antrobus, Virginia Ammendola, Mariarosaria Naddeo, Geraldine O'Hara, Chris Willberg, Abby Harrison, Fabiana Grazioli, Maria Luisa Esposito, Loredana Siani, Cinzia Traboni, Ye Oo, David Adams, Adrian Hill, Stefano Colloca, Alfredo Nicosia, Riccardo Cortese, Paul Klenerman
Affiliations
- PMID: 22218690
- PMCID: PMC3627207
- DOI: 10.1126/scitranslmed.3003155
Novel adenovirus-based vaccines induce broad and sustained T cell responses to HCV in man
Eleanor Barnes et al. Sci Transl Med. 2012.
Abstract
Currently, no vaccine exists for hepatitis C virus (HCV), a major pathogen thought to infect 170 million people globally. Many studies suggest that host T cell responses are critical for spontaneous resolution of disease, and preclinical studies have indicated a requirement for T cells in protection against challenge. We aimed to elicit HCV-specific T cells with the potential for protection using a recombinant adenoviral vector strategy in a phase 1 study of healthy human volunteers. Two adenoviral vectors expressing NS proteins from HCV genotype 1B were constructed based on rare serotypes [human adenovirus 6 (Ad6) and chimpanzee adenovirus 3 (ChAd3)]. Both vectors primed T cell responses against HCV proteins; these T cell responses targeted multiple proteins and were capable of recognizing heterologous strains (genotypes 1A and 3A). HCV-specific T cells consisted of both CD4+ and CD8+ T cell subsets; secreted interleukin-2, interferon-γ, and tumor necrosis factor-α; and could be sustained for at least a year after boosting with the heterologous adenoviral vector. Studies using major histocompatibility complex peptide tetramers revealed long-lived central and effector memory pools that retained polyfunctionality and proliferative capacity. These data indicate that an adenoviral vector strategy can induce sustained T cell responses of a magnitude and quality associated with protective immunity and open the way for studies of prophylactic and therapeutic vaccines for HCV.
Figures
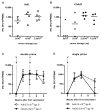
Figure 1. Magnitude of T cell responses primed after vaccination with Ad6-NSmut or ChAd3-NSmut
(A, B) Peak total IFNγ ELISpot response ex vivo: ELISpot data from individuals vaccinated with (A) Ad6-NSmut (open symbols) groups 1-3 (dose escalation 5 × 108-2.5 ×1010vp) and (B) ChAd3-NSmut (shaded symbols) groups 5-7 (dose escalation 5 × 108-7.5 ×1010vp). Vaccine dose is given as virus particles (vp). The responses shown are the total positive IFNγ ELISpot response across all pools (see methods) measured at peak response after prime (weeks 2-8) (Bars=median). (C, D) Single vs double priming: The kinetics of the priming responses after (C) double prime (groups 3 and 7) and (D) single prime (groups 9 and 10) at high vaccine dose (2.5 ×1010 vp). Group medians and IQR over time are shown. Open circles indicate Ad6-NSmut priming and closed symbols ChAd3-NSmut priming.
Figure 2. Breadth of T cell responses primed after vaccination with Ad6-NSmut or ChAd3-NSmut
(A). Breadth of primed response ex vivo: ELISpot data from individuals vaccinated with Ad6-NSmut (open symbols) groups 1-3 (dose escalation 5 × 108-2.5 ×1010vp) and ChAd3-NSmut (shaded symbols) groups 5-7 (dose escalation 5 × 108-2.5 ×1010vp). The responses shown are the number of positive pools (see methods) measured at peak magnitude after prime (weeks 2-8) (Bars=median). (B). Correlation of breadth and magnitude: the number of positive pools (from Fig. 2A) compared with the magnitude of the ELISpot response (from Fig. 1A and 1B) are plotted.
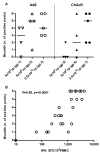
Figure 3. Specificity of T cell responses primed after vaccination with Ad6-NSmut or ChAd3-NSmut
(A) Immunodominance of primed responses: Data are taken from the IFNγ ELISpot responses from high dose groups (3, 9; Ad6-NSmut 2.5 ×1010 vp and 7, 10; ChAd3 2.5 ×1010vp) at peak magnitude after prime. The magnitude of the HCV-specific T cell response to individual peptide pools (F-M), and the corresponding NS protein below is shown (Bars=mean +/− SEM, ▯=statistical significance p=0.0001). (B) Individual volunteer data showing targeted pools after priming with Ad6-NSmut 2.5 ×1010 vp group 3 (upper panel) and ChAd3-NSmut 2.5 ×1010vp group 7 (lower panel) is shown.
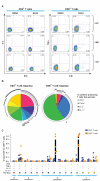
Figure 4. Functionality of T cell responses primed after vaccination with Ad6-NSmut or ChAd3-NSmut
A: Example FACS plots after Intracellular Cytokine Staining (ICS): Staining for IFNγ/IL-2, and IFNγ/TNF are shown for CD4+ and CD8+ T cell responses respectively, after stimulation with F+G+H (NS3/4) pooled peptides (see methods) in three donors (037 gp.7, 068 gp9, and 102 gp3; weeks 4-6). Plots are gated on live, CD3+, CD4+ or CD8+ T cell populations. B and C: Group data for ICS after priming. SPICE analysis for combined ICS data (after stimulation with NS3/4 peptides) for CD8+ and CD4+ T cell responses after priming (2-4 weeks) from 6 volunteers each in high dose groups (3, 7, 9 and 10; responses >0.05%). The pie charts (B) represents the proportion of cytokine secreting cells that produce one, two, three or four cytokines (IFNγ, TNFα, IL17 and/or IL-2). CD4+ and CD8+ T cells are shown on left and right plots respectively. The graph in panel C shows individual cytokines produced alone and in combination, as % of total CD4+ and CD8+ T cells. Each dot (orange= CD8+, green=CD4+ T cells) represents responses in an individual. Black bars = mean response.
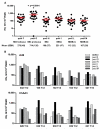
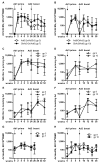
Figure 5. Cross-reactivity of T cell responses induced after vaccination with Ad6-NSmut or ChAd3-NSmut
(A); Cross-reactivity measured by IFNγ ELISpot response ex vivo against Genotype 1B with Genotype 1A and 3A peptide pools (F-M) in volunteers from group 11 (7.5×1010 vp ChAd3-NSmut) tested at week 4. (B); Correlation of IFNγ ELISpot response ex vivo against Genotype 1B with Genotype 1A and 3A peptide pools: ELISpot data from individuals in group 11 (7.5×1010 vp ChAd3-NSmut) tested at week 4. Each symbol represents a single pool response in a single volunteer.
Figure 6. Boosting of T cell responses primed after vaccination with Ad6-NSmut or ChAd3-NSmut
(A and B). Boosting of primed responses: A. Group data from IFNγ ELISpot responses in high dose double prime groups (3; Ad6-NSmut 2.5 ×1010 vp and 7; ChAd3-NSmut 2.5 ×1010 vp) over time. The responses shown are the total positive IFNγ ELISpot response across all pools (see methods) presented as group medians and IQR. Open symbols indicate Ad6-NSmut priming (ChAd3-NSmut boost) and shaded symbols ChAd3-NSmut priming (Ad6-NSmut boost). B. Similar data for groups 9 and 10 (single prime groups) (C-F) Neutralizing antibody titers (Nab): Homologous (against priming vector) (C, D) and heterologous (against boosting vector) (E, F) NAb titers are shown presented as group medians and IQR: Open symbols indicate Ad6-NSmut priming (ChAd3-NSmut boost) and shaded symbols ChAd3-NSmut priming (Ad6-NSmut boost). C and E show double-primed groups 3 and 7; D and F show single primed groups 9 and 10. (G and H) Adenovirus specific responses after priming and boosting: Group data from IFNγ ELISpot responses in high dose groups (G double prime, and H single prime groups) over time. The responses shown are the total positive IFNγ ELISpot response across to Ad5 Hexon peptides (see methods) presented as group medians and IQR. Open symbols indicate Ad6-NSmut priming (ChAd3-NSmut boost) and shaded symbols ChAd3-NSmut priming (Ad6-NSmut boost).
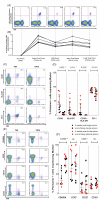
Figure 7. Characterization of epitope-specific T cell responses induced after vaccination with Ad6-NSmut or ChAd3-NSmut
(A) Staining with tetramer A2-HCV-1406: KLSGLGINAV, in a representative volunteer (volunteer 60 gp 10) over the study time course. Gating is on live CD3+ (see methods). % pentamer+/CD8+ T cells is shown. (B) Ex vivo tetramer + CD8+ T cell responses over time in 6 volunteers who had an ELISpot response to pool Gb that contains the A2 1406 peptide (Table S2 volunteers 32, 38, 105, 60, 64, and 68). Each received high dose (2.5 × 1010vp) ChAd3-NSmut or Ad6-NSmut. The Activation (C and D) and memory differentiation status. (E and F) of pentamer + cells (gated on CD8+ T cells). FACS plots (C and E) show a representative volunteer (060 gp 10) at weeks 4 and 36 after ChAd3-NSmut prime (week 0) and Ad6-NSmut boost (week 8) (both 2.5 × 1010vp). % given is the proportion of tetramer + cells expressing the phenotypic y legend characteristic. The graphs (D and F) show % pentamer + cells (HLA-A2-HCV-1406 KLSGLGINAV, and HLA-A1-HCV-1435 ATDALMTY) in 9 individuals (HLA-A2; 032, 038, 060, 064, 068, 105 and HLA-A1; 19, 37, 66, 68) expressing the phenotypic markers given in the x axis, 4 weeks after single prime (red circles) or 4 weeks after second prime (black circles) and at the end of the study (week 52 for double prime groups and week 36 for single prime groups) (squares; red in the single prime and black in the double prime group) after ChAd3-NSmut prime (week 0) and Ad6-NSmut boost (week 8) (both 2.5 × 1010vp). Gates were set using the tetramer negative cell population as a reference.
Comment in
- Hepatitis: HCV vaccine is successful in a phase I study in healthy volunteers.
Greenhill C. Greenhill C. Nat Rev Gastroenterol Hepatol. 2012 Feb 7;9(3):126. doi: 10.1038/nrgastro.2012.12. Nat Rev Gastroenterol Hepatol. 2012. PMID: 22310919 No abstract available. - Viral disease: Steps towards an HCV vaccine.
Harrison C. Harrison C. Nat Rev Drug Discov. 2012 Feb 17;11(3):187. doi: 10.1038/nrd3687. Nat Rev Drug Discov. 2012. PMID: 22338643 No abstract available.
Similar articles
- A human vaccine strategy based on chimpanzee adenoviral and MVA vectors that primes, boosts, and sustains functional HCV-specific T cell memory.
Swadling L, Capone S, Antrobus RD, Brown A, Richardson R, Newell EW, Halliday J, Kelly C, Bowen D, Fergusson J, Kurioka A, Ammendola V, Del Sorbo M, Grazioli F, Esposito ML, Siani L, Traboni C, Hill A, Colloca S, Davis M, Nicosia A, Cortese R, Folgori A, Klenerman P, Barnes E. Swadling L, et al. Sci Transl Med. 2014 Nov 5;6(261):261ra153. doi: 10.1126/scitranslmed.3009185. Sci Transl Med. 2014. PMID: 25378645 Free PMC article. Clinical Trial. - Enhanced and sustained CD8+ T cell responses with an adenoviral vector-based hepatitis C virus vaccine encoding NS3 linked to the MHC class II chaperone protein invariant chain.
Mikkelsen M, Holst PJ, Bukh J, Thomsen AR, Christensen JP. Mikkelsen M, et al. J Immunol. 2011 Feb 15;186(4):2355-64. doi: 10.4049/jimmunol.1001877. Epub 2011 Jan 21. J Immunol. 2011. PMID: 21257961 - Combined adenovirus vector and hepatitis C virus envelope protein prime-boost regimen elicits T cell and neutralizing antibody immune responses.
Chmielewska AM, Naddeo M, Capone S, Ammendola V, Hu K, Meredith L, Verhoye L, Rychlowska M, Rappuoli R, Ulmer JB, Colloca S, Nicosia A, Cortese R, Leroux-Roels G, Balfe P, Bienkowska-Szewczyk K, Meuleman P, McKeating JA, Folgori A. Chmielewska AM, et al. J Virol. 2014 May;88(10):5502-10. doi: 10.1128/JVI.03574-13. Epub 2014 Mar 5. J Virol. 2014. PMID: 24599994 Free PMC article. - Progress in the development of vaccines for hepatitis C virus infection.
Ghasemi F, Rostami S, Meshkat Z. Ghasemi F, et al. World J Gastroenterol. 2015 Nov 14;21(42):11984-2002. doi: 10.3748/wjg.v21.i42.11984. World J Gastroenterol. 2015. PMID: 26576087 Free PMC article. Review. - Progress in the development of preventive and therapeutic vaccines for hepatitis C virus.
Torresi J, Johnson D, Wedemeyer H. Torresi J, et al. J Hepatol. 2011 Jun;54(6):1273-85. doi: 10.1016/j.jhep.2010.09.040. Epub 2011 Jan 12. J Hepatol. 2011. PMID: 21236312 Review.
Cited by
- Absence of systemic toxicity changes following intramuscular administration of novel pSG2.HIVconsv DNA, ChAdV63.HIVconsv and MVA.HIVconsv vaccines to BALB/c mice.
Ondondo B, Brennan C, Nicosia A, Crome SJ, Hanke T. Ondondo B, et al. Vaccine. 2013 Nov 12;31(47):5594-601. doi: 10.1016/j.vaccine.2013.06.068. Epub 2013 Jul 2. Vaccine. 2013. PMID: 23831324 Free PMC article. - Altered Glycosylation Patterns Increase Immunogenicity of a Subunit Hepatitis C Virus Vaccine, Inducing Neutralizing Antibodies Which Confer Protection in Mice.
Li D, von Schaewen M, Wang X, Tao W, Zhang Y, Li L, Heller B, Hrebikova G, Deng Q, Ploss A, Zhong J, Huang Z. Li D, et al. J Virol. 2016 Nov 14;90(23):10486-10498. doi: 10.1128/JVI.01462-16. Print 2016 Dec 1. J Virol. 2016. PMID: 27630242 Free PMC article. - Development of prophylactic vaccines against HIV-1.
Schiffner T, Sattentau QJ, Dorrell L. Schiffner T, et al. Retrovirology. 2013 Jul 17;10:72. doi: 10.1186/1742-4690-10-72. Retrovirology. 2013. PMID: 23866844 Free PMC article. Review. - T cell responses induced by adenoviral vectored vaccines can be adjuvanted by fusion of antigen to the oligomerization domain of C4b-binding protein.
Forbes EK, de Cassan SC, Llewellyn D, Biswas S, Goodman AL, Cottingham MG, Long CA, Pleass RJ, Hill AV, Hill F, Draper SJ. Forbes EK, et al. PLoS One. 2012;7(9):e44943. doi: 10.1371/journal.pone.0044943. Epub 2012 Sep 12. PLoS One. 2012. PMID: 22984589 Free PMC article. - Prime-boost vaccination of mice and rhesus macaques with two novel adenovirus vectored COVID-19 vaccine candidates.
Luo S, Zhang P, Liu B, Yang C, Liang C, Wang Q, Zhang L, Tang X, Li J, Hou S, Zeng J, Fu Y, Allain JP, Li T, Zhang Y, Li C. Luo S, et al. Emerg Microbes Infect. 2021 Dec;10(1):1002-1015. doi: 10.1080/22221751.2021.1931466. Emerg Microbes Infect. 2021. PMID: 33993845 Free PMC article.
References
- Lauer GM, Walker BD. Hepatitis C virus infection. N Engl J Med. 2001;345:41–52. - PubMed
- Grakoui A, Shoukry NH, Woollard DJ, Han JH, Hanson HL, Ghrayeb J, Murthy KK, Rice CM, Walker CM. HCV persistence and immune evasion in the absence of memory T cell help. Science. 2003;302:659–662. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- G1002552/MRC_/Medical Research Council/United Kingdom
- 1U19AI082630-01/AI/NIAID NIH HHS/United States
- 091663/WT_/Wellcome Trust/United Kingdom
- 090532/WT_/Wellcome Trust/United Kingdom
- WT_/Wellcome Trust/United Kingdom
- U19 AI082630/AI/NIAID NIH HHS/United States
- G0901723/MRC_/Medical Research Council/United Kingdom
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous