Association among weight change, glycemic control, and markers of cardiovascular risk with exenatide once weekly: a pooled analysis of patients with type 2 diabetes - PubMed (original) (raw)
Randomized Controlled Trial
Association among weight change, glycemic control, and markers of cardiovascular risk with exenatide once weekly: a pooled analysis of patients with type 2 diabetes
Lawrence Blonde et al. Cardiovasc Diabetol. 2015.
Abstract
Background: Overweight or obesity contributes to the development of type 2 diabetes mellitus (T2DM) and increases cardiovascular risk. Exenatide, a glucagon-like peptide-1 receptor agonist, significantly reduces glycated hemoglobin (A1C) and body weight and improves cardiovascular risk markers in patients with T2DM. As weight loss alone has been shown to reduce A1C and cardiovascular risk markers, this analysis explored whether weight loss contributed importantly to clinical responses to exenatide once weekly.
Methods: A pooled analysis from eight studies of exenatide once weekly was conducted. Patients were distributed into quartiles from greatest weight loss (Quartile 1) to least loss or gain (Quartile 4). Parameters evaluated for each quartile included A1C, fasting plasma glucose (FPG), blood pressure (BP), heart rate, high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), total cholesterol, triglycerides, and the liver enzymes alanine aminotransferase (ALT) and aspartate aminotransferase (AST).
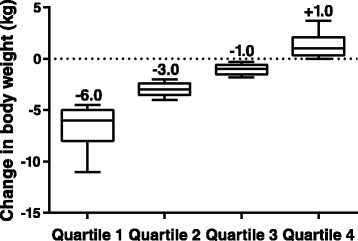
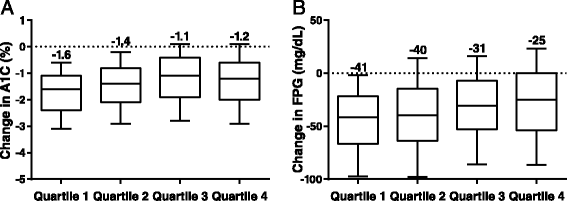
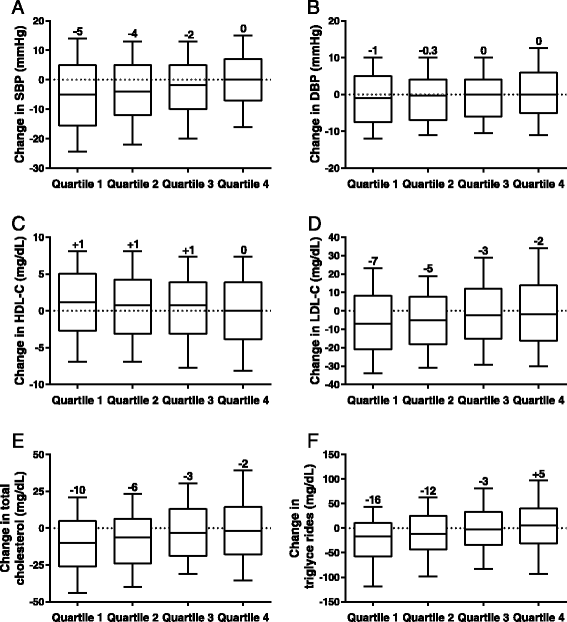
Results: The median changes from baseline in body weight in Quartiles 1-4 were -6.0, -3.0, -1.0, and +1.0 kg, respectively. All quartiles had reductions in A1C (median changes -1.6, -1.4, -1.1, and -1.2%, respectively) and FPG (-41, -40, -31, and -25 mg/dL, respectively), with the greatest decreases in Quartiles 1 and 2. Most cardiovascular risk markers (except diastolic BP) and liver enzymes improved in Quartiles 1 through 3 and were relatively unchanged in Quartile 4. Higher rates of gastrointestinal adverse events and hypoglycemia were observed in Quartile 1 compared with Quartiles 2 through 4.
Conclusions: Exenatide once weekly improved glycemic parameters independent of weight change, although the magnitude of improvement increased with increasing weight loss. The greatest trend of improvement in glycemic parameters, cardiovascular risk factors including systolic BP, LDL-C, total cholesterol, and triglycerides, and in liver enzymes, was seen in the patient quartiles with the greatest reductions in body weight.
Figures
Figure 1
Distribution of median change in body weight, by end-point body weight change quartiles. The central line represents the median (also labelled above the plot), the box encloses the 25th to 75th percentiles of the distribution, and the outer bars are drawn to the 10th and 90th percentiles.
Figure 2
Median changes in A1C and FPG, by end-point body weight change quartiles. (A) A1C. (B) FPG. A1C, hemoglobin A1C; FPG, fasting plasma glucose. The central line represents the median (also labelled above the plot), the box encloses the 25th to 75th percentiles of the distribution, and the outer bars are drawn to the 10th and 90th percentiles.
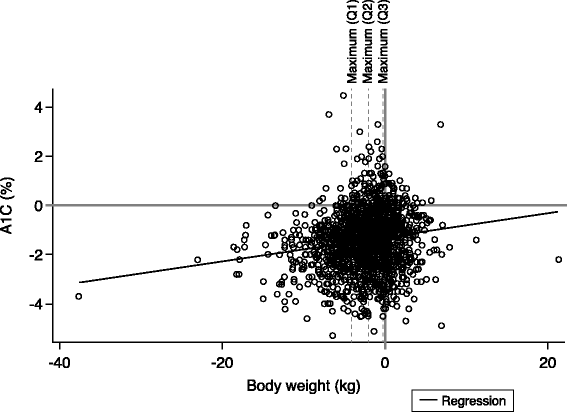
Figure 3
Pearson linear correlation of change in body weight and change in A1C. The solid black line represents the regression. Dotted lines represent the maximum body weight change in Quartiles 1 through 3. The r-value for all patients was 0.1579, and r-values for Quartiles 1 through 4 were 0.1847, 0.0267, 0.0485, and −0.0542, respectively. A1C, hemoglobin A1C; Q, quartile.
Figure 4
Median changes in blood pressure and lipids, by end-point body weight change quartiles. (A) SBP. (B) DBP. (C) HDL-C. (D) LDL-C. (E) Total cholesterol. (F) Triglycerides. DBP, diastolic blood pressure; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; SBP, systolic blood pressure. The central line represents the median (also labelled above the plot), the box encloses the 25th to 75th percentiles of the distribution, and the outer bars are drawn to the 10th and 90th percentiles.
Figure 5
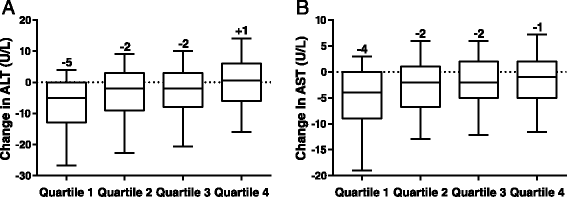
Median changes in ALT and AST, by end-point body weight change quartiles. (A) ALT. (B) AST. ALT, alanine aminotransferase; AST, aspartate aminotransferase. The central line represents the median (also labelled above the plot), the box encloses the 25th to 75th percentiles of the distribution, and the outer bars are drawn to the 10th and 90th percentiles.
Similar articles
- Long-term changes in cardiovascular risk markers during administration of exenatide twice daily or glimepiride: results from the European exenatide study.
Simó R, Guerci B, Schernthaner G, Gallwitz B, Rosas-Guzmàn J, Dotta F, Festa A, Zhou M, Kiljański J. Simó R, et al. Cardiovasc Diabetol. 2015 Sep 4;14:116. doi: 10.1186/s12933-015-0279-z. Cardiovasc Diabetol. 2015. PMID: 26338040 Free PMC article. Clinical Trial. - Exenatide once weekly improved glycaemic control, cardiometabolic risk factors and a composite index of an HbA1c < 7%, without weight gain or hypoglycaemia, over 52 weeks.
Bergenstal RM, Li Y, Porter TK, Weaver C, Han J. Bergenstal RM, et al. Diabetes Obes Metab. 2013 Mar;15(3):264-71. doi: 10.1111/dom.12026. Epub 2012 Nov 12. Diabetes Obes Metab. 2013. PMID: 23078638 Free PMC article. - Efficacy, safety, and tolerability of exenatide once weekly in patients with type 2 diabetes mellitus: an integrated analysis of the DURATION trials.
Grimm M, Han J, Weaver C, Griffin P, Schulteis CT, Dong H, Malloy J. Grimm M, et al. Postgrad Med. 2013 May;125(3):47-57. doi: 10.3810/pgm.2013.05.2660. Postgrad Med. 2013. PMID: 23748506 Clinical Trial. - Clinical implications of exenatide as a twice-daily or once-weekly therapy for type 2 diabetes.
Aroda VR, DeYoung MB. Aroda VR, et al. Postgrad Med. 2011 Sep;123(5):228-38. doi: 10.3810/pgm.2011.09.2479. Postgrad Med. 2011. PMID: 21904106 Review. - Pharmacokinetic drug evaluation of exenatide for the treatment of type 2 diabetes.
Molina Vega M, Muñoz-Garach A, Tinahones FJ. Molina Vega M, et al. Expert Opin Drug Metab Toxicol. 2018 Feb;14(2):207-217. doi: 10.1080/17425255.2018.1420160. Epub 2017 Dec 28. Expert Opin Drug Metab Toxicol. 2018. PMID: 29260924 Review.
Cited by
- Long-term changes in cardiovascular risk markers during administration of exenatide twice daily or glimepiride: results from the European exenatide study.
Simó R, Guerci B, Schernthaner G, Gallwitz B, Rosas-Guzmàn J, Dotta F, Festa A, Zhou M, Kiljański J. Simó R, et al. Cardiovasc Diabetol. 2015 Sep 4;14:116. doi: 10.1186/s12933-015-0279-z. Cardiovasc Diabetol. 2015. PMID: 26338040 Free PMC article. Clinical Trial. - A Review of the Long-Term Efficacy, Tolerability, and Safety of Exenatide Once Weekly for Type 2 Diabetes.
Genovese S, Mannucci E, Ceriello A. Genovese S, et al. Adv Ther. 2017 Aug;34(8):1791-1814. doi: 10.1007/s12325-017-0499-6. Epub 2017 Jul 3. Adv Ther. 2017. PMID: 28674957 Free PMC article. Review. - Rivaroxaban, a factor Xa inhibitor, improves neovascularization in the ischemic hindlimb of streptozotocin-induced diabetic mice.
Wu TC, Chan JS, Lee CY, Leu HB, Huang PH, Chen JS, Lin SJ, Chen JW. Wu TC, et al. Cardiovasc Diabetol. 2015 Jun 16;14:81. doi: 10.1186/s12933-015-0243-y. Cardiovasc Diabetol. 2015. PMID: 26077117 Free PMC article. - Changes in body weight and cardiovascular risk factors in a Chinese population with type 2 diabetes mellitus: a longitudinal study.
Li YY, Yang YM, Zhu S, Cheng H, Hernandez J, Huang W, Wang HHX, Li YT. Li YY, et al. Front Endocrinol (Lausanne). 2023 Apr 12;14:1112855. doi: 10.3389/fendo.2023.1112855. eCollection 2023. Front Endocrinol (Lausanne). 2023. PMID: 37124734 Free PMC article. - Glucagon-like peptide-1 receptor agonists: a review from a cardiovascular perspective.
Turkistani Y. Turkistani Y. Front Cardiovasc Med. 2025 Apr 24;12:1535134. doi: 10.3389/fcvm.2025.1535134. eCollection 2025. Front Cardiovasc Med. 2025. PMID: 40342976 Free PMC article. Review.
References
- Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Blaha MJ, Dai S, Ford ES, Fox CS, Franco S, Fullerton HJ, Gillespie C, Hailpern SM, Heit JA, Howard VJ, Huffman MD, Judd SE, Kissela BM, Kittner SJ, Lackland DT, Lichtman JH, Lisabeth LD, Mackey RH, Magid DJ, Marcus GM, Marelli A, Matchar DB, McGuire DK, Mohler ER, 3rd, Moy CS, et al. American Heart Association Statistics Committee and Stroke Statistics Subcommittee: Heart disease and stroke statistics–2014 update: a report from the American Heart Association. Circulation. 2014;129(3):e28–292. doi: 10.1161/01.cir.0000441139.02102.80. - DOI - PMC - PubMed
- Jung HS, Chang Y, Yun KE, Kim CW, Choi ES, Kwon MJ, Cho J, Zhang Y, Rampal S, Zhao D, Soo Kim H, Shin H, Guallar E, Ryu S. Impact of body mass index, metabolic health, and weight change on incident diabetes in a Korean population. Obesity (Silver Spring) 2014;22(8):1880–7. doi: 10.1002/oby.20751. - DOI - PubMed
- Garber AJ, Abrahamson MJ, Barzilay JI, Blonde L, Bloomgarden ZT, Bush MA, Dagogo-Jack S, Davidson MB, Einhorn D, Garvey WT, Grunberger G, Handelsman Y, Hirsch IB, Jellinger PS, McGill JB, Mechanick JI, Rosenblit PD, Umpierrez G, Davidson MH, American Association of Clinical Endocrinologists AACE comprehensive diabetes management algorithm 2013. Endocr Pract. 2013;19(2):327–36. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials