ZFX controls the self-renewal of human embryonic stem cells - PubMed (original) (raw)
ZFX controls the self-renewal of human embryonic stem cells
Sivan Harel et al. PLoS One. 2012.
Abstract
Embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs) offer great promise in regenerative medicine and disease modeling due to their unlimited self-renewal and broad differentiation capacity. There is evidence that the growth properties and critical signaling pathways differ between murine and human ESCs; therefore, it is essential to perform functional studies to test the putatively conserved mechanisms of pluripotent stem cell self-renewal between species. Previously, we identified the transcription factor Zfx as a key regulator of self-renewal in murine ESCs. Here we extend those findings to human ESCs. ZFX knockdown in hESCs hindered clonal growth and decreased colony size after serial replating. ZFX overexpression enhanced clone formation in the presence of Y-27632, increased colony size at low density and decreased expression of differentiation-related genes in human ESCs. ZFX-overexpressing hESCs resisted spontaneous differentiation but could be directed to differentiate into endodermal and neural cell fates when provided with the appropriate cues. Thus, ZFX acts as a molecular rheostat regulating the balance between self-renewal and differentiation in hESCs, revealing the close evolutionary conservation of the self-renewal mechanisms in murine and human ESCs.
Conflict of interest statement
Competing Interests: The authors have read the journal’s policy and have the following conflicts: CTC owns stock in Becton, Dickinson and Company. This does not alter the authors’ adherence to all the PLoS ONE policies on sharing data and materials.
Figures
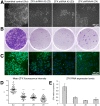
Figure 1. ZFX knockdown impairs hESC colony size.
Human ESCs were transduced with ZFX knockdown lentiviral constructs and a scrambled control before clonal replating. (A) Live cell images and (B) and the entire well stained with crystal violet seven days after replating. (C) ZFX immunofluorescence after knockdown in hESCs, and (D) ZFX quantitative immunofluorescence analysis. Each dot is the average pixel intensity of nuclear ZFX protein averaged from all cells in one microscopic field. The average pixel intensity from 50 microscopic fields derived from 3 independent experiments is shown. The crosshairs and whiskers represent the mean and SEM. All knockdowns were significantly different from the scrambled control (p<0.0001 for t-tests of each knockdown versus scrambled). 11,803 (Scr), 10,296 (Z2), 4,765 (Z3) and 6,905 (Z4) nuclei were measured in these experiments. (E) ZFX RNA levels as measured by quantitative PCR after knockdown.
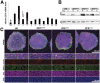
Figure 2. ZFX BAC transgenic human embryonic stem cells.
A. Normalized ZFX expression levels from G418-resistant hESC clones with the original clone names shown on the x-axis. Three clones were renamed ZFXOver1, ZFXOver2 and ZFXNormal were selected for further analysis. ZFXNormal showed normal levels of ZFX but had undergone the same clonal and selection steps as ZFXOver clones. B. ZFX and actin protein levels determined by Western blot analysis of ZFXOver clones and controls. C. Oct4 (purple), SSEA-4 (green), Tra1-81 (red) and DAPI (blue) on ZFXOver clones and controls. Scale bar = 500 µm (low) or 100 µm on the high magnification images.
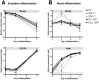
Figure 3. ZFXOver cultures have a higher clonogenic capacity.
A. ZFXOver clones, ZFXNormal and H9 hESCs (together grouped as controls) were dissociated into single cells before clonal replating. Cells were expanded for 10 days before fixation and staining with crystal violet. B. Colony counts between ZFXOver and control hESCs in 3 independent experiments with error bars representing the S.E.M.
Figure 4. ZFXOver clones resist spontaneous differentiation.
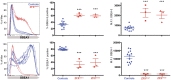
A. ZFXOver clones and controls were expanded in conditions promoting self-renewal before SSEA-3 and SSEA-1 FACS analysis. B. ZFXOver clones and controls were expanded in suboptimal conditions before SSEA-3 and SSEA-1 FACS analysis. The quantitation compares ZFXOver clones to controls in three independent experiments and error bars represent the S.E.M. See Figure S5 for all 3 independent experiments.
Figure 5. Directed differentiation of ZFXOver clones to endoderm and neural tissue.
ZFXOver clones, two control clones that express normal ZFX levels (ID1::YFPc2, Dll1::GFPc277) and H9 were directed to endoderm or neural cells, and the level of Nanog, Pax6 (neural) and CXCR4 (endoderm) mRNA at each time point was measured by quantitative PCR.
Figure 6. Gene expression analysis of ZFXOver clones compared to controls.
Two independent samples of Tra1-81HI/SSEA-3HI hESCs were isolated from ZFXOver clones, ZFXNormal and H9 hESC array analysis. A. Dendrograms of each cell line after clustering analysis. B. Up- and down-regulated genes in ZFXOver compared to H9 using an adjusted p-value of 0.05 as a cutoff. C. Quantitative PCR validation of selected genes on the array.
Similar articles
- Zfx controls the self-renewal of embryonic and hematopoietic stem cells.
Galan-Caridad JM, Harel S, Arenzana TL, Hou ZE, Doetsch FK, Mirny LA, Reizis B. Galan-Caridad JM, et al. Cell. 2007 Apr 20;129(2):345-57. doi: 10.1016/j.cell.2007.03.014. Cell. 2007. PMID: 17448993 Free PMC article. - Retinoic acid maintains self-renewal of murine embryonic stem cells via a feedback mechanism.
Wang R, Liang J, Yu HM, Liang H, Shi YJ, Yang HT. Wang R, et al. Differentiation. 2008 Nov;76(9):931-45. doi: 10.1111/j.1432-0436.2008.00272.x. Epub 2008 Jul 1. Differentiation. 2008. PMID: 18637026 - Transcription elongation factor Tcea3 regulates the pluripotent differentiation potential of mouse embryonic stem cells via the Lefty1-Nodal-Smad2 pathway.
Park KS, Cha Y, Kim CH, Ahn HJ, Kim D, Ko S, Kim KH, Chang MY, Ko JH, Noh YS, Han YM, Kim J, Song J, Kim JY, Tesar PJ, Lanza R, Lee KA, Kim KS. Park KS, et al. Stem Cells. 2013 Feb;31(2):282-92. doi: 10.1002/stem.1284. Stem Cells. 2013. PMID: 23169579 Free PMC article. - Genomic studies to explore self-renewal and differentiation properties of embryonic stem cells.
Zhan M. Zhan M. Front Biosci. 2008 Jan 1;13:276-83. doi: 10.2741/2678. Front Biosci. 2008. PMID: 17981546 Review. - The role of Ca(2+) signaling on the self-renewal and neural differentiation of embryonic stem cells (ESCs).
Hao B, Webb SE, Miller AL, Yue J. Hao B, et al. Cell Calcium. 2016 Mar;59(2-3):67-74. doi: 10.1016/j.ceca.2016.01.004. Epub 2016 Feb 27. Cell Calcium. 2016. PMID: 26973143 Review.
Cited by
- A flexible repertoire of transcription factor binding sites and a diversity threshold determines enhancer activity in embryonic stem cells.
Singh G, Mullany S, Moorthy SD, Zhang R, Mehdi T, Tian R, Duncan AG, Moses AM, Mitchell JA. Singh G, et al. Genome Res. 2021 Apr;31(4):564-575. doi: 10.1101/gr.272468.120. Epub 2021 Mar 12. Genome Res. 2021. PMID: 33712417 Free PMC article. - Transcriptomic profiling of neonatal mouse granulosa cells reveals new insights into primordial follicle activation†.
Ford EA, Frost ER, Beckett EL, Roman SD, McLaughlin EA, Sutherland JM. Ford EA, et al. Biol Reprod. 2022 Mar 19;106(3):503-514. doi: 10.1093/biolre/ioab193. Biol Reprod. 2022. PMID: 34673933 Free PMC article. - ZFX knockdown inhibits growth and migration of non-small cell lung carcinoma cell line H1299.
Li K, Zhu ZC, Liu YJ, Liu JW, Wang HT, Xiong ZQ, Shen X, Hu ZL, Zheng J. Li K, et al. Int J Clin Exp Pathol. 2013 Sep 15;6(11):2460-7. eCollection 2013. Int J Clin Exp Pathol. 2013. PMID: 24228108 Free PMC article. - Epigenetic control of metabolic identity across cell types.
Pacheco MP, Gerard D, Mangan RJ, Chapman AR, Hecker D, Kellis M, Schulz MH, Sinkkonen L, Sauter T. Pacheco MP, et al. bioRxiv [Preprint]. 2024 Dec 17:2024.07.24.604914. doi: 10.1101/2024.07.24.604914. bioRxiv. 2024. PMID: 39091778 Free PMC article. Preprint. - The Transcription Factor Zfx Regulates Peripheral T Cell Self-Renewal and Proliferation.
Smith-Raska MR, Arenzana TL, D'Cruz LM, Khodadadi-Jamayran A, Tsirigos A, Goldrath AW, Reizis B. Smith-Raska MR, et al. Front Immunol. 2018 Jul 4;9:1482. doi: 10.3389/fimmu.2018.01482. eCollection 2018. Front Immunol. 2018. PMID: 30022979 Free PMC article.
References
- Evans MJ, Kaufman MH (1981) Establishment in culture of pluripotential cells from mouse embryos. Nature 292: 154–156. - PubMed
- Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, et al. (1998) Embryonic stem cell lines derived from human blastocysts. Science 282: 1145–1147. - PubMed
- Do JT, Scholer HR (2009) Regulatory circuits underlying pluripotency and reprogramming. Trends Pharmacol Sci 30: 296–302. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources