Group 2 innate lymphoid cells are critical for the initiation of adaptive T helper 2 cell-mediated allergic lung inflammation - PubMed (original) (raw)
Group 2 innate lymphoid cells are critical for the initiation of adaptive T helper 2 cell-mediated allergic lung inflammation
Timotheus Y F Halim et al. Immunity. 2014.
Abstract
Naive CD4(+) T cell differentiation into distinct subsets of T helper (Th) cells is a pivotal process in the initiation of the adaptive immune response. Allergens predominantly stimulate Th2 cells, causing allergic inflammation. However, why allergens induce Th2 cell differentiation is not well understood. Here we show that group 2 innate lymphoid cells (ILC2s) are required to mount a robust Th2 cell response to the protease-allergen papain. Intranasal administration of papain stimulated ILC2s and Th2 cells, causing allergic lung inflammation and elevated immunoglobulin E titers. This process was severely impaired in ILC2-deficient mice. Whereas interleukin-4 (IL-4) was dispensable for papain-induced Th2 cell differentiation, ILC2-derived IL-13 was critical as it promoted migration of activated lung dendritic cells into the draining lymph node where they primed naive T cells to differentiate into Th2 cells. Papain-induced ILC2 activation and Th2 cell differentiation was IL-33-dependent, suggesting a common pathway in the initiation of Th2 cell responses to allergen.
Copyright © 2014 Elsevier Inc. All rights reserved.
Figures
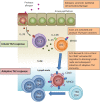
Graphical abstract
Figure 1
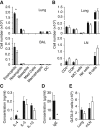
ILC2s and CD4+ T Cells Collaborated in Protease Allergen-Driven Innate and Adaptive Type-2 Lung Inflammation (A) Mice were sensitized by intranasal injection of 10 μg heat-inactivated (control) or active papain on days 0 and 1, followed by antigen challenge on days 13 and 20. (B) Serum concentrations of IgE were quantified on the days indicated (● papain, ○ control). (C–E) Mice injected with papain (▪) or control (□) were sacrificed on days 2, 14, and 21, followed by the quantification of myeloid populations in the bronchoalveolar lavage (BAL) and lung tissue by flow cytometry (C). Similarly, lung lymphoid populations were quantified. Cytokine concentrations in BAL were quantified by ELISA (D). Lymphocytes from indicated tissues were restimulated in vitro with recall antigen, followed by measurement for cytokine production (E). (F and G) Intracellular GATA3 was detected in CD4+ T cells from lung and mLN on day 21 following stimulation with papain (▪) or control (□). Shown are percent (F) and absolute numbers (G) of GATA3+ CD4+ T cells. (H) Lung cells were stained for intracellular IL-5 and IL-13 and analyzed by flow cytometry. The percentage of total IL-5+IL-13+ lung cells was measured in papain-treated or control mice on days 2 and 21. Double-positive cells were gated (left) and analyzed for surface expression of CD3 and NK1.1 to distinguish NK1.1−CD3+ T cells from NK1.1−CD3− ILC2s (for detailed analysis, see Figure S1). Cells were first gated for live (eFluor780−) CD45+ cells. The absolute numbers of lung IL-5+IL-13+ ILC2s (□) or CD4+ T cells (▪) in papain-treated mice, and IL-5+IL-13+ CD4+ T cells in control mice ( ) were calculated on days 2 and 21. Data are representative of at least three independent experiments. Mean ± SEM in (B)–(H), mean percent gated in H, ∗ = p ≤ 0.05 ∗∗ = p ≤ 0.01 (two-tailed Student’s t test). See also Figure S1.
) were calculated on days 2 and 21. Data are representative of at least three independent experiments. Mean ± SEM in (B)–(H), mean percent gated in H, ∗ = p ≤ 0.05 ∗∗ = p ≤ 0.01 (two-tailed Student’s t test). See also Figure S1.
Figure 2
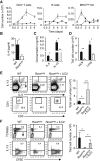
ILC2-Deficient Mice Were Incapable of Mounting an Effective Th2 Cell Response and Had Greatly Reduced Type 2 Lung Inflammation (A–G) WT (▪) and _Rora_sg/sg (□) BMT mice were treated with papain and analyzed on day 21 for myeloid cell numbers in the mLN, lung, and BAL (A). Lymphoid cell numbers were measured in the mLN and lung (B). The BAL was analyzed for concentrations of cytokines (C) and chemokines (D). Serum IgE concentrations were measured (E). The absolute number of GATA3+ CD4+ T cells was determined in the lung and mLN (F). Cells were restimulated with recall antigen, followed by an analysis for cytokine production in culture supernatant (G). (H) Formalin-fixed tissue was analyzed by histology for mucus production in PAS stained sections. Data are representative of at least three independent experiments. Scale bar represents 100 μm. Mean ± SEM in (A)–(G), ∗ = p ≤ 0.05 ∗∗ = p ≤ 0.001 (two-tailed Student’s t test). See also Figure S2.
Figure 3
IL-4 Played a Nonessential Role in the Allergic Response to Protease Allergen and Induction of Type 2 Lung Inflammation (A–E) WT (▪) and Il4 −/− (□) mice were treated with papain and analyzed on day 21 for myeloid cell numbers in the lung and BAL (A), lymphoid cell numbers in the lung and mLN (B), cytokine concentrations in the BAL (C), and IgE concentration in serum (D). Intracellular GATA3 was detected in CD4+ T cells from lung and mLN on day 21 following stimulation with papain, and absolute numbers of GATA3+ CD4+ T cells were calculated (E). Data are representative of three independent experiments. Mean ± SEM in (A)–(E), ∗ = p ≤ 0.05 (two-tailed Student’s t test). See also Figure S3.
Figure 4
ILC2 Were Instrumental for Induction of Th2 Cells in the mLN (A) WT mice received intranasal papain administration on days 0 and 1, and the absolute numbers of CD4+ T cells, B cells, and MHChi DCs were measured in the lung (●) or mLN (○) at various time-points by flow cytometry. (B and C) On day 6, cells from papain-treated WT (▪) or _Rora_sg/sg (□) BMT mouse mLN were restimulated in αCD3ε and αCD28 mAb coated wells, followed by analysis for cytokine production in culture supernatant. (D) Total numbers of IL-5+IL-13+ CD3+CD4+ Th2 cells from papain treated WT (▪) or _Rora_sg/sg (□) BMT mouse lung and mLN were measured on day 6. (E) WT BMT, _Rora_sg/sg BMT, or ILC2-transplanted _Rora_sg/sg BMT mice were stimulated with papain on days 0 and 1, and mLN lymphocytes were analyzed for intracellular IL-5 and IL-13 on day 6 by flow cytometry (top plots). Gated IL-5+IL-13+ lymphocytes were analyzed for CD3+CD4+ cells (bottom plots), and the total numbers of Th2 cells in the mLN were calculated (bar graph). (F) CFSE-labeled CD4+ T cells from OT-II mice were transplanted into WT BMT and _Rora_sg/sg BMT mice (±ILC2 transplantation), followed by intranasal injections of rmIL-33 + OVA on days 0 and 1. TCRVβ5+CFSE+ OT-II T cells in the mLN (top plots) were analyzed for intracellular IL-13 (bottom plots) by flow cytometry on day 6, and the total numbers of IL-13+ OT-II T cells in the mLN were calculated (bar graph). Data are representative of at least three independent experiments. Mean ± SEM in (A)–(F), mean percent gated in (E) and (F), ∗ = p ≤ 0.05 ∗∗ = p ≤ 0.01 (two-tailed Student’s t test). See also Figure S4.
Figure 5
IL-13 Was Required for Induction of Th2 Cells (A) WT (▪) and Il13 −/− (□) mice were treated with papain on days 0 and 1, and the total numbers of IL-5+ CD3+CD4+ Th2 cells in the mLN on day 6 were measured. (B) IL-5+IL-13+ CD3+CD4+ Th2 cells were quantified on day 6 after papain stimulation on days 0 and 1 in WT mice (▪) or mice treated with anti-IL-13 mAb ( ). (C) The induction of Th2 cells in WT BMT mice stimulated with papain (▪) was compared to _Rora_sg/sg BMT mice stimulated with papain (□) or papain + rmIL-13 (
). (C) The induction of Th2 cells in WT BMT mice stimulated with papain (▪) was compared to _Rora_sg/sg BMT mice stimulated with papain (□) or papain + rmIL-13 ( ). The total numbers of IL-5+IL-13+ CD3+CD4+ Th2 cells in the mLN were quantified on day 6. (D) CFSE-labeled CD4+ OT-II cells were transplanted into WT and _Rora_sg/sg BMT mice, followed by treatment with papain + OVA. On day 6, mLN cells were analyzed by flow cytometry for CD4+TCRVβ5+CFSE+ OT-II T cell proliferation and intracellular IL-4 and IL-13. (E) WT (▪) and _Rora_sg/sg (□) were treated as in (D). Some of the latter mice also received rmIL-13 injection (
). The total numbers of IL-5+IL-13+ CD3+CD4+ Th2 cells in the mLN were quantified on day 6. (D) CFSE-labeled CD4+ OT-II cells were transplanted into WT and _Rora_sg/sg BMT mice, followed by treatment with papain + OVA. On day 6, mLN cells were analyzed by flow cytometry for CD4+TCRVβ5+CFSE+ OT-II T cell proliferation and intracellular IL-4 and IL-13. (E) WT (▪) and _Rora_sg/sg (□) were treated as in (D). Some of the latter mice also received rmIL-13 injection ( ). OT-II T cells positively stained for intracellular IL-4 or IL-13 were quantified and the total numbers of cytokine positive OT-II in the mLN were calculated. (F) _Rora_sg/sg BMT mice were transplanted with CFSE-labeled CD4+ OT-II cells plus none, WT, or Il13 −/− ILC2s. These mice were treated on days 0 and 1 with papain, and CD3+CD4+TCRVβ5+CFSE+ OT-II T cells in the mLN were analyzed for intracellular IL-4 and IL-13 (plots) on day 6. The total numbers of IL-4+IL-13+ OT-II T cells in the mLN were calculated (bar graph). (G) CFSE-labeled CD4+ OT-II cells were transplanted into WT mice. The mice received intranasal administration of OVA + rmIL-13, OVA alone, or OVA + papain on days 0 and 1. CD3+CD4+TCRVβ5+CFSE+ cells were gated (data not shown) and analyzed for intracellular IL-4 and IL-13 on day 6 (plots). The total numbers of IL-4+IL-13+ OT-II T cells in the mLN were calculated (bar graph). Data are representative of at least three independent experiments. Mean ± SEM in (A)–(C) and (E)–(G), mean percent gated in (D), (F), and (G), ∗ = p ≤ 0.05 ∗∗ = p ≤ 0.01, ns = not significant (two-tailed Student’s t test). See also Figure S5.
). OT-II T cells positively stained for intracellular IL-4 or IL-13 were quantified and the total numbers of cytokine positive OT-II in the mLN were calculated. (F) _Rora_sg/sg BMT mice were transplanted with CFSE-labeled CD4+ OT-II cells plus none, WT, or Il13 −/− ILC2s. These mice were treated on days 0 and 1 with papain, and CD3+CD4+TCRVβ5+CFSE+ OT-II T cells in the mLN were analyzed for intracellular IL-4 and IL-13 (plots) on day 6. The total numbers of IL-4+IL-13+ OT-II T cells in the mLN were calculated (bar graph). (G) CFSE-labeled CD4+ OT-II cells were transplanted into WT mice. The mice received intranasal administration of OVA + rmIL-13, OVA alone, or OVA + papain on days 0 and 1. CD3+CD4+TCRVβ5+CFSE+ cells were gated (data not shown) and analyzed for intracellular IL-4 and IL-13 on day 6 (plots). The total numbers of IL-4+IL-13+ OT-II T cells in the mLN were calculated (bar graph). Data are representative of at least three independent experiments. Mean ± SEM in (A)–(C) and (E)–(G), mean percent gated in (D), (F), and (G), ∗ = p ≤ 0.05 ∗∗ = p ≤ 0.01, ns = not significant (two-tailed Student’s t test). See also Figure S5.
Figure 6
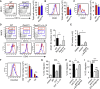
ILC2-Derived IL-13 Promoted DC Migration to the Draining LN (A–C) Lungs from WT (red) and _Rora_sg/sg (blue) BMT mice treated on days 0 and 1 with papain were analyzed on day 3 for DC activation. CD11c+MHCIIhi activated DCs were detected by flow cytometry and the total numbers in the lung calculated (A). Activated DCs in (A) were analyzed for CD40 (B) and CCR7 (C) expression. The red histograms represent WT BMT DCs, the blue histograms represent _Rora_sg/sg BMT and the solid gray histograms show the FMO control. Bar graphs are color-coded in the same way show the total numbers of DCs in the lung. (D) DCs in the mLN of WT and _Rora_sg/sg BMT mice (±rmIL-13 injection) were analyzed on day 3. Activated CD11c+MHCIIhi DCs gated by red boxes and nonactivated DCs gated by blue boxes (top plots) were analyzed for CD40 expression (bottom histograms, color-coded in the same way). Number in histogram shows the percent CD40+CD11c+ cells. Absolute numbers of CD11c+MHCIIhi CD40+ cells in mLN were calculated (bar graph). (E) WT mice treated with papain (▪), papain + anti-IL-13 mAb ( ), or control (□) were analyzed for absolute numbers of activated DCs in mLNs on day 3 as in (D). (F) Traceable DQ-OVA was injected together with papain into WT (red) or _Rora_sg/sg (blue) BMT mice, followed by an analysis for DQ-positive CD11c+MHCIIhi DCs in the lung and mLN on day 3 (histograms), and the total numbers of DQ+ DCs were calculated (bar graph). (G) Lung tissue explants were made from papain-stimulated WT, Il13 −/−, or Il13 −/− + rmIL-13 mice on day 3. Tissue explants were cultured in transwell inserts and exposed to a CCL21 chemokine gradient for 14 hr. WT lung explants without a CCL21 gradient were used as control. The total number of activated DCs was counted in both top and bottom compartments by flow cytometry, followed by a calculation of percent DC migration. (H) Lung tissue explants from naive WT mice were stimulated ex vivo with papain for 14 hr in the presence of anti-IL-13 neutralizing antibody ± EP4-agonist or PBS control. The percent of DC migration toward a CCL21 chemokine gradient was calculated. (I) Lung explants were prepared, treated with papain in the presence of PBS (control), anti-IL-13 or EP4 antagonist, and DC migration was analyzed as in (H). Data are representative of at least three independent experiments. Mean ± SEM in (A)–(I), mean percent gated in (A) and (D), ns = not significant ∗ = p ≤ 0.05, ∗∗ = p ≤ 0.01 ∗∗∗ = p ≤ 0.001 (two-tailed Student’s t test). See also Figure S6.
), or control (□) were analyzed for absolute numbers of activated DCs in mLNs on day 3 as in (D). (F) Traceable DQ-OVA was injected together with papain into WT (red) or _Rora_sg/sg (blue) BMT mice, followed by an analysis for DQ-positive CD11c+MHCIIhi DCs in the lung and mLN on day 3 (histograms), and the total numbers of DQ+ DCs were calculated (bar graph). (G) Lung tissue explants were made from papain-stimulated WT, Il13 −/−, or Il13 −/− + rmIL-13 mice on day 3. Tissue explants were cultured in transwell inserts and exposed to a CCL21 chemokine gradient for 14 hr. WT lung explants without a CCL21 gradient were used as control. The total number of activated DCs was counted in both top and bottom compartments by flow cytometry, followed by a calculation of percent DC migration. (H) Lung tissue explants from naive WT mice were stimulated ex vivo with papain for 14 hr in the presence of anti-IL-13 neutralizing antibody ± EP4-agonist or PBS control. The percent of DC migration toward a CCL21 chemokine gradient was calculated. (I) Lung explants were prepared, treated with papain in the presence of PBS (control), anti-IL-13 or EP4 antagonist, and DC migration was analyzed as in (H). Data are representative of at least three independent experiments. Mean ± SEM in (A)–(I), mean percent gated in (A) and (D), ns = not significant ∗ = p ≤ 0.05, ∗∗ = p ≤ 0.01 ∗∗∗ = p ≤ 0.001 (two-tailed Student’s t test). See also Figure S6.
Comment in
- Innate immune cells to the help.
Lambrecht BN, Hammad H. Lambrecht BN, et al. Immunity. 2014 Mar 20;40(3):313-4. doi: 10.1016/j.immuni.2014.02.010. Immunity. 2014. PMID: 24656044 - Innate lymphoid cells: breathing into allergic inflammation.
Kugelberg E. Kugelberg E. Nat Rev Immunol. 2014 May;14(5):281. doi: 10.1038/nri3668. Epub 2014 Apr 11. Nat Rev Immunol. 2014. PMID: 24722478 No abstract available.
Similar articles
- Intrinsic functional defects of type 2 innate lymphoid cells impair innate allergic inflammation in promyelocytic leukemia zinc finger (PLZF)-deficient mice.
Verhoef PA, Constantinides MG, McDonald BD, Urban JF Jr, Sperling AI, Bendelac A. Verhoef PA, et al. J Allergy Clin Immunol. 2016 Feb;137(2):591-600.e1. doi: 10.1016/j.jaci.2015.07.050. Epub 2015 Oct 23. J Allergy Clin Immunol. 2016. PMID: 26602165 Free PMC article. - Group 2 innate lymphoid cells facilitate sensitization to local, but not systemic, TH2-inducing allergen exposures.
Gold MJ, Antignano F, Halim TY, Hirota JA, Blanchet MR, Zaph C, Takei F, McNagny KM. Gold MJ, et al. J Allergy Clin Immunol. 2014 Apr;133(4):1142-8. doi: 10.1016/j.jaci.2014.02.033. J Allergy Clin Immunol. 2014. PMID: 24679471 - Innate and adaptive type 2 immunity in lung allergic inflammation.
Kubo M. Kubo M. Immunol Rev. 2017 Jul;278(1):162-172. doi: 10.1111/imr.12557. Immunol Rev. 2017. PMID: 28658559 Review. - Fibrinogen cleavage products and Toll-like receptor 4 promote the generation of programmed cell death 1 ligand 2-positive dendritic cells in allergic asthma.
Cho M, Lee JE, Lim H, Shin HW, Khalmuratova R, Choi G, Kim HS, Choi WS, Park YJ, Shim I, Kim BS, Kang CY, Kim JO, Tanaka S, Kubo M, Tung HY, Landers CT, Corry DB, Kheradmand F, Chung Y. Cho M, et al. J Allergy Clin Immunol. 2018 Aug;142(2):530-541.e6. doi: 10.1016/j.jaci.2017.09.019. Epub 2017 Oct 14. J Allergy Clin Immunol. 2018. PMID: 29038008 - Lung ILC2s link innate and adaptive responses in allergic inflammation.
Martinez-Gonzalez I, Steer CA, Takei F. Martinez-Gonzalez I, et al. Trends Immunol. 2015 Mar;36(3):189-95. doi: 10.1016/j.it.2015.01.005. Epub 2015 Feb 19. Trends Immunol. 2015. PMID: 25704560 Review.
Cited by
- Immunologic aspects of asthma: from molecular mechanisms to disease pathophysiology and clinical translation.
Xie C, Yang J, Gul A, Li Y, Zhang R, Yalikun M, Lv X, Lin Y, Luo Q, Gao H. Xie C, et al. Front Immunol. 2024 Oct 8;15:1478624. doi: 10.3389/fimmu.2024.1478624. eCollection 2024. Front Immunol. 2024. PMID: 39439788 Free PMC article. Review. - Peri-alloHCT IL-33 administration expands recipient T-regulatory cells that protect mice against acute GVHD.
Matta BM, Reichenbach DK, Zhang X, Mathews L, Koehn BH, Dwyer GK, Lott JM, Uhl FM, Pfeifer D, Feser CJ, Smith MJ, Liu Q, Zeiser R, Blazar BR, Turnquist HR. Matta BM, et al. Blood. 2016 Jul 21;128(3):427-39. doi: 10.1182/blood-2015-12-684142. Epub 2016 May 24. Blood. 2016. PMID: 27222477 Free PMC article. - ILC2s and fungal allergy.
Kita H. Kita H. Allergol Int. 2015 Jul;64(3):219-26. doi: 10.1016/j.alit.2015.04.004. Epub 2015 May 18. Allergol Int. 2015. PMID: 26117252 Free PMC article. Review. - Dendritic cells in lung immunopathology.
Cook PC, MacDonald AS. Cook PC, et al. Semin Immunopathol. 2016 Jul;38(4):449-60. doi: 10.1007/s00281-016-0571-3. Epub 2016 Jun 2. Semin Immunopathol. 2016. PMID: 27256370 Free PMC article. Review. - The interplay between innate lymphoid cells and T cells.
Cherrier M, Ramachandran G, Golub R. Cherrier M, et al. Mucosal Immunol. 2020 Sep;13(5):732-742. doi: 10.1038/s41385-020-0320-8. Epub 2020 Jul 10. Mucosal Immunol. 2020. PMID: 32651476 Review.
References
- Barlow J.L., Bellosi A., Hardman C.S., Drynan L.F., Wong S.H. Innate IL-13-producing nuocytes arise during allergic lung inflammation and contribute to airways hyperreactivity. J. Allergy Clin. Immunol. 2012;129 191–8.e1–4. - PubMed
- Besnard A.-G., Togbe D., Guillou N., Erard F., Quesniaux V., Ryffel B. IL-33-activated dendritic cells are critical for allergic airway inflammation. Eur. J. Immunol. 2011;41:1675–1686. - PubMed
- Bromley S.K., Mempel T.R., Luster A.D. Orchestrating the orchestrators: chemokines in control of T cell traffic. Nat. Immunol. 2008;9:970–980. - PubMed
- Finkelman F.D., Katona I.M., Urban J.F., Jr., Holmes J., Ohara J., Tung A.S., Sample J.V., Paul W.E. IL-4 is required to generate and sustain in vivo IgE responses. J. Immunol. 1988;141:2335–2341. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials