High rates of o'nyong nyong and Chikungunya virus transmission in coastal Kenya - PubMed (original) (raw)
. 2015 Feb 6;9(2):e0003436.
doi: 10.1371/journal.pntd.0003436. eCollection 2015 Feb.
Tamara Banda 2, Julie Brichard 2, Eric M Muchiri 3, Peter L Mungai 4, Francis M Mutuku 5, Erin Borland 6, Ginny Gildengorin 2, Sarah Pfeil 2, Crystal Y Teng 2, Kristin Long 7, Mark Heise 7, Ann M Powers 6, Uriel Kitron 8, Charles H King 4
Affiliations
- PMID: 25658762
- PMCID: PMC4319898
- DOI: 10.1371/journal.pntd.0003436
High rates of o'nyong nyong and Chikungunya virus transmission in coastal Kenya
A Desiree LaBeaud et al. PLoS Negl Trop Dis. 2015.
Erratum in
- Correction: High rates of o'nyong nyong and Chikungunya virus transmission in coastal Kenya.
PLOS Neglected Tropical Diseases Staff. PLOS Neglected Tropical Diseases Staff. PLoS Negl Trop Dis. 2015 Apr 7;9(4):e0003674. doi: 10.1371/journal.pntd.0003674. eCollection 2015 Apr. PLoS Negl Trop Dis. 2015. PMID: 25849863 Free PMC article. No abstract available.
Abstract
Background: Chikungunya virus (CHIKV) and o'nyong nyong virus (ONNV) are mosquito-borne alphaviruses endemic in East Africa that cause acute febrile illness and arthritis. The objectives of this study were to measure the seroprevalence of CHIKV and ONNV in coastal Kenya and link it to demographics and other risk factors.
Methodology: Demographic and exposure questionnaires were administered to 1,848 participants recruited from two village clusters (Milalani-Nganja and Vuga) in 2009. Sera were tested for alphavirus exposure using standardized CHIKV IgG ELISA protocols and confirmed with plaque reduction neutralization tests (PRNT). Logistic regression models were used to determine the variables associated with seropositivity. Weighted K test for global clustering of houses with alphavirus positive participants was performed for distance ranges of 50-1,000 meters, and G* statistic and kernel density mapping were used to identify locations of higher seroprevalence.
Principal findings: 486 (26%) participants were seropositive by IgG ELISA. Of 443 PRNT confirmed positives, 25 samples (6%) were CHIKV+, 250 samples (56%) were ONNV+, and 168 samples (38%) had high titers for both. Age was significantly associated with seropositivity (OR 1.01 per year, 95% C.I. 1.00-1.01); however, younger adults were more likely to be seropositive than older adults. Males were less likely to be seropositive (p<0.05; OR 0.79, 95% C.I. 0.64-0.97). Adults who owned a bicycle (p<0.05; OR 1.37, 95% C.I. 1.00-1.85) or motor vehicle (p<0.05; OR 4.64, 95% C.I. 1.19-18.05) were more likely to be seropositive. Spatial analysis demonstrated hotspots of transmission within each village and clustering among local households in Milalani-Nganja, peaking at the 200-500m range.
Conclusions/significance: Alphavirus exposure, particularly ONNV exposure, is common in coastal Kenya with ongoing interepidemic transmission of both ONNV and CHIKV. Women and adults were more likely to be seropositive. Household location may be a defining factor for the ecology of alphaviral transmission in this region.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
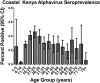
Figure 1. Alphavirus seroprevalence as measured by ELISA displayed by village and age group.
95% CI indicated by bars.
Figure 2. Alphavirus seroprevalence as measured by ELISA displayed by age.
95% CI indicated by bars.
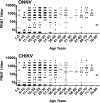
Figure 3. Plaque reduction neutralization titer results by age.
ONNV (top) and CHIKV (bottom) titers by 5 year age group. Geometric means indicated by horizontal bars.
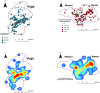
Figure 4. Analytic maps of alphavirus seropositivity in Milalani-Nganja (right) and Vuga (left).
The top panels show household locations as dots proportional to the household density of seropositives; the bottom panels show the kernel density estimates for alphaviral seropositivity as a color range, with red having the highest vales and blue the lowest values.
Figure 5. G*-statistic maps of Vuga (top) and Milalani-Nganja (bottom) alphavirus clustering.
Clusters of low values are demonstrated in blue circles and clusters of high values are in red circles.
References
- Powers AM, Brault AC, Tesh RB and Weaver SC. (2000) Re-emergence of Chikungunya and O’nyong-nyong viruses: evidence for distinct geographical lineages and distant evolutionary relationships. J Gen Virol 81:471–479. -PubMed
- Pan American Health Organization (Paho) (2014) Chikungunya, Statistics: Number of reported cases of Chikungunya fever in the Americas. Available: http://www.paho.org/hq/index.php?option=com_content&view=article&id=9053...
- Haddow A.J., Davies C.W., Walker A.J.. (1960) O’nyong nyong fever: an epidemic virus disease in West Africa: Introduction. Trans. R. Soc. Trop. Med. Hyg., 54: 517–522. 10.1016/0035-9203(60)90025-0 -DOI
Publication types
MeSH terms
Grants and funding
- U54 AI081680/AI/NIAID NIH HHS/United States
- NIH AI102918/AI/NIAID NIH HHS/United States
- R01 AI102918/AI/NIAID NIH HHS/United States
- AI81680:RP012/AI/NIAID NIH HHS/United States
- R01 TW008067/TW/FIC NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical