Improving adherence to web-based cessation programs: a randomized controlled trial study protocol - PubMed (original) (raw)
Randomized Controlled Trial
Improving adherence to web-based cessation programs: a randomized controlled trial study protocol
Amanda L Graham et al. Trials. 2013.
Abstract
Background: Reducing smoking prevalence is a public health priority that can save more lives and money than almost any other known preventive intervention. Internet interventions have the potential for enormous public health impact given their broad reach and effectiveness. However, most users engage only minimally with even the best designed websites, diminishing their impact due to an insufficient 'dose'. Two approaches to improve adherence to Internet cessation programs are integrating smokers into an online social network and providing free nicotine replacement therapy (NRT). Active participation in online communities is associated with higher rates of cessation. Integrating smokers into an online social network can increase support and may also increase utilization of cessation tools and NRT. Removing barriers to NRT may increase uptake and adherence, and may also increase use of online cessation tools as smokers look for information and support while quitting. The combination of both strategies may exert the most powerful effects on adherence compared to either strategy alone.
Methods/design: This study compares the efficacy of a smoking cessation website (WEB) alone and in conjunction with free NRT and a social network (SN) protocol designed to integrate participants into the online community. Using a 2 (SN, no SN) x 2 (NRT, no NRT) randomized, controlled factorial design with repeated measures at baseline, 3 months, and 9 months, this study will recruit N = 4,000 new members of an internet cessation program and randomize them to: 1) WEB, 2) WEB + SN, 3) WEB + NRT, or 4) WEB + SN + NRT. Hypotheses are that all interventions will outperform WEB and that WEB + SN + NRT will outperform WEB + NRT and WEB + SN on 30-day point prevalence abstinence at 9 months. Exploratory analyses will examine theory-driven hypotheses about the mediators and moderators of outcome.
Discussion: Addressing adherence in internet cessation programs is critical and timely to leverage their potential public health impact. This study is innovative in its use of a social network approach to improve behavioral and pharmacological treatment utilization to improve cessation. This approach is significant for reducing tobacco's devastating disease burden and for optimizing behavior change in other arenas where adherence is just as critical.
Trial registration: ISRCTN:ISRCTN45127327.
Figures
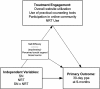
Figure 1
Research model. Theoretical model depicting the intervention components, main outcome variable, and mediating variables.
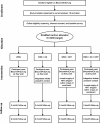
Figure 2
Study procedures. Flowchart of study procedures from enrollment through follow-up data collection.
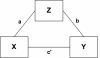
Figure 3
Mediational model. Conceptual diagram depicting the intervention (X), outcome (Y), and mediator (Z) variables as well as theory (a), action (b), and outcome (c’) pathways which will be examined in mediational analyses.
Similar articles
- Improving Adherence to Smoking Cessation Treatment: Smoking Outcomes in a Web-based Randomized Trial.
Graham AL, Papandonatos GD, Cha S, Erar B, Amato MS. Graham AL, et al. Ann Behav Med. 2018 Mar 15;52(4):331-341. doi: 10.1093/abm/kax023. Ann Behav Med. 2018. PMID: 29878062 Free PMC article. Clinical Trial. - Improving Adherence to Smoking Cessation Treatment: Intervention Effects in a Web-Based Randomized Trial.
Graham AL, Papandonatos GD, Cha S, Erar B, Amato MS, Cobb NK, Niaura RS, Abrams DB. Graham AL, et al. Nicotine Tob Res. 2017 Mar 1;19(3):324-332. doi: 10.1093/ntr/ntw282. Nicotine Tob Res. 2017. PMID: 27836982 Free PMC article. Clinical Trial. - Effect of nicotine replacement therapy on quitting by young adults in a trial comparing cessation services.
Buller DB, Halperin A, Severson HH, Borland R, Slater MD, Bettinghaus EP, Tinkelman D, Cutter GR, Woodall WG. Buller DB, et al. J Public Health Manag Pract. 2014 Mar-Apr;20(2):E7-E15. doi: 10.1097/PHH.0b013e3182a0b8c7. J Public Health Manag Pract. 2014. PMID: 24458316 Free PMC article. Clinical Trial. - Interventions for Tobacco Cessation in Adults, Including Pregnant Women: An Evidence Update for the U.S. Preventive Services Task Force [Internet].
Patnode CD, Henderson JT, Melnikow J, Coppola EL, Durbin S, Thomas R. Patnode CD, et al. Rockville (MD): Agency for Healthcare Research and Quality (US); 2021 Jan. Report No.: 20-05264-EF-1. Rockville (MD): Agency for Healthcare Research and Quality (US); 2021 Jan. Report No.: 20-05264-EF-1. PMID: 33523610 Free Books & Documents. Review. - Nicotine replacement therapy for smoking cessation.
Stead LF, Perera R, Bullen C, Mant D, Hartmann-Boyce J, Cahill K, Lancaster T. Stead LF, et al. Cochrane Database Syst Rev. 2012 Nov 14;11:CD000146. doi: 10.1002/14651858.CD000146.pub4. Cochrane Database Syst Rev. 2012. PMID: 23152200 Updated. Review.
Cited by
- Improving Adherence to Smoking Cessation Treatment: Smoking Outcomes in a Web-based Randomized Trial.
Graham AL, Papandonatos GD, Cha S, Erar B, Amato MS. Graham AL, et al. Ann Behav Med. 2018 Mar 15;52(4):331-341. doi: 10.1093/abm/kax023. Ann Behav Med. 2018. PMID: 29878062 Free PMC article. Clinical Trial. - Study protocol for a non-inferiority trial of a blended smoking cessation treatment versus face-to-face treatment (LiveSmokefree-Study).
Siemer L, Pieterse ME, Brusse-Keizer MG, Postel MG, Ben Allouch S, Sanderman R. Siemer L, et al. BMC Public Health. 2016 Nov 24;16(1):1187. doi: 10.1186/s12889-016-3851-x. BMC Public Health. 2016. PMID: 27881108 Free PMC article. Clinical Trial. - Young Adult Utilization of a Smoking Cessation Website: An Observational Study Comparing Young and Older Adult Patterns of Use.
Cantrell J, Ilakkuvan V, Graham AL, Richardson A, Xiao H, Mermelstein RJ, Curry SJ, Sporer AK, Vallone DM. Cantrell J, et al. JMIR Res Protoc. 2016 Jul 11;5(3):e142. doi: 10.2196/resprot.4881. JMIR Res Protoc. 2016. PMID: 27401019 Free PMC article. - Exposure to positive peer sentiment about nicotine replacement therapy in an online smoking cessation community is associated with NRT use.
Pearson JL, Amato MS, Papandonatos GD, Zhao K, Erar B, Wang X, Cha S, Cohn AM, Graham AL. Pearson JL, et al. Addict Behav. 2018 Dec;87:39-45. doi: 10.1016/j.addbeh.2018.06.022. Epub 2018 Jun 19. Addict Behav. 2018. PMID: 29940390 Free PMC article. - A prospective examination of online social network dynamics and smoking cessation.
Graham AL, Zhao K, Papandonatos GD, Erar B, Wang X, Amato MS, Cha S, Cohn AM, Pearson JL. Graham AL, et al. PLoS One. 2017 Aug 23;12(8):e0183655. doi: 10.1371/journal.pone.0183655. eCollection 2017. PLoS One. 2017. PMID: 28832621 Free PMC article.
References
- Centers for Disease Control and Prevention. Smoking-attributable mortality, years of potential life lost, and productivity losses--United States, 2000–2004. MMWR Morb Mortal Wkly Rep. 2008;57:1226–1228. - PubMed
- Centers for Disease Control and Prevention. Early Release of Selected Estimates Based on Data From the January-June 2009 National Health Interview Survey. http://www.cdc.gov/nchs/data/nhis/earlyrelease/earlyrelease200912.pdf.
- Institute of Medicine. Ending the Tobacco Problem: A Blueprint for the Nation. Washington, DC: The National Academies Press; 2007.
- National Tobacco Cessation Collaborative. Innovations in Building Consumer Demand for Tobacco Cessation Products and Services. http://www.consumer-demand.org/national_pdf/ConsumerDemandReportConferen.... - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical