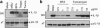ER stress activates the NLRP3 inflammasome via an UPR-independent pathway - PubMed (original) (raw)
ER stress activates the NLRP3 inflammasome via an UPR-independent pathway
P Menu et al. Cell Death Dis. 2012.
Abstract
Uncontrolled endoplasmic reticulum (ER) stress responses are proposed to contribute to the pathology of chronic inflammatory diseases such as type 2 diabetes or atherosclerosis. However, the connection between ER stress and inflammation remains largely unexplored. Here, we show that ER stress causes activation of the NLRP3 inflammasome, with subsequent release of the pro-inflammatory cytokine interleukin-1β. This ER-triggered proinflammatory signal shares the same requirement for reactive oxygen species production and potassium efflux compared with other known NLRP3 inflammasome activators, but is independent of the classical unfolded protein response (UPR). We thus propose that the NLRP3 inflammasome senses and responds to ER stress downstream of a previously uncharacterized ER stress response signaling pathway distinct from the UPR, thus providing mechanistic insight to the link between ER stress and chronic inflammatory diseases.
Figures
Figure 1
ER stress triggers the release of mature interleukin-1_β_ by macrophages. (a) PMA-differentiated THP-1 cells were stimulated with uric acid crystals (MSU, positive control), BFA, thapsigargin or tunicamycin for 6 h. Precipitated supernatant and whole-cell extracts were then analyzed by western blot. (b) Human primary macrophages were stimulated with MSU, BFA or tunicamycin for 6 h, and analyzed by western blot. IL-β, mature IL-1_β_; pIL-1_β_, proIL-1_β_; Casp1, mature caspase-1; pCasp1, procaspase-1; SN, supernatant; xT, whole-cell extract
Figure 2
ER stress specifically activates the NLRP3 inflammasome. (a–c) LPS-primed bone marrow-derived macrophages from Nlrp3+/+, Nlrp3_−/−, Asc+/+, Asc_−/−, Nlrc4+/+ and _Nlrc4_−/− mice, respectively, were stimulated with MSU, tunicamycin, thapsigargin or BFA as indicated for 6 h, and analyzed by western blot
Figure 3
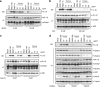
Mechanism of ER stress-induced inflammasome activation is similar to that of other known NLRP3 activators. (a, b and c) PMA-differentiated THP-1 cells were pre-incubated for 30 min with 50-_μ_M ammonium pyrrolidinedithiocarbamate, 130-mM extracellular KCl or 2-_μ_M cytochalasin D (Cyto D), respectively, stimulated with MSU, R837 or tunicamycin for a further 6 h, and analyzed by western blot. (d) PMA-differentiated THP-1 cells overexpressing an anti-VDAC1, anti-VDAC2 or scramble shRNA were stimulated with MSU or tunicamycin for 6 h, and analyzed by western blot
Figure 4
ER stress activates the NLRP3 inflammasome independently of the UPR. (a) PMA-differentiated THP-1 cells overexpressing an shRNA against IRE1_α_ or an empty vector were stimulated with MSU, BFA or R837 for 6 h, and analyzed by western blot. (b) PMA-differentiated THP-1 cells stably overexpressing a scramble or anti-PERK shRNA were stimulated with MSU or tunicamycin for 6 h, and analyzed by western blot. (c) LPS-primed BMDM isolated from Atf6α+/+ and _Atf6α_−/− mice were stimulated with MSU, thapsigargin or tunicamycin for 6 h, and analyzed by western blot. (d) PMA-differentiated THP-1 cells were pretreated with actinomycin D for 30 min, stimulated with R837 or BFA for 6 h, and analyzed by western blot
References
- Schroder K, Tschopp J. The inflammasomes. Cell. 2010;140:821–832. -PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources