Thyroid hormone reduces cholesterol via a non-LDL receptor-mediated pathway - PubMed (original) (raw)
Thyroid hormone reduces cholesterol via a non-LDL receptor-mediated pathway
Ira J Goldberg et al. Endocrinology. 2012 Nov.
Abstract
Although studies in vitro and in hypothyroid animals show that thyroid hormone can, under some circumstances, modulate the actions of low-density lipoprotein (LDL) receptors, the mechanisms responsible for thyroid hormone's lipid-lowering effects are not completely understood. We tested whether LDL receptor (LDLR) expression was required for cholesterol reduction by treating control and LDLR-knockout mice with two forms of thyroid hormone T(3) and 3,5-diiodo-l-thyronine. High doses of both 3,5-diiodo-l-thyronine and T(3) dramatically reduced circulating total and very low-density lipoprotein/LDL cholesterol (∼70%) and were associated with reduced plasma T(4) level. The cholesterol reduction was especially evident in the LDLR-knockout mice. Circulating levels of both apolipoprotein B (apo)B48 and apoB100 were decreased. Surprisingly, this reduction was not associated with increased protein or mRNA expression of the hepatic lipoprotein receptors LDLR-related protein 1 or scavenger receptor-B1. Liver production of apoB was markedly reduced, whereas triglyceride production was increased. Thus, thyroid hormones reduce apoB lipoproteins via a non-LDLR pathway that leads to decreased liver apoB production. This suggests that drugs that operate in a similar manner could be a new therapy for patients with genetic defects in the LDLR.
Figures
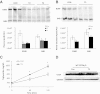
Fig. 1.
Plasma apoB distribution, apoB, triglyceride production in _Ldlr_−/− mice and Western blot analysis of LDLR in WT mice. A, Fasting plasma apoB100 and B48. Plasma (1 μl) proteins from _Ldlr_−/− mice were separated in 4% SDS-PAGE and stained with Coomassie blue. Representative samples are shown on top, and average scanned densities of each protein shown in arbitrary units (AU) are shown on the bottom. B, Representative autoradiogram of plasma showing newly synthesized apoB at the 120-min time point. _Ldlr_−/− mice were treated for 1 wk with vehicle or T3 by gavage, injected with [35S]methionine, and Triton WR1339 and plasma (1 μl) containing liver-secreted apoB was subjected to SDS-PAGE analysis. Average scanned densities (AU) of each protein are shown on bottom. C, Triglyceride production in Triton WR1339-treated _Ldlr_−/− mice receiving control vehicle or T3 by gavage for 1 wk. P values: *, P < 0.05; and **, P < 0.01. Con, Control. D, WT C57BL/6 mice were placed on a Western diet for 1 wk and then maintained on the diet and treated with T3, 0.75 mg/kg, by daily gavage for 1 wk. LDLR were detected by Western blot analysis. GAPDH, Glyceraldehyde 3-phosphate dehydrogenase.
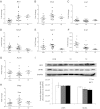
Fig. 2.
Hepatic gene expression and Western blot analysis of lipoprotein receptors in control and thyroid hormone-treated _Ldlr_−/− mice. Male mice were fed a Western diet for 1 wk and then received a daily gavage of control (Con) or thyroid hormone-containing vehicle. After 1 wk the mice were killed, and livers were collected. Data are from mice receiving the highest dose of T3 (0.75 mg/kg) or T2 (12.5 mg/kg). A–H, Liver mRNA levels were assessed by real-time PCR analysis. Me1, Malic enzyme 1; Me2, malic enzyme 2; Lrp1, LDLR-related protein 1; Sdc1, proteoglycan syndecan 1; Ndst1, _N_-deacetylase/_N_-sulfotransferase 1; Srb1, scavenger receptor B-1; Apob, apolipoprotein B; Mttp, microsomal triglyceride transfer protein. Gene expression was normalized to 18S RNA and expressed as fold change of control. I, Hepatic lipoprotein receptors (LRP1 and SR-B1) were analyzed by Western blot analysis. Representative autoradiograms are shown on top, and average scanned densities of each protein [normalized to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and shown in arbitrary units (AU)] are shown on the bottom.
References
- Mason RL, Hunt HM, Hurzthal L. 1930. Blood cholesterol values in hyperthyroidism and hypothyroidism-their significance. N Engl J Med 26:1273–1278
- Pramfalk C, Pedrelli M, Parini P. 2011. Role of thyroid receptor β in lipid metabolism. Biochim Biophys Acta 1812:929–937 - PubMed
- Faber J, Kirkegaard C, Lumholtz IB, Siersbaek-Nielsen K, Friis T. 1982. Simultaneous measurement of 3,5-diiodothyronine and 3,5,3′-triiodothyronine turnover kinetics in euthyroid hyperthyroid, and hypothyroid subjects. J Clin Endocrinol Metab 55:8–12 - PubMed
- McClure J, De Mowbray RR, Gilliland IC. 1961. The effect of 3:5-diiodo-D-thyronine on serum cholesterol. J Endocrinol 22:87–93 - PubMed
- Goglia F. 2005. Biological effects of 3,5-diiodothyronine (T2). Biochemistry (Mosc) 70:164–172 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R21 HL112233/HL/NHLBI NIH HHS/United States
- R01 HL045095/HL/NHLBI NIH HHS/United States
- AA020561/AA/NIAAA NIH HHS/United States
- R37 HL045095/HL/NHLBI NIH HHS/United States
- R21 AA020561/AA/NIAAA NIH HHS/United States
- HL45095/HL/NHLBI NIH HHS/United States
- HL112233/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous