Dietary carnosine prevents early atherosclerotic lesion formation in apolipoprotein E-null mice - PubMed (original) (raw)
Comparative Study
Dietary carnosine prevents early atherosclerotic lesion formation in apolipoprotein E-null mice
Oleg A Barski et al. Arterioscler Thromb Vasc Biol. 2013 Jun.
Abstract
Objective: Atherosclerotic lesions are associated with the accumulation of reactive aldehydes derived from oxidized lipids. Although inhibition of aldehyde metabolism has been shown to exacerbate atherosclerosis and enhance the accumulation of aldehyde-modified proteins in atherosclerotic plaques, no therapeutic interventions have been devised to prevent aldehyde accumulation in atherosclerotic lesions.
Approach and results: We examined the efficacy of carnosine, a naturally occurring β-alanyl-histidine dipeptide, in preventing aldehyde toxicity and atherogenesis in apolipoprotein E-null mice. In vitro, carnosine reacted rapidly with lipid peroxidation-derived unsaturated aldehydes. Gas chromatography mass-spectrometry analysis showed that carnosine inhibits the formation of free aldehydes 4-hydroxynonenal and malonaldialdehyde in Cu(2+)-oxidized low-density lipoprotein. Preloading bone marrow-derived macrophages with cell-permeable carnosine analogs reduced 4-hydroxynonenal-induced apoptosis. Oral supplementation with octyl-D-carnosine decreased atherosclerotic lesion formation in aortic valves of apolipoprotein E-null mice and attenuated the accumulation of protein-acrolein, protein-4-hydroxyhexenal, and protein-4-hydroxynonenal adducts in atherosclerotic lesions, whereas urinary excretion of aldehydes as carnosine conjugates was increased.
Conclusions: The results of this study suggest that carnosine inhibits atherogenesis by facilitating aldehyde removal from atherosclerotic lesions. Endogenous levels of carnosine may be important determinants of atherosclerotic lesion formation, and treatment with carnosine or related peptides could be a useful therapy for the prevention or the treatment of atherosclerosis.
Keywords: aldehydes; atherosclerosis; carnosine; oxidized low-density lipoprotein.
Figures
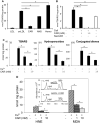
Figure 1. Histidyl dipeptides prevent LDL oxidation
Oxidation of LDL (measured by the formation of TBARS) in the presence of A, CuSO4 (10 μM) and 10 mM carnosine (CAR), anserine (ANS), or homocarnosine (HOMO) or B, murine bone marrow-derived macrophages (BMM), and carnosine or N-acetyl-carnoine (NAcCAR). For the experiments with N-acetyl-carnosine, BMM were pre-incubated with NacCAR (10 mM) for 24 h and rinsed with PBS prior to the addition of LDL. C, Formation of TBARS (i), hydroperoxides (ii), and conjugated dienes (iii) in LDL oxidized in the presence of Cu2+ and 1 or 10 mM carnosine. D. Inhibition of HNE and MDA formation in Cu2+-oxidized LDL in the presence of carnosine. Inset shows GC-CI/MS identification of MDA and HNE. Values are mean ± SE from 3 independent experiments. *P<0.05.
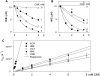
Figure 2. Carnosine reacts with oxidized lipids-derived aldehydes
The rate of disappearance of A, HNE and B, HHE (10 μM each) from a reaction mixture containing indicated concentrations of carnosine in 0.15 M potassium phosphate, pH 7.4. The reaction mixture was incubated at 37°C and aliquots were removed at indicated times to measure free aldehyde by GC-CI/MS. Data are shown as discrete points and the curves are the best fit of a single exponential equation to the data [_y=Ae−kobst_]. C, Concentration dependence of kobs. Best fits of the linear dependence were used to calculate the second order rate constants listed in Table 1. ACR indicates acrolein; OCT, octenal; ONE, 4-oxononenal.
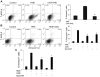
Figure 3. Carnosine protects macrophages from HNE-induced apoptosis
Flow cytometry analysis of bone marrow macrophages treated with A, HNE or carnosine-HNE or B, pre-incubated with the cell-permeable carnosine analogs, octyl-D-carnosine (ODC) or N-acetyl-carnosine (NacCAR) for 16h and then incubated for 2h with HBSS or HNE as indicated. Cells were labeled with FITC-Annexin V/7-AAD to monitor apoptosis. The lower right quadrant shows the early apoptotic cells and the upper right quadrant shows the late apoptotic/necrotic cells. Panel C shows the effect of ODC and NacCAR on HNE-induced apoptosis in the presence of 5% fetal bovine serum in RPMI medium. Values are mean ± SEM of 3 independent experiments, each conducted in triplicate or quadruplicate. *P<0.01 versus HNE-treated cells.
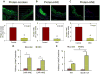
Figure 4. Carnosine-feeding inhibits early phase of atherogenesis and decreases the accumulation of macrophages in lesions
A, Carnosine level in the plasma (i), spleen (ii) and aorta (iii) of 14-week-old female apoE-null mice maintained on high fat diet and fed ODC (60 mg/kg in drinking water) for 6 weeks starting at 8 weeks of age. Concentration of carnosine was measured by LC/ESI+-MS as described in Methods. B, Lesions in the aortic valve. Lipids were visualized by Oil red O staining. Panels (i) shows the representative photomicrographs of aortic valve of control and ODC-fed mice. Panel (ii) shows the group data. C, Macrophage accumulation in the aortic valve was examined by staining with Alexa 647-conjugated CD-68 antibody. Panels (i) and (ii) shows the representative photomicrographs of aortic valve of control and ODC-fed mice and the group data, respectively. Values are means ± SEM. *P < 0.05 versus controls.
Figure 5. Carnosine removes aldehydes from atherosclerotic lesions of apoE-null mice
Photomicrographs of aortic valves stained with A, anti-KLH-acrolein B, anti-KLH-HHE, or C, anti-KLH-HNE antibodies. Mice were treated with ODC as described in Fig. 4. Nuclei were stained with DAPI. Values are means ± SEM. #P < 0.01 versus controls. D, Abundance of CAR-HNE and CAR-HHE conjugates in the urine of apoE-null mice, described in panels A-C. Urine was collected 2 days prior to sacrifice. E, Abundance of carnosine-HHE in the urine of 30 week-old female C57BL/6 and apoE-null mice before and after feeding ODC. Control urine was collected for 24 hours. Mice were then fed ODC (60 mg/kg in water by gavage) and urine was collected for an additional 24 hours. MRM traces of the conjugates are shown in Supplemental Figure IV. Values are means ± SEM. *P < 0.05.
Similar articles
- Aldose reductase protects against early atherosclerotic lesion formation in apolipoprotein E-null mice.
Srivastava S, Vladykovskaya E, Barski OA, Spite M, Kaiserova K, Petrash JM, Chung SS, Hunt G, Dawn B, Bhatnagar A. Srivastava S, et al. Circ Res. 2009 Oct 9;105(8):793-802. doi: 10.1161/CIRCRESAHA.109.200568. Epub 2009 Sep 3. Circ Res. 2009. PMID: 19729598 Free PMC article. - Aldehyde adducts generated during lipid peroxidation modification of proteins.
Uchida K. Uchida K. Free Radic Res. 2015;49(7):896-904. doi: 10.3109/10715762.2015.1036052. Epub 2015 May 26. Free Radic Res. 2015. PMID: 25968950 Review. - Cardiospecific Overexpression of ATPGD1 (Carnosine Synthase) Increases Histidine Dipeptide Levels and Prevents Myocardial Ischemia Reperfusion Injury.
Zhao J, Conklin DJ, Guo Y, Zhang X, Obal D, Guo L, Jagatheesan G, Katragadda K, He L, Yin X, Prodhan MAI, Shah J, Hoetker D, Kumar A, Kumar V, Wempe MF, Bhatnagar A, Baba SP. Zhao J, et al. J Am Heart Assoc. 2020 Jun 16;9(12):e015222. doi: 10.1161/JAHA.119.015222. Epub 2020 Jun 9. J Am Heart Assoc. 2020. PMID: 32515247 Free PMC article. - Histamine H1 receptor promotes atherosclerotic lesion formation by increasing vascular permeability for low-density lipoproteins.
Rozenberg I, Sluka SH, Rohrer L, Hofmann J, Becher B, Akhmedov A, Soliz J, Mocharla P, Borén J, Johansen P, Steffel J, Watanabe T, Lüscher TF, Tanner FC. Rozenberg I, et al. Arterioscler Thromb Vasc Biol. 2010 May;30(5):923-30. doi: 10.1161/ATVBAHA.109.201079. Epub 2010 Mar 4. Arterioscler Thromb Vasc Biol. 2010. PMID: 20203300 - Aldehyde-sequestering drugs: tools for studying protein damage by lipid peroxidation products.
Burcham PC, Kaminskas LM, Fontaine FR, Petersen DR, Pyke SM. Burcham PC, et al. Toxicology. 2002 Dec 27;181-182:229-36. doi: 10.1016/s0300-483x(02)00287-1. Toxicology. 2002. PMID: 12505316 Review.
Cited by
- Use of carnosine in the prevention of cardiometabolic risk factors in overweight and obese individuals: study protocol for a randomised, double-blind placebo-controlled trial.
Menon K, Cameron JD, de Courten M, de Courten B. Menon K, et al. BMJ Open. 2021 May 13;11(5):e043680. doi: 10.1136/bmjopen-2020-043680. BMJ Open. 2021. PMID: 33986049 Free PMC article. - The protective role of carnosine against type 2 diabetes-induced cognitive impairment.
Wang Q, Tripodi N, Valiukas Z, Bell SM, Majid A, de Courten B, Apostolopoulos V, Feehan J. Wang Q, et al. Food Sci Nutr. 2024 Mar 12;12(6):3819-3833. doi: 10.1002/fsn3.4077. eCollection 2024 Jun. Food Sci Nutr. 2024. PMID: 38873448 Free PMC article. Review. - Modulation of mitochondrial function and autophagy mediates carnosine neuroprotection against ischemic brain damage.
Baek SH, Noh AR, Kim KA, Akram M, Shin YJ, Kim ES, Yu SW, Majid A, Bae ON. Baek SH, et al. Stroke. 2014 Aug;45(8):2438-2443. doi: 10.1161/STROKEAHA.114.005183. Epub 2014 Jun 17. Stroke. 2014. PMID: 24938837 Free PMC article. - Biogenic Aldehydes as Therapeutic Targets for Cardiovascular Disease.
Nelson MM, Baba SP, Anderson EJ. Nelson MM, et al. Curr Opin Pharmacol. 2017 Apr;33:56-63. doi: 10.1016/j.coph.2017.04.004. Epub 2017 May 18. Curr Opin Pharmacol. 2017. PMID: 28528297 Free PMC article. Review. - Metabolic Pathways for Removing Reactive Aldehydes are Diminished in Atrophic Muscle During Heart Failure.
Chaudhari M, Zelko I, Lorkiewicz P, Hoetker D, Doelling B, Brittian K, Bhatnagar A, Srivastava S, Baba SP. Chaudhari M, et al. Res Sq [Preprint]. 2023 Nov 21:rs.3.rs-3621159. doi: 10.21203/rs.3.rs-3621159/v1. Res Sq. 2023. PMID: 38045249 Free PMC article. Updated. Preprint.
References
- Steinberg D, Witztum JL. Oxidized low-density lipoprotein and atherosclerosis. Arterioscler Thromb Vasc Biol. 2010;30:2311–2316. - PubMed
- Tsimikas S, Witztum JL. The role of oxidized phospholipids in mediating lipoprotein(a) atherogenicity. Curr Opin Lipidol. 2008;19:369–377. - PubMed
- Tsimikas S, Kiechl S, Willeit J, Mayr M, Miller ER, Kronenberg F, Xu Q, Bergmark C, Weger S, Oberhollenzer F, Witztum JL. Oxidized phospholipids predict the presence and progression of carotid and femoral atherosclerosis and symptomatic cardiovascular disease: Five-year prospective results from the bruneck study. J Am Coll Cardiol. 2006;47:2219–2228. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- ES17260/ES/NIEHS NIH HHS/United States
- P20 RR024489/RR/NCRR NIH HHS/United States
- HL-78825/HL/NHLBI NIH HHS/United States
- R01 HL095593/HL/NHLBI NIH HHS/United States
- HL95593/HL/NHLBI NIH HHS/United States
- R01 HL122581/HL/NHLBI NIH HHS/United States
- GM103492/GM/NIGMS NIH HHS/United States
- R01 HL059378/HL/NHLBI NIH HHS/United States
- P20 GM103492/GM/NIGMS NIH HHS/United States
- P01 HL078825/HL/NHLBI NIH HHS/United States
- R01 ES017260/ES/NIEHS NIH HHS/United States
- HL-59378/HL/NHLBI NIH HHS/United States
- R01 HL055477/HL/NHLBI NIH HHS/United States
- HL-55477/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical