The metabolic profile of tumors depends on both the responsible genetic lesion and tissue type - PubMed (original) (raw)
Comparative Study
. 2012 Feb 8;15(2):157-70.
doi: 10.1016/j.cmet.2011.12.015.
Teresa W M Fan, Thaddeus D Allen, Richard M Higashi, Dana V Ferraris, Takashi Tsukamoto, José M Matés, Francisco J Alonso, Chunmei Wang, Youngho Seo, Xin Chen, J Michael Bishop
Affiliations
- PMID: 22326218
- PMCID: PMC3282107
- DOI: 10.1016/j.cmet.2011.12.015
Comparative Study
The metabolic profile of tumors depends on both the responsible genetic lesion and tissue type
Mariia O Yuneva et al. Cell Metab. 2012.
Abstract
The altered metabolism of tumors has been considered a target for anticancer therapy. However, the relationship between distinct tumor-initiating lesions and anomalies of tumor metabolism in vivo has not been addressed. We report that MYC-induced mouse liver tumors significantly increase both glucose and glutamine catabolism, whereas MET-induced liver tumors use glucose to produce glutamine. Increased glutamine catabolism in MYC-induced liver tumors is associated with decreased levels of glutamine synthetase (Glul) and the switch from Gls2 to Gls1 glutaminase. In contrast to liver tumors, MYC-induced lung tumors display increased expression of both Glul and Gls1 and accumulate glutamine. We also show that inhibition of Gls1 kills cells that overexpress MYC and catabolize glutamine. Our results suggest that the metabolic profiles of tumors are likely to depend on both the genotype and tissue of origin and have implications regarding the design of therapies targeting tumor metabolism.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures
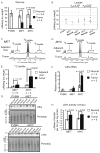
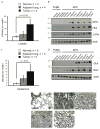
Figure 1. Lactate Levels are Significantly Increased in _MYC_-induced but not in _MET_-induced Murine Liver Tumors
(A and B) Glucose and lactate levels in tumor and normal liver tissue extracts measured by enzyme-based assays. Normal tissue was from wild type FVBN mice and transgenic mice kept on doxycycline. (C and D) Representative profiles of 1H NMR performed on extracts from tumors and adjacent liver tissues from animals injected with [U-13C]-glucose (number of animals analyzed is indicated in 1E). Two satellite peaks at 1.21 and 1.42 ppm represent fully-labeled 13C-lactate. The profiles were normalized to the initial dry weight of tissue. (E) Quantification of 13C-lactate concentration in the samples from 1C and 1D. #, p = 0.050 for _MET_-induced tumors vs. _MYC_-induced tumors. (F) Ldha RNA levels estimated by real-time PCR in the samples outlined in A and B. Mouse β-actin served as an internal control. One of the FVBN normal livers was used as a reference. (G) Ldha expression estimated by immunoblotting of whole tissue lysates of normal livers and tumors. Ponceau staining of membranes was used to confirm the protein loading. (H) LDH activity measured in the cytosolic fractions of tumors and normal livers from transgenic mice kept on doxycycline. All values are given as the mean ± s.d. See also Figure S1.
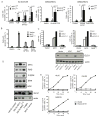
Figure 2. Glucose Levels are Controlled Differently in Tumors Induced by either MYC or MET
(A) PET/CT imaging of 18F-FDG uptake performed on normal and tumor-bearing animals. After imaging tumor presence was confirmed as demonstrated by photographs under each scan image. Tumors are indicated by arrows. Images of representative animals are shown. (B) 18F-FDG uptake quantified as described in Experimental Procedures. The normal group contained one FVBN mouse, and one each of mice carrying either MYC or MET transgenes and kept on doxycycline. *, p = 0.038 for MET vs. normal, **, p = 0.023 for MYC vs. normal and #, p = 0.036 for MET vs. MYC. (C and E) RNA levels for glucokinase (C) and Hk2 (E) measured in normal and tumor tissues using mouse β-actin as an internal control. Results are normalized as in Fig. 1F. Values are given as the mean ± s.d. (D) Glucokinase expression estimated in the cytosolic fractions of normal and tumor tissues by immunoblotting. Ponceau staining was used to confirm the protein loading. (F) Hk2 protein levels measured in whole tissue lysates from normal and tumor samples by immunoblotting using β-actin as a loading control. See also Figure S2 and Table S1.
Figure 3. Glutamine Metabolism is Different in Liver Tumors Induced by either MYC or MET
(A and B) Glutamine levels determined by NMR in tumors and normal livers from transgenic mice kept on doxycycline. Representative 1H NMR profiles of normal livers and tumors are shown (at least four animals per group). Glutamine peaks are in 2.42–2.47 ppm region and glutamate peaks are in 2.32–2.37 ppm region. The profiles were normalized to the initial dry weight. (C) Glutamine concentration quantified in normal livers and _MET_-induced tumors based on 1H-NMR data in B. (D) Expression of indicated proteins evaluated by immunoblotting of whole tissue lysates using β-actin as a loading control. (E) Gls1 RNA levels measured in normal and tumor tissues using mouse β-actin as an internal control. (F) Phosphate-dependent glutaminase activity measured in the mitochondrial fraction of _MYC_-induced tumors and normal livers from transgenic mice kept on doxycycline. All values are given as the mean ± s.d. (G) Detection of 13C-labeled glutamine by 1H-13C heteronuclear single quantum coherence spectroscopy (HSQC) NMR performed on extracts of tumor-adjacent and tumor tissues from animals injected with [U-13C]-glucose (5 animals per group). The representative HSQC spectra are shown. 13C-4-glutamine peak is at 2.45 ppm. Consistent with glutamine depletion a corresponding 13C-4-glutamine peak was not observed in _MYC_-induced tumors. The profiles were normalized to the initial dry weight of tissue. See also Figure S3.
Figure 4. Catabolism of Glucose and Glutamine Contributes to Increased Synthesis of Krebs Cycle Intermediates in _MYC_-induced Liver Tumors
(A and B) The levels of Krebs cycle intermediates measured in tissue extracts by GC-MS. Values are given as the mean ± s.d. (C and D) The representative spectra of 1H-13C HSQC NMR performed on extracts of tumor-adjacent and tumor tissues from animals injected with either [U-13C]-glucose (C) or [U-13C]-glutamine (D) (at least 3 animals per group). The profiles were normalized to the initial dry weight. Glc, glucose; G6P, glucose-6-phosphate; Glu, glutamate; GSH, reduced glutathione; GSSG, oxidized glutathione; Gln, glutamine; Asp, aspartate; Suc, succinate; Ala, alanine; Lac, lactate. See also Figures S4.
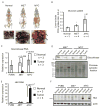
Figure 5. Metabolism of _MYC_-induced Lung Tumors Differs from Metabolism of _MYC_-induced Liver Tumors
(A) Lactate levels measured by 1H NMR in lung tumors induced by MYC (Tumor) and either adjacent lung lobes from the same animals (Adjacent lung) or normal lungs from mice with MYC transgene kept without doxycycline (Normal). Values are given as the mean ± s.d. (B and D) The levels of indicated proteins estimated by immunoblotting in whole tissue lysates using β-actin as a loading control. (C) Glutamine levels in tissue extracts determined by GC-MS. Values are given as the mean ± s.d. (E–I) Expression of Glul examined by immunochemistry in normal lung (E), _MYC_-induced tumors (F–H) and adjacent lung (I). Expression of Glul is apparent in bronchial epithelium of normal lung, in tumor cells and in infiltrating immune cells of tumor-adjacent lungs (indicated by arrows).
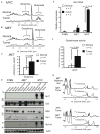
Figure 6. GLS1 Inhibitors Affect Glutamine Catabolism through the Krebs Cycle
(A) IMR90-ERMYC and HA1E-MYCER cells were incubated for 24 hours with either OHT (MYCOn) or 0.1% ethanol (MYCOff). Expression of GLUL, GLS2 and GLS1 was detected in whole lysates of these cells, normal liver and lung as well as of _MYC_-induced liver and lung tumors by immunoblotting using β-actin as a loading control. *, indicates cross-reactive band recognized by anti-Gls2 antibody in human cell lines. (B and C) IMR90-ERMYC cells with induced MYC activity were incubated with 2 mM [U-13C]-glutamine and indicated concentrations of BPTES for 12 hours. 0.1% DMSO was used as a vehicle control. Absolute concentrations (B) and 13C isotopologue enrichment (C) of indicated compounds were measured by GC-MS. In C metabolate+0 represents monoisotopic or unlabeled isotopologue while metabolite+1 to 5 are isotopologues with 1 to 5 13C atoms. The results were referenced to the initial dry weight of cell pellet. Average of two replicate samples is presented. The experiment was repeated twice. (D) IMR90-ERMYC cells with induced MYC were incubated with 2 mM 15N2-glutamine and indicated concentrations of BPTES, 20767 or DON. Incorporation of 15N into adenine nucleotides (AXP) was evaluated by 1H NMR. The profiles are referenced to the dry weight of cell pellet and the internal standard concentration. The representative profiles from two independent experiments are presented. See also Figure S5 and S6.
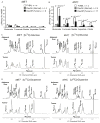
Figure 7. GLS1 Inhibitors Induce _MYC_-dependent Apoptosis
(A) MYC activity was induced in HA1E-MYCER and IMR90-ERMYC cells as described in Fig. 6A. Cells then were treated with indicated concentrations of BPTES or 20767 or with 5 mM DON. 0.1% DMSO was used as a vehicle control for the experiments with BPTES. HA1E-MYCER cells were treated for 36 hours and IMR90-ERMYC cells were treated for 48 hours and percentage of apoptotic nuclei was scored. The average of three independent experiments is presented ± s.d. (B) HA1E cells transduced with either empty vector or vectors carrying either MYC or H-RASV12 were either incubated in glutamine-free media or treated with indicated concentrations of BPTES for 24 hours. Apoptosis was scored in three independent replicate samples. The values are given as the mean ± s.d. A representative of two independent experiments is shown. (C) HA1E-derived cell lines were infected with lentiviruses expressing either a scrambled shRNA (shScr) or two independent shRNAs targeting GLS1 (shGLS1_1 and shGLS1_2). Cells were collected 36 or 48 hours after the infection, GLS1 expression was evaluated by immunoblotting, and apoptosis was scored by counting apoptotic nuclei. For each shRNA the experiment was repeated at least twice. Transduction with empty pLKO.1 vector (EV) was used to account for the exceptional level of cell death induced by lentiviral infection observed specifically in HA1E cells with ectopic expression of MYC. (D) The ectopic expression of MYC and H-RASV12, as well as expression of ERK, P-ERK and GLS1 was estimated by western blotting in whole cell lysates. Increased expression of PERK indicates increased activity of RAS. (E) Indicated Burkitt’s lymphoma cell lines were cultured in the presence of 150 μM 20767. Cell death was analyzed by Annexin V and PI staining. A representative of two independent experiments is shown.
Comment in
- METabolic adaptations in the tumor MYCroenvironment.
Davidson SM, Vander Heiden MG. Davidson SM, et al. Cell Metab. 2012 Feb 8;15(2):131-3. doi: 10.1016/j.cmet.2012.01.005. Cell Metab. 2012. PMID: 22326214
Similar articles
- Tumor suppressor NDRG2 inhibits glycolysis and glutaminolysis in colorectal cancer cells by repressing c-Myc expression.
Xu X, Li J, Sun X, Guo Y, Chu D, Wei L, Li X, Yang G, Liu X, Yao L, Zhang J, Shen L. Xu X, et al. Oncotarget. 2015 Sep 22;6(28):26161-76. doi: 10.18632/oncotarget.4544. Oncotarget. 2015. PMID: 26317652 Free PMC article. - Myc promotes glutaminolysis in human neuroblastoma through direct activation of glutaminase 2.
Xiao D, Ren P, Su H, Yue M, Xiu R, Hu Y, Liu H, Qing G. Xiao D, et al. Oncotarget. 2015 Dec 1;6(38):40655-66. doi: 10.18632/oncotarget.5821. Oncotarget. 2015. PMID: 26528759 Free PMC article. - GLS-driven glutamine catabolism contributes to prostate cancer radiosensitivity by regulating the redox state, stemness and ATG5-mediated autophagy.
Mukha A, Kahya U, Linge A, Chen O, Löck S, Lukiyanchuk V, Richter S, Alves TC, Peitzsch M, Telychko V, Skvortsov S, Negro G, Aschenbrenner B, Skvortsova II, Mirtschink P, Lohaus F, Hölscher T, Neubauer H, Rivandi M, Labitzky V, Lange T, Franken A, Behrens B, Stoecklein NH, Toma M, Sommer U, Zschaeck S, Rehm M, Eisenhofer G, Schwager C, Abdollahi A, Groeben C, Kunz-Schughart LA, Baretton GB, Baumann M, Krause M, Peitzsch C, Dubrovska A. Mukha A, et al. Theranostics. 2021 Jun 26;11(16):7844-7868. doi: 10.7150/thno.58655. eCollection 2021. Theranostics. 2021. PMID: 34335968 Free PMC article. - Rethinking the Warburg effect with Myc micromanaging glutamine metabolism.
Dang CV. Dang CV. Cancer Res. 2010 Feb 1;70(3):859-62. doi: 10.1158/0008-5472.CAN-09-3556. Epub 2010 Jan 19. Cancer Res. 2010. PMID: 20086171 Free PMC article. Review. - Therapeutic targeting of Myc-reprogrammed cancer cell metabolism.
Dang CV. Dang CV. Cold Spring Harb Symp Quant Biol. 2011;76:369-74. doi: 10.1101/sqb.2011.76.011296. Epub 2011 Sep 29. Cold Spring Harb Symp Quant Biol. 2011. PMID: 21960526 Review.
Cited by
- Stable isotope-resolved metabolomics analyses of metabolic phenotypes reveal variable glutamine metabolism in different patient-derived models of non-small cell lung cancer from a single patient.
Kinslow CJ, Ll MB, Cai Y, Yan J, Lorkiewicz PK, Al-Attar A, Tan J, Higashi RM, Lane AN, Fan TW. Kinslow CJ, et al. Metabolomics. 2024 Jul 27;20(4):87. doi: 10.1007/s11306-024-02126-x. Metabolomics. 2024. PMID: 39068202 - Molecular Pathways: Targeting MYC-induced metabolic reprogramming and oncogenic stress in cancer.
Li B, Simon MC. Li B, et al. Clin Cancer Res. 2013 Nov 1;19(21):5835-41. doi: 10.1158/1078-0432.CCR-12-3629. Epub 2013 Jul 29. Clin Cancer Res. 2013. PMID: 23897900 Free PMC article. Review. - Divergent in vitro/in vivo responses to drug treatments of highly aggressive NIH-Ras cancer cells: a PET imaging and metabolomics-mass-spectrometry study.
Gaglio D, Valtorta S, Ripamonti M, Bonanomi M, Damiani C, Todde S, Negri AS, Sanvito F, Mastroianni F, Di Campli A, Turacchio G, Di Grigoli G, Belloli S, Luini A, Gilardi MC, Colangelo AM, Alberghina L, Moresco RM. Gaglio D, et al. Oncotarget. 2016 Aug 9;7(32):52017-52031. doi: 10.18632/oncotarget.10470. Oncotarget. 2016. PMID: 27409831 Free PMC article. - Mechanisms and Implications of Metabolic Heterogeneity in Cancer.
Kim J, DeBerardinis RJ. Kim J, et al. Cell Metab. 2019 Sep 3;30(3):434-446. doi: 10.1016/j.cmet.2019.08.013. Cell Metab. 2019. PMID: 31484055 Free PMC article. Review. - Combined thioredoxin reductase and glutaminase inhibition exerts synergistic anti-tumor activity in MYC-high high-grade serous ovarian carcinoma.
Raninga PV, He Y, Datta KK, Lu X, Maheshwari UR, Venkat P, Mayoh C, Gowda H, Kalimutho M, Hooper JD, Khanna KK. Raninga PV, et al. Mol Ther. 2023 Mar 1;31(3):729-743. doi: 10.1016/j.ymthe.2022.12.011. Epub 2022 Dec 21. Mol Ther. 2023. PMID: 36560881 Free PMC article.
References
- Abcouwer SF, Lukaszewicz GC, Souba WW. Glucocorticoids regulate glutamine synthetase expression in lung epithelial cells. Am J Physiol. 1996;270:L141–151. - PubMed
- Baggetto LG. Deviant energetic metabolism of glycolytic cancer cells. Biochimie. 1992;74:959–974. - PubMed
- Bode BP. Recent molecular advances in mammalian glutamine transport. J Nutr. 2001;131:2475S–2485S. discussion 2486S–2477S. - PubMed
- Bustamante E, Morris HP, Pedersen PL. Energy metabolism of tumor cells. Requirement for a form of hexokinase with a propensity for mitochondrial binding. J Biol Chem. 1981;256:8699–8704. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P20RR018733/RR/NCRR NIH HHS/United States
- R21 NS074151/NS/NINDS NIH HHS/United States
- 1R01CA118434-01A2/CA/NCI NIH HHS/United States
- 3R01 CA118434-02S1/CA/NCI NIH HHS/United States
- P20 RR018733/RR/NCRR NIH HHS/United States
- R35 CA044338/CA/NCI NIH HHS/United States
- R21NS074151/NS/NINDS NIH HHS/United States
- R01 CA118434/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous