Multidrug resistant pulmonary tuberculosis treatment regimens and patient outcomes: an individual patient data meta-analysis of 9,153 patients - PubMed (original) (raw)
Meta-Analysis
doi: 10.1371/journal.pmed.1001300. Epub 2012 Aug 28.
David Ashkin, Monika Avendano, Rita Banerjee, Melissa Bauer, Jamie N Bayona, Mercedes C Becerra, Andrea Benedetti, Marcos Burgos, Rosella Centis, Eward D Chan, Chen-Yuan Chiang, Helen Cox, Lia D'Ambrosio, Kathy DeRiemer, Nguyen Huy Dung, Donald Enarson, Dennis Falzon, Katherine Flanagan, Jennifer Flood, Maria L Garcia-Garcia, Neel Gandhi, Reuben M Granich, Maria G Hollm-Delgado, Timothy H Holtz, Michael D Iseman, Leah G Jarlsberg, Salmaan Keshavjee, Hye-Ryoun Kim, Won-Jung Koh, Joey Lancaster, Christophe Lange, Wiel C M de Lange, Vaira Leimane, Chi Chiu Leung, Jiehui Li, Dick Menzies, Giovanni B Migliori, Sergey P Mishustin, Carole D Mitnick, Masa Narita, Philly O'Riordan, Madhukar Pai, Domingo Palmero, Seung-kyu Park, Geoffrey Pasvol, Jose Peña, Carlos Pérez-Guzmán, Maria I D Quelapio, Alfredo Ponce-de-Leon, Vija Riekstina, Jerome Robert, Sarah Royce, H Simon Schaaf, Kwonjune J Seung, Lena Shah, Tae Sun Shim, Sonya S Shin, Yuji Shiraishi, José Sifuentes-Osornio, Giovanni Sotgiu, Matthew J Strand, Payam Tabarsi, Thelma E Tupasi, Robert van Altena, Martie Van der Walt, Tjip S Van der Werf, Mario H Vargas, Pirett Viiklepp, Janice Westenhouse, Wing Wai Yew, Jae-Joon Yim; Collaborative Group for Meta-Analysis of Individual Patient Data in MDR-TB
Affiliations
- PMID: 22952439
- PMCID: PMC3429397
- DOI: 10.1371/journal.pmed.1001300
Meta-Analysis
Multidrug resistant pulmonary tuberculosis treatment regimens and patient outcomes: an individual patient data meta-analysis of 9,153 patients
Shama D Ahuja et al. PLoS Med. 2012.
Abstract
Background: Treatment of multidrug resistant tuberculosis (MDR-TB) is lengthy, toxic, expensive, and has generally poor outcomes. We undertook an individual patient data meta-analysis to assess the impact on outcomes of the type, number, and duration of drugs used to treat MDR-TB.
Methods and findings: Three recent systematic reviews were used to identify studies reporting treatment outcomes of microbiologically confirmed MDR-TB. Study authors were contacted to solicit individual patient data including clinical characteristics, treatment given, and outcomes. Random effects multivariable logistic meta-regression was used to estimate adjusted odds of treatment success. Adequate treatment and outcome data were provided for 9,153 patients with MDR-TB from 32 observational studies. Treatment success, compared to failure/relapse, was associated with use of: later generation quinolones, (adjusted odds ratio [aOR]: 2.5 [95% CI 1.1-6.0]), ofloxacin (aOR: 2.5 [1.6-3.9]), ethionamide or prothionamide (aOR: 1.7 [1.3-2.3]), use of four or more likely effective drugs in the initial intensive phase (aOR: 2.3 [1.3-3.9]), and three or more likely effective drugs in the continuation phase (aOR: 2.7 [1.7-4.1]). Similar results were seen for the association of treatment success compared to failure/relapse or death: later generation quinolones, (aOR: 2.7 [1.7-4.3]), ofloxacin (aOR: 2.3 [1.3-3.8]), ethionamide or prothionamide (aOR: 1.7 [1.4-2.1]), use of four or more likely effective drugs in the initial intensive phase (aOR: 2.7 [1.9-3.9]), and three or more likely effective drugs in the continuation phase (aOR: 4.5 [3.4-6.0]).
Conclusions: In this individual patient data meta-analysis of observational data, improved MDR-TB treatment success and survival were associated with use of certain fluoroquinolones, ethionamide, or prothionamide, and greater total number of effective drugs. However, randomized trials are urgently needed to optimize MDR-TB treatment. Please see later in the article for the Editors' Summary.
Conflict of interest statement
JR is a Consultant for bioMérieux. WWY has been indirectly sponsored to participate in International Conferences by GlaxoSmithKline and Pfizer in the last 3 years. CDM is on the Scientific Advisory Board for Otsuka pharmaceuticals development of OPC67683 (Delaminid), a new anti-TB compound. SK received salary support from the Eli Lilly Foundation as part of funding for the activities of Partners In Health by the Foundation's MDR-TB Partnership. This funder was not involved in the study design; collection, analysis and interpretation of data; writing of the paper; and/or decision to submit for publication. The Partners In Health project in Tomsk received funding from Mr. Tom White, the Open Society Institute, the Bill and Melinda Gates Foundation, and the Global Fund to fight AIDS, Tuberculosis and Malaria. None of these funders were involved in the study design; collection, analysis and interpretation of data; writing of the paper; and/or decision to submit for publication. KD is an unpaid, volunteer member of the New Diagnostics Working Group (NDWG), formed of members of the Stop TB Partnership. The Secretariat of the NDWG is hosted by FIND (Foundation for New Innovative Diagnostics). JB was working as consultant for Otsuka Pharmaceutical for the implementation of clinical trial in Peru. JB was co PI of a NIH grant in Peru, Epidemiology of Tuberculosis. MP and GP are members of the Editorial Board of PLOS Medicine. All other authors have declared that no competing interests exist.
Figures
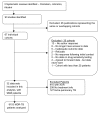
Figure 1. Study selection.
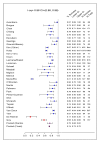
Figure 2. Treatment success versus failure and relapse and death.
Crude treatment success versus failure or relapse or death by study with exact 95% CI, as well as number of subjects with success and number of subjects treated. Fixed and der Simonian and Laird random effects pooled estimates are given (purple dots). Two studies that used only first-line TB drugs are indicated by a red square.
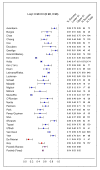
Figure 3. Treatment success versus failure and relapse and death.
Fixed and der Simonian and Laird random effects pooled estimates are given (purple dots). Two studies that used only first-line TB drugs are indicated by a red square.
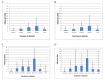
Figure 4. Association of treatment success with duration (adjusted odds and upper bound of CI shown).
(A) Duration of initial intensive phase in all patients (reference group 1.0–2.5 mo). (B) Duration of initial intensive phase—restricted to patients not previously treated with second-line drugs (reference group 1.0–2.5 mo). (C) Total duration of therapy in all patients (reference group is 6.0–12.5 mo). Patients receiving therapy for less than 6 or more than 36 mo excluded from analysis. Note: For duration of 24.6–27.5 mo the upper limit of the CI was 30.2. This is truncated at 21. (D) Total duration of therapy—analysis restricted to patients not previously treated with second-line drugs (reference group is 6.0–12.5 mo. Patients receiving therapy for less than 6 or more than 36 mo excluded from analysis). Note: For duration of 24.6–27.5 mo, the upper limit of the CI was 56.5. This is truncated at 21.
References
- World Health Organization (2008) Guidelines for the programmatic management of drug resistant tuberculosis - emergency update. World Health Organization 247 p.
- Caminero JA, Sotgiu G, Zumla A, Migliori GB (2010) Best drug treatment for multidrug-resistant and extensively drug-resistant tuberculosis. Lancet 10: 621–629. -PubMed
- Akcakir Y (2010) Correlates of treatment outcomes of multidrug-resistant tuberculosis (MDR-TB): a systematic review and meta-analysis [PhD dissertation]. Montreal: Department of Epidemiology & Biostatistics, McGill University. 110 p.
- Orenstein EW, Basu S, Shah NS, Andrews JR, Friedland GH, et al. (2009) Treatment outcomes among patients with multidrug-resistant tuberculosis:systematic review and meta-analysis. Lancet Infect Dis 9: 153–161. -PubMed
- Johnston JC, Shahidi NC, Sadatsafavi M, FitzGerald JM (2009) Treatment outcomes of multidrug-resistant tuberculosis: a systematic review and meta-analysis. PLoS One 4: e6914 doi:10.1371/journal.pone.0006914. -DOI -PMC -PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 176W009/WT_/Wellcome Trust/United Kingdom
- K01TW000001/TW/FIC NIH HHS/United States
- R01 AI089349/AI/NIAID NIH HHS/United States
- R01 AI087465/AI/NIAID NIH HHS/United States
- WT_/Wellcome Trust/United Kingdom
- K01 TW000001/TW/FIC NIH HHS/United States
- 55000632/HHMI/Howard Hughes Medical Institute/United States
- A135969/PHS HHS/United States
- CAPMC/ CIHR/Canada