Type I interferon upregulates Bak and contributes to T cell loss during human immunodeficiency virus (HIV) infection - PubMed (original) (raw)
Clinical Trial
doi: 10.1371/journal.ppat.1003658. Epub 2013 Oct 10.
Yvonne M Mueller, Guibin Yang, Alina C Boesteanu, Donald T Gracias, Duc H Do, Jennifer L Hope, Noshin Kathuria, Shannon E McGettigan, Mark G Lewis, Luis D Giavedoni, Jeffrey M Jacobson, Peter D Katsikis
Affiliations
- PMID: 24130482
- PMCID: PMC3795023
- DOI: 10.1371/journal.ppat.1003658
Clinical Trial
Type I interferon upregulates Bak and contributes to T cell loss during human immunodeficiency virus (HIV) infection
Joseph A Fraietta et al. PLoS Pathog. 2013.
Abstract
The role of Type I interferon (IFN) during pathogenic HIV and SIV infections remains unclear, with conflicting observations suggesting protective versus immunopathological effects. We therefore examined the effect of IFNα/β on T cell death and viremia in HIV infection. Ex vivo analysis of eight pro- and anti-apoptotic molecules in chronic HIV-1 infection revealed that pro-apoptotic Bak was increased in CD4+ T cells and correlated directly with sensitivity to CD95/Fas-mediated apoptosis and inversely with CD4+ T cell counts. Apoptosis sensitivity and Bak expression were primarily increased in effector memory T cells. Knockdown of Bak by RNA interference inhibited CD95/Fas-induced death of T cells from HIV-1-infected individuals. In HIV-1-infected patients, IFNα-stimulated gene expression correlated positively with ex vivo T cell Bak levels, CD95/Fas-mediated apoptosis and viremia and negatively with CD4+ T cell counts. In vitro IFNα/β stimulation enhanced Bak expression, CD95/Fas expression and CD95/Fas-mediated apoptosis in healthy donor T cells and induced death of HIV-specific CD8+ T cells from HIV-1-infected patients. HIV-1 in vitro sensitized T cells to CD95/Fas-induced apoptosis and this was Toll-like receptor (TLR)7/9- and Type I IFN-dependent. This sensitization by HIV-1 was due to an indirect effect on T cells, as it occurred in peripheral blood mononuclear cell cultures but not purified CD4+ T cells. Finally, peak IFNα levels and viral loads correlated negatively during acute SIV infection suggesting a potential antiviral effect, but positively during chronic SIV infection indicating that either the virus drives IFNα production or IFNα may facilitate loss of viral control. The above findings indicate stage-specific opposing effects of Type I IFNs during HIV-1 infection and suggest a novel mechanism by which these cytokines contribute to T cell depletion, dysregulation of cellular immunity and disease progression.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
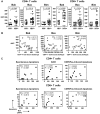
Figure 1. CD95/Fas-mediated, but not TRAIL- or TNFα-induced T cell apoptosis is increased in HIV-1-infected individuals and correlates inversely with CD4+ T cell counts.
(A) Representative flow cytometry plots depicting apoptosis of CD4+ T cells and HIV-specific CD8+ T cells induced by anti-CD95/Fas antibody, SuperKiller TRAIL or TNFα treated PBMC from HIV-1+ individuals for 14 hours. (B) Pooled data showing percent apoptosis of CD4+ T cells (n = 8), total CD8+ T cells (n = 8), HIV-specific (n = 5) and CMV/EBV-specific (n = 5) CD8+ T cells after anti-CD95/Fas-, TRAIL-, and TNFα-treatment. Bars depict means ± standard errors. The P values were calculated using Student t test for paired samples. (C) Spearman's rho correlation shown between the frequency of CD95/Fas-mediated apoptosis of CD4+ T cells and absolute CD4+ T cell counts (n = 31).
Figure 2. Bak expression is increased and correlates with CD4+ T cell counts and CD95-induced apoptosis in HIV-1-infected individuals.
(A) Mean fluorescence intensity (MFI) of Bak, Bax and Bim expression ex vivo shown in CD4+ T cells and CD8+ T cells from HIV-1+ patients and healthy controls. Each dot depicts a donor, the lines indicate 10% and 90% and the boxes depict 25%, median and 75% quantiles. The P values were calculated by using the nonparametric Wilcoxon signed rank test for unpaired samples. (B) Spearman's rho correlation shown between ex vivo Bak (n = 25), Bax (n = 24) and Bim (n = 19) expression in CD4+ T cells and CD4+ T counts in HIV-1-infected individuals. (C) Spearman's rho or Pearson's r correlations shown between ex vivo Bak expression and the percentage of spontaneous apoptosis, AICD and CD95/Fas-mediate apoptosis in CD4+ and CD8+ T cells from HIV-1-infected individuals (n = 10).
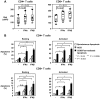
Figure 3. Bak silencing markedly reduces the sensitivity of T cells in chronic HIV-1 infection to CD95/Fas-induced apoptosis.
(A) Efficiency of siRNA uptake in T cells transfected with a pool of siRNAs and siGlo fluorescent oligonucleotides (to monitor transfection) compared to an electroporated negative control (no RNA or siGlo). Silencing of (B) Bak and (C) Bax (off-target control) by siRNA sequences targeting Bak in primary PBMC from a representative HIV+ patient measured by quantitative PCR. Expression (mean, SEM) normalized to 18S rRNA is presented as fold change relative to non-targeting control siRNA. (D) CD95/Fas-specific apoptosis of CD4+ T cells and CD8+ T cells in chronic HIV-1 infection following transfection with a pool of siRNAs directed against Bak, relative to a pool of non-targeting siRNAs (negative control). (E) Percent inhibition of CD95/Fas-specific cell death in T cells from chronically HIV-1-infected individuals following transfection with a Bak siRNA pool. Results were obtained from 3 independent experiments with 3 different HIV-1-infected patients.
Figure 4. Type I IFN increases Bak expression in T cells from healthy donors and sensitizes them to CD95/Fas-mediated apoptosis.
(A) Bak expression shown in CD4+ T cells and CD8+ T cells from healthy individuals after PBMC were untreated or treated with Type I IFN (1000 U/ml) for 72 hours. Each symbol depicts one donor (n = 6). Lines indicate 10% and 90% and the box depicts 25%, median and 75% quantiles. (B) Apoptosis sensitivity of T cells shown following a 72 hour Type I IFN treatment. Cells were stimulated with media alone (spontaneous apoptosis), plate-bound anti-CD3 antibody (AICD) or plate bound anti-CD95 antibody for 14 hours after pre-stimulation with Type I IFN as described in A and apoptosis was measured (n = 5). Bars depict mean ± standard error. P values shown in Panel A and B graphs were calculated by using the Student's t-test for paired samples.
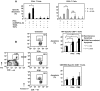
Figure 5. Type I IFN increases the sensitivity of T cells from HIV-1-infected individuals to CD95/Fas-induced death, but not TRAIL- and TNFα-mediated apoptosis.
(A) Treatment-specific apoptosis shown for CD4+ T cells (n = 10) and CD8+ (n = 10) T cells pre-treated with IFNα (1000 U/ml) for 72 hours prior to a 14 hour stimulation with plate-bound anti-CD95/Fas antibody, TRAIL (10 ng/ml) or TNFα (10 ng/ml). n.s. = not significant. (B) Representative flow cytometry (left panel) and pooled data (right panel) showing apoptosis sensitivity of HIV-specific CD8+ T cells (n = 7) and CMV/EBV-specific CD8+ T cells (n = 5) after pre-incubation of PBMC with IFNα or IFNβ (1000 U/ml) for 72 hours and subsequent stimulation with plate-bound anti-CD95/Fas antibody for 14 hours. Bars in graphs indicate mean ± standard error. P values shown in graphs were calculated by using the parametric Student's t-test or nonparametric Wilcoxon signed rank test for paired samples.
Figure 6. CD95/Fas-induced apoptosis of T cells exposed to HIV-1 is Type I IFN-dependent.
(A) TLR7/9 inhibitors inhibit HIV-1Ba-L-induced production of IFNα in infected PBMC from healthy donors. PBMC were exposed to 105 TCID50/ml HIV-1Ba-L in the presence or absence of a TLR7/9 antagonist for 24 hours (n = 13) and IFNα in supernatants was measured by ELISA. Bars indicate mean ± standard error. P values were calculated by using the Wilcoxon signed rank test for paired samples. (B) HIV-1Ba-L exposure increases CD95/Fas apoptosis of T cells, which is inhibited by a TLR7/9 antagonist and anti-IFNα/β receptor blocking antibodies. Sensitivity to CD95/Fas-induced apoptosis shown for CD4+ T cells and CD8+ T cells from healthy donors following a 72 hour exposure to HIV-1Ba-L in the presence or absence of anti-IFNα/β receptor blocking antibodies or a TLR7/9 antagonist (n = 5). Bars depict mean ± standard error. P values were calculated by using the Student's t-test for paired samples. (C) Representative flow cytometry plots from one out of three healthy donors depicting the sensitivity of CD4+ T cells to CD95/Fas-mediated apoptosis following exposure to non-infectious (AT-2 treated) HIV-1Ba-L for 72 hours in the presence or absence of an isotype control antibody, anti-IFNα/β receptor blocking antibody, and a TLR7/9-specific antagonist. Numbers indicate the percentage of apoptosis.
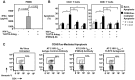
Figure 7. IFNα-induced gene expression is increased in HIV-1 infection and correlates with Bak expression, CD95/Fas-induced T cell apoptosis and CD4+ T cell loss.
(A) Ex vivo IFI6-16 and (B) ISG56 levels as measured by real-time RT PCR are significantly increased in HIV-1-infected patients (n = 7–14) compared to healthy donors (n = 6–8). P values were calculated by using the Wilcoxon signed rank test for unpaired samples. Spearman's rho correlations shown between IFI6-16 expression in PBMC and (C) ex vivo T cell Bak expression (n = 11), (D) ex vivo T cell Bax expression (n = 11) (E) ex vivo T cell Bim expression (n = 7) (F) ex vivo CD95/Fas-mediated T cell apoptosis sensitivity (n = 12) and (G) CD4+ T cell counts (n = 10) in ART-naive HIV-1-infected individuals. Correlations are shown for ART-naïve HIV-1-infected patients (n = 7) between plasma viral load and (H) IFI6-16 mRNA levels, (I) ISG56 mRNA levels. Spearman's rho correlations depicted between CD95/Fas-specific apoptosis of CD4+ T cells and (J) ISG56 expression, (K) IFI6-16 expression and (L) plasma viral load in ART-naïve HIV-1 patients.
Figure 8. Contrasting relationships between viral loads and Type I IFN levels during acute and chronic SIV infection.
(A) Plasma IFNα during pathogenic SIV infection as measured by a modified Luminex assay. (B) Plasma SIV viral load was determined by PCR (dashed line indicates assay detection limit) (C) CD4+ T cell counts during the acute and chronic stages of SIV infection. Spearman's rho correlations shown between plasma IFNα levels and viral loads during (D) acute and (E) chronic SIV infection of rhesus macaques. (F) Spearman's rho correlations shown between IFNα expression and CD4+ T cell counts in chronic SIV infection. Measurements are shown for SIV-infected rhesus macaques (n = 10).
Similar articles
- Activation of the CD95 (APO-1/Fas) system in T cells from human immunodeficiency virus type-1-infected children.
Bäumler CB, Böhler T, Herr I, Benner A, Krammer PH, Debatin KM. Bäumler CB, et al. Blood. 1996 Sep 1;88(5):1741-6. Blood. 1996. PMID: 8781430 - Reduced Chronic Lymphocyte Activation following Interferon Alpha Blockade during the Acute Phase of Simian Immunodeficiency Virus Infection in Rhesus Macaques.
Carnathan D, Lawson B, Yu J, Patel K, Billingsley JM, Tharp GK, Delmas OM, Dawoud R, Wilkinson P, Nicolette C, Cameron MJ, Sekaly RP, Bosinger SE, Silvestri G, Vanderford TH. Carnathan D, et al. J Virol. 2018 Apr 13;92(9):e01760-17. doi: 10.1128/JVI.01760-17. Print 2018 May 1. J Virol. 2018. PMID: 29467313 Free PMC article. - Induction of CD95 ligand expression on T lymphocytes and B lymphocytes and its contribution to apoptosis of CD95-up-regulated CD4+ T lymphocytes in macaques by infection with a pathogenic simian/human immunodeficiency virus.
Sasaki Y, Ami Y, Nakasone T, Shinohara K, Takahashi E, Ando S, Someya K, Suzaki Y, Honda M. Sasaki Y, et al. Clin Exp Immunol. 2000 Dec;122(3):381-9. doi: 10.1046/j.1365-2249.2000.01327.x. Clin Exp Immunol. 2000. PMID: 11122244 Free PMC article. - Are blockers of gp120/CD4 interaction effective inhibitors of HIV-1 immunopathogenesis?
Herbeuval JP, Shearer GM. Herbeuval JP, et al. AIDS Rev. 2006 Jan-Mar;8(1):3-8. AIDS Rev. 2006. PMID: 16736946 Review. - HIV-1 immunopathogenesis: how good interferon turns bad.
Herbeuval JP, Shearer GM. Herbeuval JP, et al. Clin Immunol. 2007 May;123(2):121-8. doi: 10.1016/j.clim.2006.09.016. Epub 2006 Nov 16. Clin Immunol. 2007. PMID: 17112786 Free PMC article. Review.
Cited by
- Natural amines inhibit activation of human plasmacytoid dendritic cells through CXCR4 engagement.
Smith N, Pietrancosta N, Davidson S, Dutrieux J, Chauveau L, Cutolo P, Dy M, Scott-Algara D, Manoury B, Zirafi O, McCort-Tranchepain I, Durroux T, Bachelerie F, Schwartz O, Münch J, Wack A, Nisole S, Herbeuval JP. Smith N, et al. Nat Commun. 2017 Feb 9;8:14253. doi: 10.1038/ncomms14253. Nat Commun. 2017. PMID: 28181493 Free PMC article. - HIV-1 and interferons: who's interfering with whom?
Doyle T, Goujon C, Malim MH. Doyle T, et al. Nat Rev Microbiol. 2015 Jul;13(7):403-13. doi: 10.1038/nrmicro3449. Epub 2015 Apr 27. Nat Rev Microbiol. 2015. PMID: 25915633 Free PMC article. Review. - Regulation of antiviral T cell responses by type I interferons.
Crouse J, Kalinke U, Oxenius A. Crouse J, et al. Nat Rev Immunol. 2015 Apr;15(4):231-42. doi: 10.1038/nri3806. Epub 2015 Mar 20. Nat Rev Immunol. 2015. PMID: 25790790 Review. - Type I interferon and HIV: Subtle balance between antiviral activity, immunopathogenesis and the microbiome.
Scagnolari C, Antonelli G. Scagnolari C, et al. Cytokine Growth Factor Rev. 2018 Apr;40:19-31. doi: 10.1016/j.cytogfr.2018.03.003. Epub 2018 Mar 16. Cytokine Growth Factor Rev. 2018. PMID: 29576284 Free PMC article. Review. - Investigational drugs with dual activity against HBV and HIV (Review).
Sun S, Yang Q, Sheng Y, Fu Y, Sun C, Deng C. Sun S, et al. Exp Ther Med. 2021 Jan;21(1):35. doi: 10.3892/etm.2020.9467. Epub 2020 Nov 11. Exp Ther Med. 2021. PMID: 33262821 Free PMC article. Review.
References
- Finkel TH, Tudor-Williams G, Banda NK, Cotton MF, Curiel T, et al. (1995) Apoptosis occurs predominantly in bystander cells and not in productively infected cells of HIV- and SIV-infected lymph nodes. Nat Med 1: 129–134. - PubMed
- Gougeon ML, Garcia S, Heeney J, Tschopp R, Lecoeur H, et al. (1993) Programmed cell death in AIDS-related HIV and SIV infections. AIDS Res Hum Retroviruses 9: 553–563. - PubMed
- Meyaard L, Otto SA, Jonker RR, Mijnster MJ, Keet RP, et al. (1992) Programmed death of T cells in HIV-1 infection. Science 257: 217–219. - PubMed
- Gougeon ML, Lecoeur H, Dulioust A, Enouf MG, Crouvoiser M, et al. (1996) Programmed cell death in peripheral lymphocytes from HIV-infected persons: increased susceptibility to apoptosis of CD4 and CD8 T cells correlates with lymphocyte activation and with disease progression. J Immunol 156: 3509–3520. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R24 OD013096/OD/NIH HHS/United States
- P51 OD011133/OD/NIH HHS/United States
- R01 AI046719/AI/NIAID NIH HHS/United States
- R33 AI082680/AI/NIAID NIH HHS/United States
- R21 AI082680/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous