Exercise and sodium butyrate transform a subthreshold learning event into long-term memory via a brain-derived neurotrophic factor-dependent mechanism - PubMed (original) (raw)
Exercise and sodium butyrate transform a subthreshold learning event into long-term memory via a brain-derived neurotrophic factor-dependent mechanism
Karlie A Intlekofer et al. Neuropsychopharmacology. 2013 Sep.
Abstract
We demonstrate that exercise enables hippocampal-dependent learning in conditions that are normally subthreshold for encoding and memory formation, and depends on hippocampal induction of brain-derived neurotrophic factor (BDNF) as a key mechanism. Using a weak training paradigm in an object location memory (OLM) task, we show that sedentary mice are unable to discriminate 24 h later between familiar and novel object locations. In contrast, 3 weeks of prior voluntary exercise enables strong discrimination in the spatial memory task. Cognitive benefits of exercise match those attained with post-training sodium butyrate (NaB), a histone deacetylase (HDAC) inhibitor previously shown to enable subthreshold learning. We demonstrate that the enabling effects of exercise and NaB on subthreshold OLM learning are dependent on hippocampal BDNF upregulation, and are blocked by hippocampal infusion of BDNF short-interfering RNA. Exercise and NaB increased bdnf transcripts I and IV, and the increases were associated with BDNF promoter acetylation on H4K8 but not H4K12. These data provide support for the concept that exercise engages epigenetic control mechanisms and serves as a natural stimulus that operates in part like NaB and potentially other HDAC inhibitors, placing the brain into a state of readiness for plasticity.
Figures
Figure 1
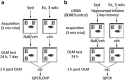
Experimental design. (a) Sedentary (Sed) or exercised (EX) mice were given a 3-min acquisition trial (as a subthreshold learning task as shown previously; Stefanko et al, 2009; McQuown_et al_, 2011), followed immediately with an i.p. injection of sodium butyrate (NaB) or vehicle (veh), and returning all animals to clean home cages without access to running wheels. Twenty-four hours or 7 days later, a 5-min object location memory (OLM) retention test was administered where one object was moved to a novel location. Time spent exploring the identical objects was recorded and a discrimination index (DI) was calculated to determine preference for the object in the novel location. (b) To determine whether a brain-derived neurotrophic factor (BDNF)-dependent mechanism was involved in the enhancement of subthreshold learning, short-interfering RNA (siRNA) against bdnf, or control siRNA, were infused to the hippocampus 2 days before OLM acquisition. Mice were given a 3-min acquisition trial, followed 24 h later by a 5-min OLM retention test. Animals were killed 1 h after the OLM test, and hippocampi were removed for molecular assays.
Figure 2
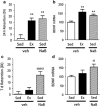
In a subthreshold object location memory (OLM) paradigm, exercise (Ex) improves learning equivalent to sedentary (Sed) mice injected with post-acquisition sodium butyrate (NaB), a histone deacetylase (HDAC) inhibitor. (a) Twenty-four hours after a 3-min acquisition trial, vehicle-treated Sed animals do not show preference for the novel location over the familiar location, as measured by the discrimination index ratio (DI). Following 3 weeks voluntary exercise, Ex animals show marked preference for the novel location to a similar extent as post-acquisition injection of NaB. (b) Quantitative reverse transcription PCR (RT-qPCR) revealed that hippocampal brain-derived neurotrophic factor (bdnf) mRNA levels are increased by exercise and NaB, in parallel with the cognitive enhancement. (c) Effects of NaB on cognition persist 7 days after the acquisition trial, whereas the exercise effects on spatial memory in the subthreshold paradigm eventually decay after daily physical activity ceases. (d) RT-qPCR revealed that bdnf mRNA expression patterns at the 7-day timepoint paralleled the cognitive data across the treatment groups, remaining elevated after NaB treatment but not with exercise. Data are expressed as means±SEM. For the 24-h timepoint: **p<0.01 vs sedentary. For the 7-day timepoint:σ_p_<0.05;σσσ_p_<0.001 vs sedentary.
Figure 3
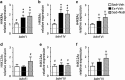
Exercise (Ex) and sodium butyrate (NaB) increase acetylation of histone H4, specifically acetylation at H4K8 of promoters for (a) brain-derived neurotrophic factor (bdnf) I and (b) bdnf IV, but not (c) bdnf VI. Acetylation at H4K12 was not significantly increased by Ex or NaB at either promoters (d)bdnf I, (e) bdnf IV, or (f) bdnf VI. Comparing sedentary (Sed) animals with or without object location memory (OLM) testing, behavioral testing had a small effect on H4K8 acetylation on bdnf promoters I and IV, and H4K12 acetylation in bdnf promoter IV. Data are expressed as means±SEM, *p<0.05 vs Sed+Veh (OLM tested),#p<0.05, vs baseline acetylation level in sedentary unhandled animals (indicated by dotted line).
Figure 4
Preventing increased brain-derived neurotrophic factor (bdnf) mRNA expression with intrahippocampal short-interfering RNA (siRNA) blocks the cognitive benefits of exercise (Ex) and sodium butyrate (NaB) in a subthreshold object location memory (OLM) learning task. (a) Exercise-dependent improvements in the subthreshold OLM paradigm were abolished by intrahippocampal infusion of bdnf siRNA, but not by control siRNA. Intrahippocampal bdnf siRNA blocked induction of (b) bdnf mRNA and (c) BDNF protein expression by exercise and decreased levels of mRNA and protein below baseline. (d) Cognitive improvements in the subthreshold OLM paradigm with NaB treatment were blocked by intrahippocampal infusion of bdnf siRNA, but not control siRNA. Data are expressed as means±SEM. *p<0.05; **p<0.01; ***p<0.001.
Similar articles
- Exercise and pharmacological inhibition of histone deacetylase improves cognitive function accompanied by an increase of gene expressions crucial for neuronal plasticity in the hippocampus.
Kitahara M, Inoue T, Mani H, Takamatsu Y, Ikegami R, Tohyama H, Maejima H. Kitahara M, et al. Neurosci Lett. 2021 Apr 1;749:135749. doi: 10.1016/j.neulet.2021.135749. Epub 2021 Feb 18. Neurosci Lett. 2021. PMID: 33610667 - Effects of exercise and pharmacological inhibition of histone deacetylases (HDACs) on epigenetic regulations and gene expressions crucial for neuronal plasticity in the motor cortex.
Maejima H, Kitahara M, Takamatsu Y, Mani H, Inoue T. Maejima H, et al. Brain Res. 2021 Jan 15;1751:147191. doi: 10.1016/j.brainres.2020.147191. Epub 2020 Nov 2. Brain Res. 2021. PMID: 33152341 - Temporal endurance of exercise-induced benefits on hippocampus-dependent memory and synaptic plasticity in female mice.
Dong TN, Kramár EA, Beardwood JH, Al-Shammari A, Wood MA, Keiser AA. Dong TN, et al. Neurobiol Learn Mem. 2022 Oct;194:107658. doi: 10.1016/j.nlm.2022.107658. Epub 2022 Jul 8. Neurobiol Learn Mem. 2022. PMID: 35811066 Free PMC article. - The weekend warrior effect: Consistent intermittent exercise induces persistent cognitive benefits.
La Tour S, Shaikh H, Beardwood JH, Augustynski AS, Wood MA, Keiser AA. La Tour S, et al. Neurobiol Learn Mem. 2024 Oct;214:107971. doi: 10.1016/j.nlm.2024.107971. Epub 2024 Aug 11. Neurobiol Learn Mem. 2024. PMID: 39137861 Free PMC article. - Use it and boost it with physical and mental activity.
Korol DL, Gold PE, Scavuzzo CJ. Korol DL, et al. Hippocampus. 2013 Nov;23(11):1125-35. doi: 10.1002/hipo.22197. Hippocampus. 2013. PMID: 23996382 Free PMC article. Review.
Cited by
- Effect of a multi-strain probiotic mixture consumption on anxiety and depression symptoms induced in adult mice by postnatal maternal separation.
De Santa F, Strimpakos G, Marchetti N, Gargari G, Torcinaro A, Arioli S, Mora D, Petrella C, Farioli-Vecchioli S. De Santa F, et al. Microbiome. 2024 Feb 19;12(1):29. doi: 10.1186/s40168-024-01752-w. Microbiome. 2024. PMID: 38369490 Free PMC article. - Exercise opens a temporal window for enhanced cognitive improvement from subsequent physical activity.
Butler CW, Keiser AA, Kwapis JL, Berchtold NC, Wall VL, Wood MA, Cotman CW. Butler CW, et al. Learn Mem. 2019 Nov 15;26(12):485-492. doi: 10.1101/lm.050278.119. Print 2019 Dec. Learn Mem. 2019. PMID: 31732709 Free PMC article. - Relationship Between Short-chain Fatty Acids and Parkinson's Disease: A Review from Pathology to Clinic.
Duan WX, Wang F, Liu JY, Liu CF. Duan WX, et al. Neurosci Bull. 2024 Apr;40(4):500-516. doi: 10.1007/s12264-023-01123-9. Epub 2023 Sep 27. Neurosci Bull. 2024. PMID: 37755674 Free PMC article. Review. - Histone deacetylase inhibition induces long-lasting changes in maternal behavior and gene expression in female mice.
Stolzenberg DS, Stevens JS, Rissman EF. Stolzenberg DS, et al. Endocrinology. 2014 Sep;155(9):3674-83. doi: 10.1210/en.2013-1946. Epub 2014 Jun 16. Endocrinology. 2014. PMID: 24932804 Free PMC article. - Train and Reprogram Your Brain: Effects of Physical Exercise at Different Stages of Life on Brain Functions Saved in Epigenetic Modifications.
Kukla-Bartoszek M, Głombik K. Kukla-Bartoszek M, et al. Int J Mol Sci. 2024 Nov 9;25(22):12043. doi: 10.3390/ijms252212043. Int J Mol Sci. 2024. PMID: 39596111 Free PMC article. Review.
References
- Alarcon JM, Malleret G, Touzani K, Vronskaya S, Ishii S, Kandel ER, et al. Chromatin acetylation, memory, and LTP are impaired in CBP+/− mice: a model for the cognitive deficit in Rubinstein-Taybi syndrome and its amelioration. Neuron. 2004;42:947–959. - PubMed
- Bekinschtein P, Oomen CA, Saksida LM, Bussey TJ. Effects of environmental enrichment and voluntary exercise on neurogenesis, learning and memory, and pattern separation: BDNF as a critical variable. Semin Cell Dev Biol. 2011;22:536–542. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- F31 DA029368/DA/NIDA NIH HHS/United States
- AG34667/AG/NIA NIH HHS/United States
- R01 MH081004/MH/NIMH NIH HHS/United States
- R01 AG034667/AG/NIA NIH HHS/United States
- P01 AG000538/AG/NIA NIH HHS/United States
- T32AG9629/AG/NIA NIH HHS/United States
- F31DA29368/DA/NIDA NIH HHS/United States
- MH081004/MH/NIMH NIH HHS/United States
- AG00538/AG/NIA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources