Middle East respiratory syndrome coronavirus (MERS-CoV) causes transient lower respiratory tract infection in rhesus macaques - PubMed (original) (raw)
. 2013 Oct 8;110(41):16598-603.
doi: 10.1073/pnas.1310744110. Epub 2013 Sep 23.
Angela L Rasmussen, Darryl Falzarano, Trenton Bushmaker, Friederike Feldmann, Douglas L Brining, Elizabeth R Fischer, Cynthia Martellaro, Atsushi Okumura, Jean Chang, Dana Scott, Arndt G Benecke, Michael G Katze, Heinz Feldmann, Vincent J Munster
Affiliations
- PMID: 24062443
- PMCID: PMC3799368
- DOI: 10.1073/pnas.1310744110
Middle East respiratory syndrome coronavirus (MERS-CoV) causes transient lower respiratory tract infection in rhesus macaques
Emmie de Wit et al. Proc Natl Acad Sci U S A. 2013.
Abstract
In 2012, a novel betacoronavirus, designated Middle East respiratory syndrome coronavirus or MERS-CoV and associated with severe respiratory disease in humans, emerged in the Arabian Peninsula. To date, 108 human cases have been reported, including cases of human-to-human transmission. The availability of an animal disease model is essential for understanding pathogenesis and developing effective countermeasures. Upon a combination of intratracheal, ocular, oral, and intranasal inoculation with 7 × 10(6) 50% tissue culture infectious dose of the MERS-CoV isolate HCoV-EMC/2012, rhesus macaques developed a transient lower respiratory tract infection. Clinical signs, virus shedding, virus replication in respiratory tissues, gene expression, and cytokine and chemokine profiles peaked early in infection and decreased over time. MERS-CoV caused a multifocal, mild to marked interstitial pneumonia, with virus replication occurring mainly in alveolar pneumocytes. This tropism of MERS-CoV for the lower respiratory tract may explain the severity of the disease observed in humans and the, up to now, limited human-to-human transmission.
Keywords: DPP4; emerging infectious disease.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
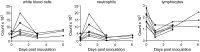
Fig. 1.
Hematological changes in rhesus macaques inoculated with MERS-CoV. Total number of white blood cells (Left), neutrophils (Center), and lymphocytes (Right) were determined in blood samples obtained from animals 0, 1, 3, and 6 dpi. Of note, CoV-2, CoV-4, and CoV-6 were euthanized 3 dpi, and thus no samples are available for 6 dpi. Each line represents one animal: CoV-1 (●), CoV-2 (■), CoV-3 (▲), CoV-4 (▼), CoV-5 (♦), CoV-6 (○).
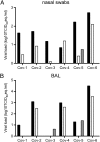
Fig. 2.
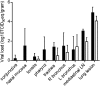
Virus shedding in rhesus macaques inoculated with MERS-CoV. Nasal swabs (A) and BAL fluid (B) were collected 1, 3, and 6 dpi. Of note, CoV-2, CoV-4, and CoV-6 were euthanized 3 dpi and thus no samples are available for 6 dpi. RNA was extracted and viral load was determined as TCID50 equivalents by qRT-PCR. TCID50 equivalents were extrapolated from standard curves generated by adding dilutions of RNA extracted from a HCoV-EMC/2012 stock with known virus titer in parallel to each run. Animal numbers are indicated on the x axis. Black bars indicate viral load at 1 dpi, white bars indicate viral load at 3 dpi, and gray bars indicate viral load at 6 dpi.
Fig. 3.
Viral load in respiratory tissues of rhesus macaques inoculated with MERS-CoV. Rhesus macaques were euthanized on day 3 (black bars) and day 6 (white bars) postinfection and tissue samples were collected. RNA was extracted and viral load was determined as TCID50 equivalents by qRT-PCR. TCID50 equivalents were extrapolated from standard curves generated by adding dilutions of RNA extracted from a HCoV-EMC/2012 stock with known virus titer in parallel to each run. Geometric mean viral loads were calculated; error bars represent SD. R, right; L, left; LN, lymph node.
Fig. 4.
Virus replication in the lungs of MERS-CoV inoculated rhesus macaques. Transmission electron microscopy was performed on lung lesion samples collected from MERS-CoV–inoculated macaques at 6 dpi. Virus particles (arrows) are visible in type I pneumocytes (Left and Right). (Scale bar, Right, 250 nm; Inset, 50 nm). B, basement membrane; E, endothelial cell; P, pneumocytes; R, red blood cell.
Fig. 5.
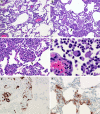
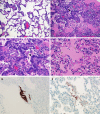
Histopathological changes at 3 dpi in rhesus macaques inoculated with MERS-CoV. Rhesus macaques were euthanized on day 3 postinfection and lung tissue was collected and stained with H&E. (A) Mild acute interstitial pneumonia demonstrated by mild thickening of alveolar septae by small numbers of neutrophils, macrophages, fibrin, and edema. Small numbers of inflammatory cells and fibrin also extend into alveolar spaces. (B) Marked interstitial pneumonia with bronchiolitis obliterans (arrow). Airways are filled with organizing fibrin, necrotic debris, collagen and fibroblasts. There are numerous multinucleate syncytial cells scattered throughout the alveoli (arrowhead). (C) Bronchiolitis obliterans organizing pneumonia. (D) Alveolar capillary (asterisk) bounded by degenerate, vacuolated type I pulmonary epithelium. The alveolar space contains inflammatory cells and a multinucleate syncytial cell (arrowhead). (E) ISH demonstrates viral RNA predominantly within alveolar pneumocytes. (F) Anticoronaviral IHC reveals viral antigen within alveolar pneumocytes. (Magnification: A and B, 200×; C, E, and F, 400×; D, 1,000×.)
Fig. 6.
Histopathological changes at 6 dpi in rhesus macaques inoculated with MERS-CoV. Rhesus macaques were euthanized at 6 dpi and lung tissue was collected and stained with H&E. (A) Mild acute interstitial pneumonia demonstrated by mild thickening of alveolar septae by small numbers of neutrophils, macrophages, fibrin, and edema. Small numbers of inflammatory cells and fibrin also extend into alveolar spaces. (B) Marked interstitial pneumonia with extensive type II pneumocyte hyperplasia (arrow). Alveolar spaces are flooded with edema fluid, fibrin, and inflammatory cells. (C) Type II pneumocyte hyperplasia (arrow) with alveolar flooding edema and fibrin. (D) Hyaline membranes line alveolar space (arrowhead). (E) ISH demonstrates viral RNA predominantly within alveolar pneumocytes. (F) Anticoronaviral IHC reveals viral antigen within alveolar pneumocytes. (Magnification: A and B, 200×; C_–_F, 400×.)
Fig. 7.
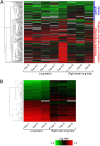
Microarray analysis of gene expression in lungs of rhesus macaques inoculated with MERS-CoV. The heatmap shows log10 ratios to uninfected controls of differentially expressed genes in the right lung lower lobe and lung lesion samples as determined by Welch’s t test (P < 0.01, fold-change ≥ 2), and grouped by hierarchical clustering. A representation is shown of 173 differentially expressed genes from individual animals and sample sites at 3 dpi (A) and of 37 differentially expressed genes from individual animals and sample sites at 6 dpi (B). Functional analysis was performed on subgroups of the day 3 differentially expressed genes, and enriched functional categories identified by IPA are shown in the blue and red vertical bars to the right of the heatmap in A. Differentially expressed genes are specified in
Table S2
(3 dpi) and
Table S3
(6 dpi).
Similar articles
- Animal models of Middle East respiratory syndrome coronavirus infection.
van Doremalen N, Munster VJ. van Doremalen N, et al. Antiviral Res. 2015 Oct;122:28-38. doi: 10.1016/j.antiviral.2015.07.005. Epub 2015 Jul 17. Antiviral Res. 2015. PMID: 26192750 Free PMC article. Review. - An animal model of MERS produced by infection of rhesus macaques with MERS coronavirus.
Yao Y, Bao L, Deng W, Xu L, Li F, Lv Q, Yu P, Chen T, Xu Y, Zhu H, Yuan J, Gu S, Wei Q, Chen H, Yuen KY, Qin C. Yao Y, et al. J Infect Dis. 2014 Jan 15;209(2):236-42. doi: 10.1093/infdis/jit590. Epub 2013 Nov 11. J Infect Dis. 2014. PMID: 24218506 Free PMC article. - Reverse genetics with a full-length infectious cDNA of the Middle East respiratory syndrome coronavirus.
Scobey T, Yount BL, Sims AC, Donaldson EF, Agnihothram SS, Menachery VD, Graham RL, Swanstrom J, Bove PF, Kim JD, Grego S, Randell SH, Baric RS. Scobey T, et al. Proc Natl Acad Sci U S A. 2013 Oct 1;110(40):16157-62. doi: 10.1073/pnas.1311542110. Epub 2013 Sep 16. Proc Natl Acad Sci U S A. 2013. PMID: 24043791 Free PMC article. - Comparative pathology of rhesus macaque and common marmoset animal models with Middle East respiratory syndrome coronavirus.
Yu P, Xu Y, Deng W, Bao L, Huang L, Xu Y, Yao Y, Qin C. Yu P, et al. PLoS One. 2017 Feb 24;12(2):e0172093. doi: 10.1371/journal.pone.0172093. eCollection 2017. PLoS One. 2017. PMID: 28234937 Free PMC article. - A Comparative Review of Animal Models of Middle East Respiratory Syndrome Coronavirus Infection.
Baseler L, de Wit E, Feldmann H. Baseler L, et al. Vet Pathol. 2016 May;53(3):521-31. doi: 10.1177/0300985815620845. Epub 2016 Feb 11. Vet Pathol. 2016. PMID: 26869154 Review.
Cited by
- Animal models of Middle East respiratory syndrome coronavirus infection.
van Doremalen N, Munster VJ. van Doremalen N, et al. Antiviral Res. 2015 Oct;122:28-38. doi: 10.1016/j.antiviral.2015.07.005. Epub 2015 Jul 17. Antiviral Res. 2015. PMID: 26192750 Free PMC article. Review. - Some pathological observations on the naturally infected dromedary camels (Camelus dromedarius) with the Middle East respiratory syndrome coronavirus (MERS-CoV) in Saudi Arabia 2018-2019.
Alnaeem A, Kasem S, Qasim I, Al-Doweriej A, Al-Houfufi A, Alwazan A, Albadrani A, Alshaammari K, Refaat M, Al-Shabebi A, Hemida MG. Alnaeem A, et al. Vet Q. 2020 Dec;40(1):190-197. doi: 10.1080/01652176.2020.1781350. Vet Q. 2020. PMID: 32543343 Free PMC article. - Passive immunotherapy with dromedary immune serum in an experimental animal model for Middle East respiratory syndrome coronavirus infection.
Zhao J, Perera RA, Kayali G, Meyerholz D, Perlman S, Peiris M. Zhao J, et al. J Virol. 2015 Jun;89(11):6117-20. doi: 10.1128/JVI.00446-15. Epub 2015 Mar 18. J Virol. 2015. PMID: 25787284 Free PMC article. - Development of Medical Countermeasures to Middle East Respiratory Syndrome Coronavirus.
Uyeki TM, Erlandson KJ, Korch G, O'Hara M, Wathen M, Hu-Primmer J, Hojvat S, Stemmy EJ, Donabedian A. Uyeki TM, et al. Emerg Infect Dis. 2016 Jul;22(7):e160022. doi: 10.3201/eid2207.160022. Epub 2016 Jul 15. Emerg Infect Dis. 2016. PMID: 27191188 Free PMC article. - Use of a rapid electronic survey methodology to estimate blood donors' potential exposure to emerging infectious diseases: Application of a statistically representative sampling methodology to assess risk in US blood centers.
Whitaker BI, Walderhaug M, Hinkins S, Steele WR, Custer B, Kessler D, Leparc G, Gottschall JL, Bialkowski W, Stramer SL, Dodd RY, Crowder L, Vahidnia F, Shaz BH, Kamel H, Rebosa M, Stern M, Anderson SA. Whitaker BI, et al. Transfusion. 2020 Sep;60(9):1987-1997. doi: 10.1111/trf.15941. Epub 2020 Aug 3. Transfusion. 2020. PMID: 32743798 Free PMC article.
References
- Zaki AM, van Boheemen S, Bestebroer TM, Osterhaus AD, Fouchier RA. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med. 2012;367(19):1814–1820. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous